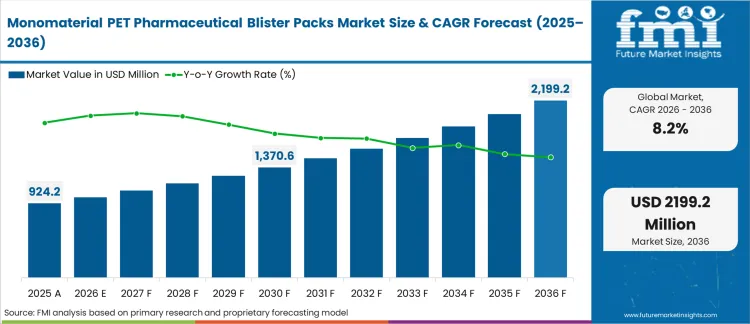
The monomaterial PET pharmaceutical blister packs market crossed a valuation of USD 920.0 million in 2025. Industry is expected to surpass USD 1,000.0 million in 2026 at a CAGR of 8.20% during the forecast. Demand outlook carries the valuation to USD 2,200.0 million through 2036 as pharmaceutical manufacturers qualify fully recyclable mono-PET films on existing form-fill-seal lines to meet extended producer responsibility obligations without reformulating solid-dose products.
Formulation scientists and packaging engineers executing new product launches now reject multi-layer laminates that complicate mechanical recycling streams. Contract development and manufacturing organizations must integrate virgin and recycled PET grades into every new blister specification to satisfy downstream EPR audits. Suppliers entering 2027 tender cycles without verified mono-material compatibility face a disqualification window they cannot close after shortlisting begins.
Branded pharmaceutical procurement teams face mounting pressure to abandon legacy PVC-aluminum formats and commit capital to converged mono-PET tooling. The underlying mechanism sustaining market growth is the absolute necessity of maintaining moisture and oxygen barriers while enabling closed-loop recycling, a capability legacy laminates physically cannot deliver at scale. The geographic distribution of this architectural migration reveals distinct adoption velocities tied directly to national circular-economy policies.
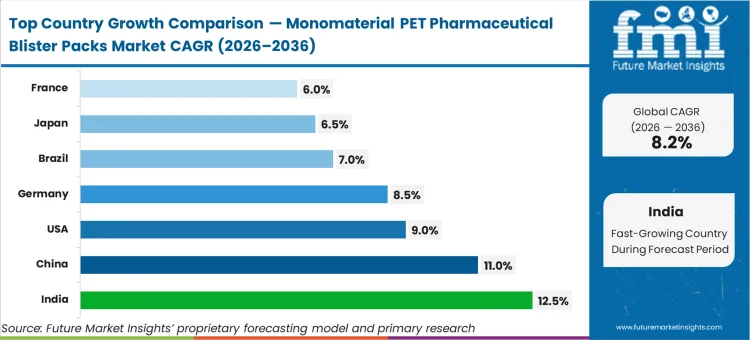
As regional pharmaceutical hubs navigate this transition, India sets the pace with a 12.5% CAGR, followed closely by China at 11.0% as both nations leverage greenfield CDMO facilities to bypass legacy retrofit hurdles. The United States advances at a 9.0% rate, supported by federal EPR frameworks. Germany registers an 8.5% expansion while Brazil tracks at 7.0%, both driven by export compliance to EU markets. Japan expands at a 6.5% trajectory anchored by its domestic generics sector. France maintains a steady 6.0% pace as energy-intensive blister lines implement synchronized mono-PET conversion.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1,000.0 million |
| Industry Value (2036) | USD 2,200.0 million |
| CAGR (2026-2036) | 8.20% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
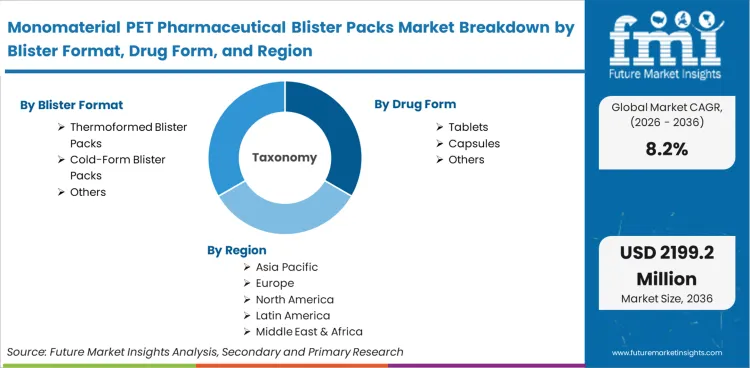
Monomaterial PET Pharmaceutical Blister Packs represent single-polymer primary packaging solutions engineered from polyethylene terephthalate film and lidding that deliver guaranteed moisture and oxygen protection for solid oral dosage forms while enabling full mechanical recyclability in existing PET streams. The ecosystem includes thermoformed cavities, cold-formed structures, and compatible heat-seal lidding compliant with pharmacopeial stability requirements. A core inclusion is film supporting high-barrier coatings without additional polymer layers.
Market scope includes pharmaceutical-grade mono-PET base films, preformed blister trays, lidding foils with PET-compatible heat-seal coatings, and integrated form-fill-seal consumables featuring native recyclability. Configuration tools for cavity design, inline barrier coating applicators, and associated stability-testing protocols fall within the boundaries. Products utilizing 100% PET structures for critical solid-dose protection are fully incorporated. For deeper insights into related blister packaging solutions see the blister packaging solutions.
Multi-material laminates containing PVC, PVDC, or aluminum layers are excluded because they disrupt mechanical recycling streams and require separate sorting infrastructure. Pure secondary cartons, non-pharmaceutical consumer blister formats, and liquid or injectable primary packaging fall outside the defined parameters. Legacy non-PET films without verified mono-material certification are explicitly omitted from the valuation.
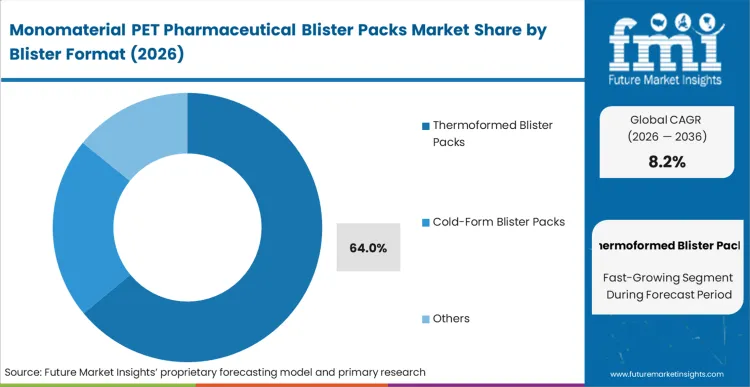
With legacy multi-layer laminates proving inadequate for closed-loop recyclability, capital projects directors are executing full-scale tooling replacement programs across pharmaceutical packaging lines. Thermoformed Blister Packs command a dominant 64.0% share in 2026, reflecting the large installed base of form-fill-seal infrastructure already capable of PET conversion with targeted process adjustments. Pharmaceutical manufacturers specifying mono-PET thermoformed cavities establish the practical foundation for recyclable primary packaging without forcing immediate reformulation of solid-dose products. This material-level transition reduces conversion complexity and allows packaging engineers to retain optical clarity, forming precision, and line efficiency within existing production environments. According to FMI’s estimates, facilities qualifying thermoformed mono-PET structures can shorten sustainability transition timelines while preserving compatibility with high-volume pharmaceutical packaging operations. Suppliers unable to provide verified thermoformable PET structures with acceptable barrier performance risk immediate exclusion from the next wave of branded and generic blister conversion tenders.
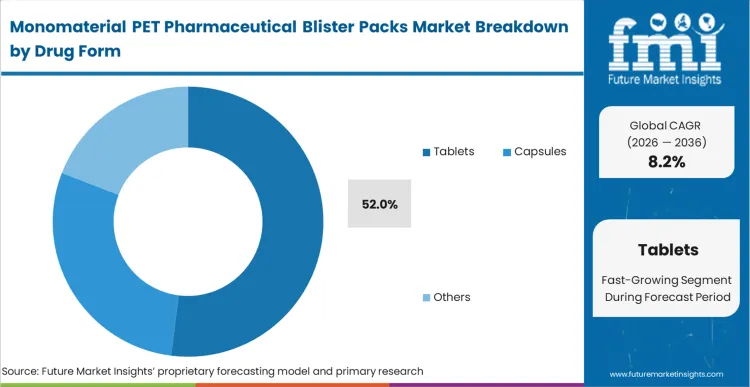
Tablets emerge as the dominant drug form, expected to represent 52.0% of total market share in 2026. High-volume solid oral dosage manufacturing increasingly depends on packaging formats that can combine unit-dose protection with verifiable recyclability performance. FMI analysts opine that tablet packaging provides the most scalable entry point for mono-PET adoption because tablet stability profiles, dispensing formats, and retail distribution models already align closely with standardized blister architectures. Packaging teams qualifying mono-PET formats for tablet applications can accelerate commercialization programs while avoiding the more complex containment challenges associated with certain alternative dosage forms. The widespread concentration of tablet volumes in branded and generic pharmaceutical supply chains further reinforces mono-PET demand across this segment.
The convergence of extended producer responsibility legislation and brand net-zero commitments forces packaging specification leads to extract verifiable recyclability data directly from every blister cavity. This architectural requirement renders fragmented multi-layer structures obsolete. Asset owners upgrading brownfield lines face a strict binary choice between deploying complex separation processes or overhauling the base film. Transitioning to a unified mono-PET backbone simplifies supply-chain logistics and enables direct inline recycling for advanced analytics. Facilities that fail to modernize their core packaging layers risk operational blind spots and reduced overall equipment effectiveness.
The intricate qualification parameters required to optimize mono-PET barrier coatings create steep learning curves for traditional stability scientists. Designing conflict-free stability profiles across multi-vendor tooling demands specialized expertise that most pharmaceutical facilities lack internally. To mitigate this skill gap, capital project directors increasingly rely on advanced simulation software that automates cavity design and validates performance before physical deployment.
Opportunities in the Monomaterial PET Pharmaceutical Blister Packs Market
Based on the regional analysis, the Monomaterial PET Pharmaceutical Blister Packs market is segmented into Asia Pacific, North America, Europe, Latin America, Middle East & Africa across 40+ countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 12.5% |
| China | 11.0% |
| USA | 9.0% |
| Germany | 8.5% |
| Brazil | 7.0% |
| Japan | 6.5% |
| France | 6.0% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Greenfield infrastructure development across the Asia Pacific region accelerates the bypass of legacy multi-layer constraints. Capital project directors constructing new CDMO campuses specify unified mono-PET tooling in their initial site blueprints. FMI analysts opine that this clean-slate approach entirely eliminates the costly and time-consuming retrofit phase that plagues older manufacturing centers. By building native recyclability capabilities into the foundation, regional asset owners establish highly flexible production environments capable of rapid retooling. This aggressive greenfield expansion strategy directly fuels the demand for high-performance mono-PET film. The integration of robust packaging architecture forms the critical prerequisite for deploying advanced stability monitoring applications. The specific manufacturing requirements of individual nations dictate the precise implementation parameters for these packaging technologies.
FMI's report includes extensive coverage of the Asia Pacific mono-PET blister landscape. It incorporates detailed analysis of South Korea, Indonesia, Australia, and the broader ASEAN region. A primary trend shaping these nations is the rapid localization of pharmaceutical assembly, forcing contract manufacturers to deploy unified architectures to satisfy stringent global brand traceability requirements.
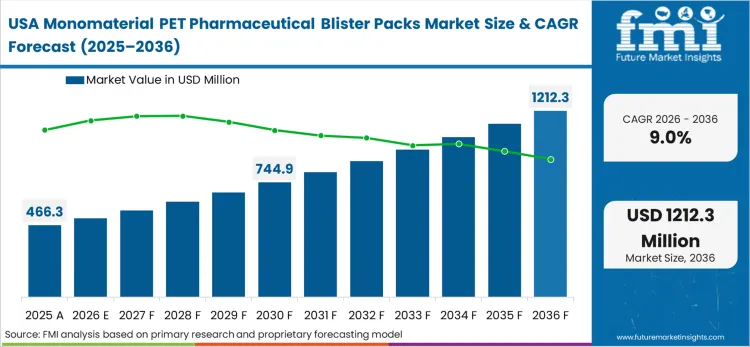
Industrial modernization mandates across North America target the systematic eradication of isolated multi-layer blister networks. Control systems engineers leading critical infrastructure overhauls face strict directives to unify disparate communication domains under a single manageable architecture. According to FMI's estimates, the branded pharmaceutical sectors actively drive this consolidation to secure their supply chain data flows against sophisticated regulatory audits. Implementing a standardized, mono-PET backbone enables facilities to deploy advanced predictive maintenance algorithms without compromising real-time stability. This strategic shift requires significant capital allocation toward robust film hardware capable of isolating mixed-criticality requirements. The success of these modernization initiatives depends entirely on the availability of certified interoperable components. National security frameworks and pharmacopeial standards govern the exact specifications required for these critical deployments.
FMI's report includes comprehensive evaluation of the North American mono-PET blister sector. It features specific analysis of the Canadian and Mexican industrial markets. A defining dynamic in these countries involves the integration of cross-border supply chains, which requires standardized protocols to coordinate just-in-time delivery sequences and maintain synchronized production schedules across multiple facilities.
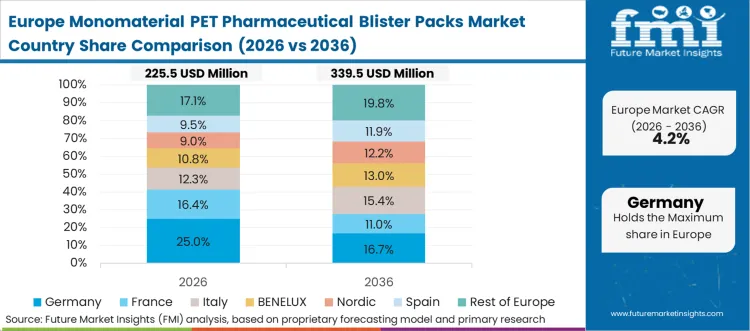
European industrial policy actively penalizes the continued operation of energy-inefficient and technologically isolated manufacturing facilities. Automation architects redesigning legacy production lines must integrate continuous energy monitoring data alongside critical machine control signals. This dual-purpose network requirement forces the rapid adoption of traffic-shaping protocols to guarantee control stability while transmitting heavy diagnostic payloads. The transition requires a complete overhaul of existing communication topologies, shifting from hierarchical polling structures to event-driven deterministic architectures. Upgrading the core switching infrastructure provides the necessary bandwidth and synchronization precision to support advanced sustainable manufacturing practices. Facility directors recognize that maintaining outdated network standards severely limits their ability to compete in highly regulated European markets. Regional engineering standards and safety certifications heavily influence the technical requirements for these network transformations.
FMI's report includes thorough investigation of the European industrial networking framework. The analysis encompasses Italy, Spain, the Nordics, and the Benelux region. A prevailing structural condition across these nations is the mandatory compliance with strict machinery safety directives, forcing asset owners to specify deterministic networks that can reliably transport fail-safe signals alongside standard operational data.
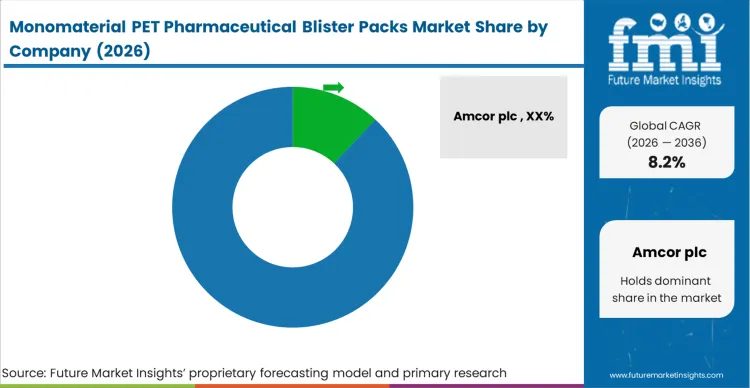
The introduction of standardized testing profiles under the EU Packaging and Packaging Waste Regulation is reshaping how pharmaceutical buyers evaluate blister hardware. Instead of accepting multi-layer timing mechanisms, procurement teams now demand certified, interoperable mono-PET silicon in all new tenders. This shift has raised the performance bar across the industry, meaning vendors that continue to rely on closed, single-vendor ecosystems risk being excluded from major modernization programs.
Industry leaders have already begun adapting. Manufacturers such as Amcor, Huhtamaki, and Klöckner Pentaplast have aligned portions of their portfolios with open, mono-PET networking standards, setting a new competitive baseline for others to follow. As a result, automation suppliers are now pushed to integrate native mono-PET-compliant capabilities directly into their hardware instead of using add-on gateways or proprietary extensions.
Vendors embedding precision barrier coatings and synchronization protocols directly into edge devices gain a significant architectural advantage. Companies like Constantia Flexibles and Tekni-Plex, which increasingly incorporate mono-PET silicon at the device level, help reduce engineering overhead during commissioning and shrink the physical control-cabinet footprint. This silicon-first approach accelerates brownfield migration projects, allowing network architects and project leads to prioritize platforms that avoid the complexity associated with external timing gateways. In contrast, component manufacturers slow to adopt native mono-PET designs risk losing position during early-stage vendor shortlisting. For detailed competitive insights see the pharmaceutical plastic packaging.
| Metric | Value |
|---|---|
| Quantitative Units | USD 1,000.0 million to USD 2,200.0 million, at a CAGR of 8.20% |
| Market Definition | Monomaterial PET Pharmaceutical Blister Packs encompass specialized PET films and lidding built to guarantee bounded moisture and oxygen protection for solid oral dosage forms while enabling full recyclability in standard PET streams. |
| Blister Format Segmentation | Thermoformed Blister Packs, Cold-Form Blister Packs, Others |
| Drug Form Segmentation | Tablets, Capsules, Others |
| End Use Segmentation | Branded Pharmaceuticals, Generic Pharmaceuticals, Others |
| Barrier Requirement Segmentation | High Barrier, Standard Barrier, Others |
| Regions Covered | Asia Pacific, North America, Europe, Latin America, Middle East & Africa |
| Countries Covered | India, China, USA, Germany, Brazil, Japan, France, and 40 plus countries |
| Key Companies Profiled | Amcor plc, Huhtamaki, Klöckner Pentaplast, Constantia Flexibles, Tekni-Plex, Inc., Berry Global Inc., Sonoco Products Company |
| Forecast Period | 2026 to 2036 |
| Approach | The baseline value derives from a bottom-up aggregation of mono-PET film shipments, applying region-specific sustainability adoption curves to project future conversion velocity. Projections undergo cross-validation against quarterly PET resin shipment volumes for pharmaceutical-grade film. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the demand for Monomaterial PET Pharmaceutical Blister Packs in the global market in 2026?
Demand for Monomaterial PET Pharmaceutical Blister Packs in the global market is estimated to be valued at USD 1,000.0 million in 2026 as branded manufacturers accelerate qualification programs to meet impending EPR deadlines.
What will be the market size of Monomaterial PET Pharmaceutical Blister Packs in the global market by 2036?
Market size for Monomaterial PET Pharmaceutical Blister Packs is projected to reach USD 2,200.0 million by 2036 driven by sustained regulatory tailwinds and cost parity improvements in recycled PET supply.
What CAGR is projected for Monomaterial PET Pharmaceutical Blister Packs from 2026 to 2036?
Demand for Monomaterial PET Pharmaceutical Blister Packs is expected to grow at a CAGR of 8.20% between 2026 and 2036 sustained by the structural necessity of maintaining drug stability while enabling closed-loop recycling.
Which Blister Format is poised to lead global sales by 2026?
Thermoformed Blister Packs accounts for 64.0% in 2026 as capital project directors execute full-scale hardware replacement programs to eliminate multi-layer separation steps.
How significant is the role of Tablets in driving Monomaterial PET Pharmaceutical Blister Packs adoption in 2026?
Tablets represent 52.0% of segment share as solid oral dosage forms dominate unit-dose dispensing in both retail and institutional channels.
What is driving demand in India?
India’s pharmaceutical export sector operates under stringent FDA Pre-Approval Inspection frameworks requiring immutable electronic records for all batch production parameters, anchoring 12.5% CAGR through regulatory compliance pressure.
What compliance standards or regulations are referenced for Germany?
The German machine tool export sector operates under rigorous functional safety certifications that mandate unified diagnostic layers, driving 8.5% expansion.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 12.5% during 2026 to 2036 through greenfield CDMO expansion.
Why is Asia Pacific described as a priority region in this report?
The Asia Pacific greenfield CDMO campuses actively drive network consolidation to secure supply chain traceability against sophisticated regulatory audits.
What type of demand dominates in North America?
Demand heavily focuses on replacing vulnerable legacy multi-layer formats with standardized mono-PET backbones that comply with federal EPR mandates.
What is the China growth outlook in this report?
China is projected to expand at a CAGR of 11.0% during 2026 to 2036 driven by massive new capacity buildout.
Does the report cover USA in its regional analysis?
Yes, USA is included within North America under the regional scope of analysis.
What are the sources referred to for analyzing USA?
Federal infrastructure modernization initiatives and pharmacopeial qualification standards form the analytical basis.
What is the main demand theme linked to USA in its region coverage?
Strict cybersecurity and data visibility mandates across multi-tier supply chains force the deployment of converged mono-PET architectures.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe under the regional coverage framework.
What is the main Germany related demand theme in its region coverage?
Stringent validation criteria for mixed-criticality network traffic on advanced production lines compel the adoption of scheduled mono-PET flows.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Embedded mono-PET cavities integrated directly into field devices are critical for streamlining localized pharmaceutical assembly.
What is Monomaterial PET Pharmaceutical Blister Packs and what is it mainly used for?
Monomaterial PET provides time-bound moisture protection and full recyclability. It is primarily used to synchronize stability requirements for tablets and capsules in high-volume dispensing environments.
What does Monomaterial PET Pharmaceutical Blister Packs mean in this report?
The scope encompasses specialized hardware and software built on single-polymer PET standards to guarantee low-latency stability protection.
What is included in the scope of this Monomaterial PET Pharmaceutical Blister Packs report?
Market covers pharmaceutical-grade mono-PET films, preformed trays, lidding foils, and integrated form-fill-seal consumables featuring native recyclability capabilities.
What is excluded from the scope of this report?
Multi-material laminates containing PVC or aluminum, non-pharmaceutical consumer formats, and liquid or injectable primary packaging are explicitly excluded.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on defined technology adoption assumptions for strategic planning purposes.
How does FMI build and validate the Monomaterial PET Pharmaceutical Blister Packs forecast?
The model applies a bottom-up methodology starting with global installed base metrics and cross-validates projections against publicly reported capital expenditure guidance.
What does zero reliance on speculative third-party market research mean here?
Primary interviews, verified regulatory filings, and official industry compliance registries are used exclusively instead of unverified syndicated estimates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.