Osteoarthritis Gene Therapy Market
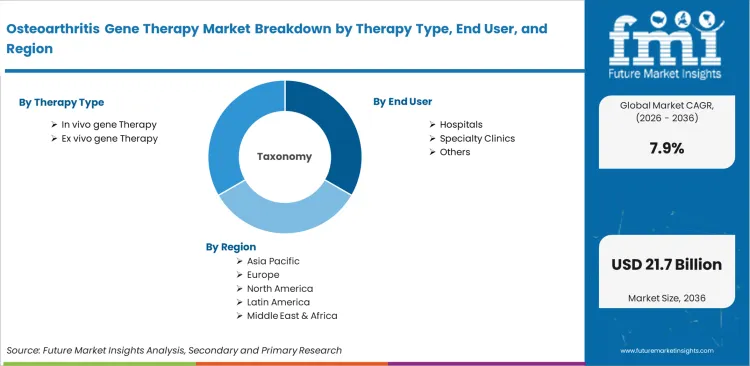
The osteoarthritis gene therapy market is segmented by Therapy Type (In vivo gene therapy, Ex vivo gene therapy), End User (Hospitals, Specialty clinics, Research institutes), Vector Type (Viral vectors, Non-viral vectors, Hybrid vectors), and Region. Forecast for 2026 to 2036.
Osteoarthritis Gene Therapy Market Size, Market Forecast and Outlook By FMI
The osteoarthritis gene therapy market crossed a valuation of USD 8.7 billion in 2025 and is projected to reach USD 9.4 billion in 2026. FMI estimates the market will expand at a CAGR of 7.9% from 2026 to 2036, taking total valuation to USD 20.1 billion by 2036.
Summary of Osteoarthritis Gene Therapy Market
- The market is forecast to reach USD 20.1 billion by 2036.
- The market is expected to grow at a CAGR of 7.9% from 2026 to 2036.
- The market was estimated at USD 8.7 billion in 2025.
- The forecast period represents an incremental opportunity of USD 10.7 billion.
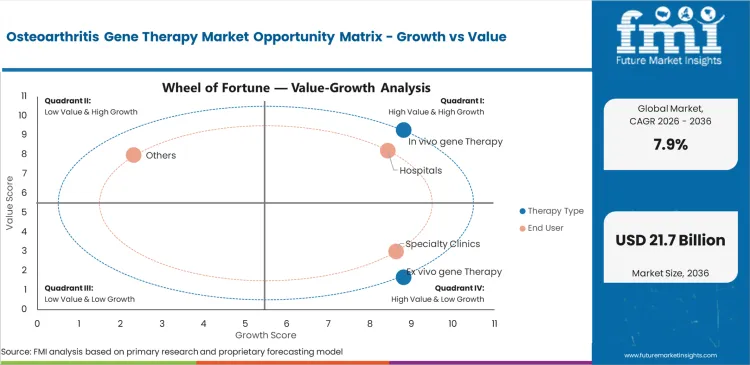
- In vivo gene therapy leads the therapy type segment with 52.8% share, driven by direct intra-articular delivery advantages.
- Viral vectors dominate the vector segment with 68.0% share, supported by strong clinical pipeline adoption.
- Hospitals account for 61.3% share in end use, due to specialized infrastructure and monitoring requirements.
- East Asia contributes 34.0% share in regional demand, led by strong growth in China and Japan.
- Key companies in the market include Pacira BioSciences, Kolon TissueGene, Genascence, Xalud Therapeutics, Belief BioMed, and GQ Bio Therapeutics.
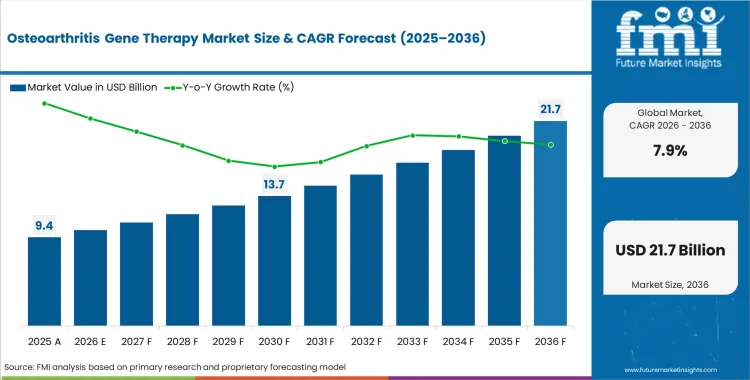
This outlook reflects a market moving from exploratory promise toward more measurable clinical and commercial direction, especially as trial programs place greater emphasis on cartilage regeneration, improvement, and longer-duration joint preservation.
Orthopedic specialists assessing osteoarthritis gene therapy are increasingly looking beyond short-duration symptom management and focusing on treatments that can change the course of joint degeneration with fewer interventions. That shift matters commercially because single-injection or limited-dose approaches fit the long-term economics of care only when they can show durable benefit against delayed surgery, lower repeat procedure burden, and improved outcomes. Hospitals and payers are still weighing the high upfront treatment cost against the possibility of reducing future arthroplasty volumes and chronic disease-management expense.
Commercial scale for cartilage repair will depend heavily on regulatory clarity around disease-modification endpoints in intra-articular osteoarthritis gene therapy. Standardized approval pathways would make formulary review, pricing discussion, and institutional adoption far more manageable across large hospital systems. Current cartilage-repair and gene-delivery pipelines suggest the market is moving closer to that point, but broader uptake will still depend on how quickly clinical evidence translates into reimbursement confidence and treatment-protocol acceptance.
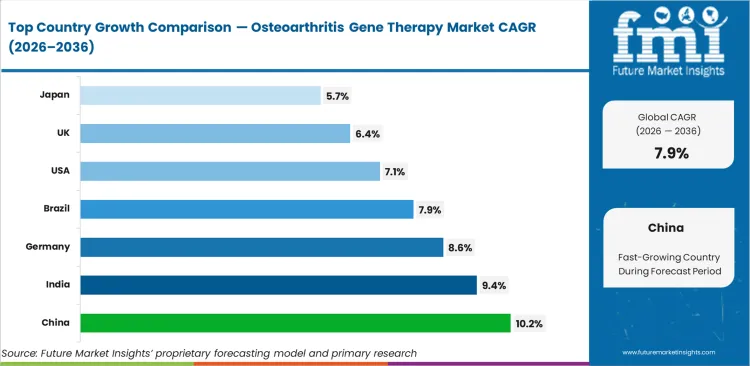
China is projected to expand at a CAGR of 10.2% through 2036, supported by strong state-backed investment in cell and gene therapy capabilities. India is expected to register 9.4% CAGR, helped by rising clinical trial capacity and a broader translational research base. Germany is likely to grow at 8.6% as advanced-therapy pricing and reimbursement frameworks become more defined. Brazil is set to record 7.9% CAGR during the forecast period, while the United States is anticipated to advance at 7.1%, reflecting tighter pricing scrutiny despite strong innovation depth. The United Kingdom is expected to grow at 6.4%, and Japan is projected to post 5.7%, showing a more measured adoption curve shaped by market-access timing and regulatory progression.
Segmental Analysis
Osteoarthritis Gene Therapy Market Analysis by Therapy Type
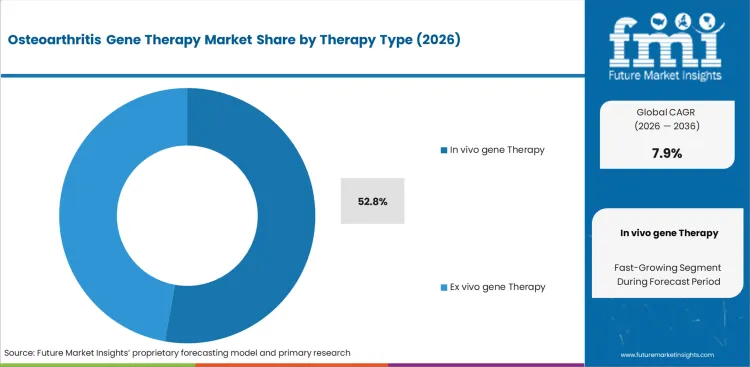
Direct injection formats fit more easily into orthopedic practice than treatment models that depend on cell extraction, lab handling, and reinfusion. Orthopedic department heads often prefer approaches that can be inserted into established procedural routines without building dedicated cell-processing capacity. Direct vector delivery into synovial fluid also aligns well with existing viscosupplementation workflows. In 2026, in vivo gene therapy is expected to contribute 52.8% of total market share. Pre-existing neutralizing antibodies remain an important clinical limitation, since prior viral exposure can reduce therapeutic response before treatment even begins. Providers comparing in vivo and ex vivo osteoarthritis gene therapy often underweight antibody screening, which can lead to avoidable efficacy gaps and payer disputes.
- Workflow integration: Direct intra-articular delivery follows familiar injection pathways. Orthopedic surgeons can adopt the modality without building an entirely new procedural model.
- Cold-chain dependency: Strict temperature control shapes distribution and storage discipline. Pharmacy teams face real spoilage risk when monitoring systems break or transfer conditions slip.
- Immunogenicity screening: Patient selection depends on accurate antibody testing. Clinical coordinators need tight pre-treatment protocols to protect vector performance.
Osteoarthritis Gene Therapy Market Analysis by End User
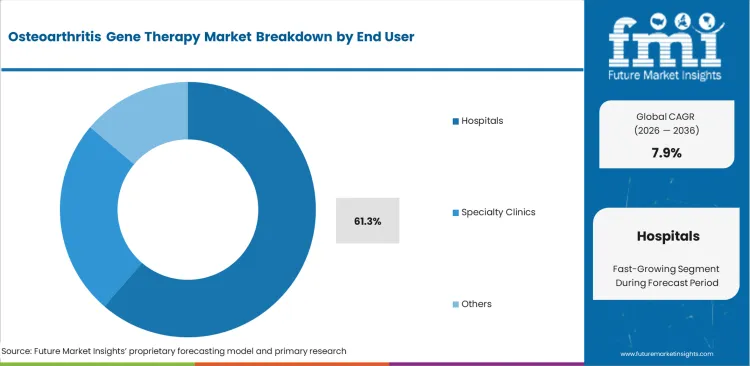
Major medical centers are likely to remain the first point of commercial adoption because advanced biologics require infrastructure that smaller care settings usually do not maintain. Hospitals are anticipated to capture 61.3% share in 2026. Specialized biosafety systems, controlled storage conditions, and trained pharmacy support continue to limit broader migration into outpatient settings. Tertiary centers are better placed to manage ultra-low temperature storage, biological handling controls, and multidisciplinary treatment planning. Reimbursement adds another operational barrier, since knee hyaluronic acid injections move through established pathways while emerging gene therapies often require new inpatient billing logic and payer review. Institutions that delay coding preparation can expose themselves to large unreimbursed product costs.
- Infrastructure qualification: Handling advanced biologics requires certified facilities and controlled workflows. Hospital administrators need these approvals in place before taking product shipments.
- Reimbursement navigation: High one-time treatment cost draws close payer scrutiny. Financial teams often face lengthy authorization cycles before each scheduled procedure.
- Multidisciplinary coordination: Treatment involves surgeons, pharmacists, and genetic counselors. Clinical pathway leaders must keep communication aligned across departments that usually work separately.
Osteoarthritis Gene Therapy Market Analysis by Vector Type
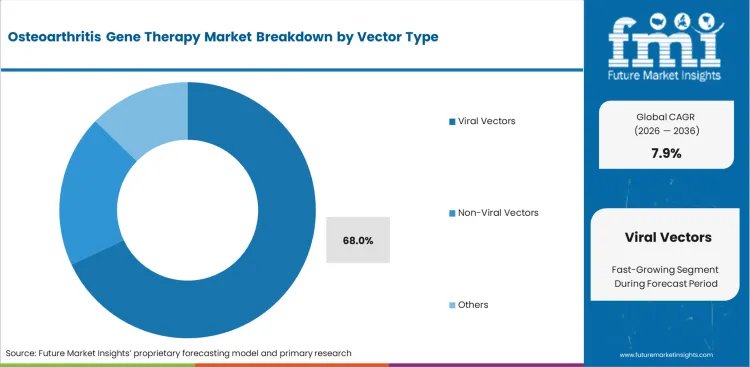
Reliable gene delivery in dense, relatively avascular joint tissue keeps viral systems ahead of alternative platforms. Chief scientific officers continue to rely on adeno-associated viral systems because they support sustained transgene expression in chondrocytes and give development teams a more established clinical path. Evidence from broader gene therapy in CNS disorder programs also strengthens confidence in the safety and durability profile of these vector platforms. Viral vectors are set to represent 68.0% share in 2026. Manufacturing remains the main commercial constraint, as global capacity for clinical-grade viral vector production is still limited. Companies that fail to lock in production slots early often face trial delays that have little to do with the underlying science.
- Expression durability: Adeno-associated viruses can support therapeutic protein expression over long periods. Investigators rely on that persistence when evaluating joint preservation.
- Production bottlenecks: Specialized bioreactor capacity remains limited across the supply base. Supply chain teams must compete early for scarce manufacturing availability.
- Neutralizing immunity: Pre-existing antibodies can inactivate viral delivery systems before they reach the target site. R&D teams need capsid design strategies that reduce this immune interference.
Osteoarthritis Gene Therapy Market Drivers, Restraints, and Opportunities
Escalating revision surgery burdens force hospital administrators to demand interventions that modify disease progression rather than simply masking pain. Orthopaedic department heads face mounting pressure from value-based care contracts to prevent joint deterioration entirely. Delaying adoption of biological solutions ensures continued reliance on mechanical joint replacement, a pathway facing severe long-term cost constraints. Widespread implementation of joint replacement alternative pathways accelerates as clinical data proves true cartilage regeneration.
Strict FDA and EMA regulatory requirements for demonstrating disease modification slow clinical advancement globally. Regulators demand extensive long-term follow-up to prove cartilage thickening, forcing multi-year trial timelines. This friction remains persistent because standard pain reduction metrics no longer satisfy advanced therapy approval criteria. Emerging biomarker analysis offers partial solutions, yet validating these surrogate endpoints requires significant preliminary investment limiting fast-track approvals for hips and knees reconstructive alternatives.
Opportunities in the Osteoarthritis Gene Therapy Market
- Non-viral delivery platforms: Advancing non-viral osteoarthritis gene therapy technology bypasses existing viral manufacturing bottlenecks. R&D directors gain faster clinical trial initiation capabilities.
- Biomarker validation: Discovering reliable early-response indicators shortens clinical evaluation periods. Chief scientific officers can prove efficacy before definitive radiological changes appear.
- Treatment substitution: Comparing osteoarthritis gene therapy vs stem cell therapy reveals distinct advantages in localized tissue regeneration without complex cell harvesting. Product managers create distinct competitive advantages through integrated procedural kits augmenting standard soft tissue repair procedures.
Regional Analysis
Based on regional analysis, osteoarthritis gene therapy market is segmented into North America, Europe, East Asia, South Asia, Latin America, Oceania, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 10.2% |
| India | 9.4% |
| Germany | 8.6% |
| Brazil | 7.9% |
| USA | 7.1% |
| UK | 6.4% |
| Japan | 5.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
East Asia Osteoarthritis Gene Therapy Market Analysis
State-backed cellular biology investments accelerate clinical translation across major research centers. Rapidly aging populations strain existing orthopedic infrastructure, forcing healthcare ministries to fast-track novel biological interventions. FMI analysts note that regional regulatory bodies approve advanced therapy trials significantly earlier than their Western counterparts. This aggressive regulatory stance attracts deep biotechnology investment targeting massive, previously underserved patient pools.
- China: Acquisition directors across major provincial hospitals actively secure localized biomanufacturing supply chains to eliminate reliance on imported viral vectors. Driven by this aggressive localization strategy, the osteoarthritis gene therapy market in China is poised to expand at a CAGR of 10.2% through 2036. Regional companies achieving early commercialization gain large initial volume advantages superseding traditional bone grafts and substitutes.
- Japan: Japan is anticipated to see osteoarthritis gene therapy demand grow at a CAGR of 5.7% over the forecast period, anchored by deeply established regenerative medicine frameworks. Clinical investigators utilize these existing accelerated approval pathways to initiate human trials rapidly. Strict post-market surveillance requirements define the ongoing operational realities for manufacturers seeking to maintain long-term institutional formulary access.
North America Osteoarthritis Gene Therapy Market Analysis
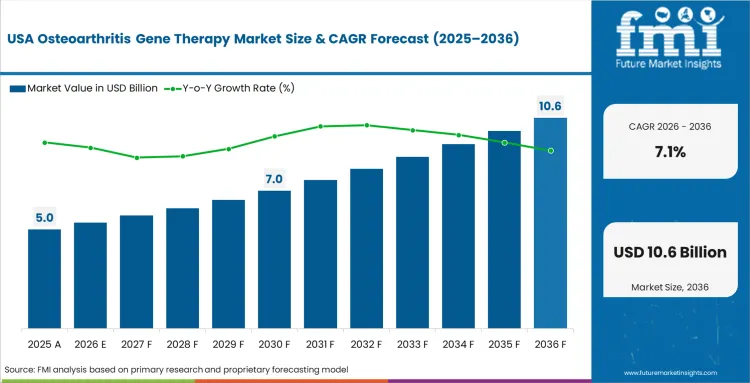
Considerable pricing scrutiny from public and private health insurers shapes commercialization strategies. High baseline procedural costs for traditional arthroplasty establish a specific economic threshold that new interventions must overcome. In FMI's view, biotech companies must design clinical trials explicitly proving definitive long-term cost offsets. Failure to confirm significant mechanical joint preservation guarantees highly restrictive hospital formulary placement.
- USA: Stringent FDA requirements demanding proof of structural modification completely dictate clinical trial designs for novel joint preservation therapeutics. Despite these regulatory hurdles, adoption of osteoarthritis gene therapy in the USA is likely to advance at a CAGR of 7.1% by 2036. Securing specific reimbursement codes remains an urgent priority for broad hospital adoption of these artificial cartilage implant alternatives.
Europe Osteoarthritis Gene Therapy Market Analysis
Fragmented national healthcare systems force pharmaceutical developers into complex, country-by-country pricing negotiations. Regional value assessment frameworks strictly prioritize long-term clinical utility over short-term symptom relief. FMI observes that successful companies build rigorous health economic models early during clinical development. These extensive dossiers must definitively prove absolute superiority over existing palliative treatments to secure sustainable institutional funding.
- Germany: Advanced therapy pricing frameworks explicitly encourage early market access for complex biological treatments. Capitalizing on this predictable regulatory environment, Germany is forecast to record steady growth in osteoarthritis gene therapy sales at a CAGR of 8.6% through 2036. These early movers establish the crucial real-world clinical evidence necessary for validating expensive cell free cartilage repair innovations.
- UK: Health economic leads face strict NICE evaluation criteria demanding pristine cost-effectiveness data before approving institutional funding for advanced therapeutics. These exceptionally rigorous evidence thresholds shape competitive positioning against deeply entrenched traditional orthopedic interventions. Despite these constraints, the osteoarthritis gene therapy industry in the UK is expected to grow at a CAGR of 6.4% during the study period, driven by proven clinical superiority.
FMI's report includes India and Brazil. Regulatory harmonization efforts across these specific emerging markets actively aim to attract massive international clinical trial operations seeking viable arthralgia management solutions.
Competitive Aligners for Market Players
Manufacturing constraints dictate competitive positioning far more than theoretical biological efficacy, a dynamic detailed within any osteoarthritis gene therapy competitive landscape report. Organizations tracking which companies are developing osteoarthritis gene therapy observe that entities like Pacira BioSciences and Kolon TissueGene focus heavily on securing reliable vector production capacity to ensure uninterrupted clinical trial supply. Small-scale academic spin-offs frequently possess superior genetic constructs but fail commercially due to insurmountable production bottlenecks. Establishing detailed chemistry, manufacturing, and controls documentation early separates viable commercial entities from pure research projects.
Incumbent players possess crucial regulatory experience navigating complex FDA advanced therapy pathways, solidifying their status among top osteoarthritis gene therapy companies. These established teams maintain vast libraries of validated assay protocols necessary for lot-release testing. Challengers must build identical analytical capabilities from scratch, adding years to development timelines. FMI's analysis reveals that partnering with organizations experienced in retinal gene therapy accelerates orthopedic vector validation significantly compared to teams handling rheumatoid arthritis therapeutics alone.
Hospital sourcing committees resist purchasing ultra-expensive single-dose therapies lacking definitive outcome guarantees. Large buyer networks demand risk-sharing financial agreements tied directly to patient clinical responses. Tension exists between biotechnology firms requiring upfront payment and hospitals demanding long-term efficacy proof. Value-based contracting mechanisms define commercial success criteria as new products exit clinical phases targeting reactive arthritis treatment and related joint degradation markets.
Key Players in Osteoarthritis Gene Therapy Market
- Pacira BioSciences
- Kolon Tissue Gene
- Genascence
- Xalud Therapeutics
- Belief BioMed
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 9.4 billion to USD 20.1 billion, at a CAGR of 7.9% |
| Market Definition | Direct genetic modification of joint tissues via specialized delivery vectors to halt degradation and induce localized therapeutic protein expression. |
| Segmentation | By Therapy Type, End User, Vector Type, and Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | USA, Canada, Brazil, Mexico, Germany, UK, France, Spain, Italy, China, Japan, South Korea, India, Australia, New Zealand, GCC Countries, South Africa |
| Key Companies Profiled | Pacira BioSciences, Kolon TissueGene, Genascence, Xalud Therapeutics, Belief BioMed |
| Forecast Period | 2026 to 2036 |
| Approach | Clinical stage progression probabilities applied to total addressable patient populations failing conservative management protocols. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Osteoarthritis Gene Therapy Market Analysis by Segments
Therapy Type:
- In vivo gene therapy
- Ex vivo gene therapy
End User:
- Hospitals
- Specialty clinics
- Research institutes
Vector Type:
- Viral vectors
- Non-viral vectors
- Hybrid vectors
Region:
- North America
- United States
- Canada
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia Pacific
- China
- Japan
- South Korea
- Taiwan
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- GCC Countries
- South Africa
Bibliography
- Grol, M. W. (2024). The evolving landscape of gene therapy strategies for the treatment of osteoarthritis. Osteoarthritis and Cartilage, 32(4), 372-384.
- Evans, C. H., Ghivizzani, S. C., & Robbins, P. D. (2024). The 2024 OREF Clinical Research Award: Progress toward a gene therapy for arthritis. The Journal of the American Academy of Orthopaedic Surgeons, 32(23), 1052-1060.
- DeJulius, C. R., Walton, B. L., Colazo, J. M., d’Arcy, R., Francini, N., Brunger, J. M., & Duvall, C. L. (2024). Engineering approaches for RNA-based and cell-based osteoarthritis therapies. Nature Reviews Rheumatology, 20(2), 81-100.
- Chen, Y., Luo, X., Kang, R., Cui, K., Ou, J., Zhang, X., & Liang, P. (2024). Current therapies for osteoarthritis and prospects of CRISPR-based genome, epigenome, and RNA editing in osteoarthritis treatment. Journal of Genetics and Genomics, 51(2), 159-183.
- Shakeri, M., Aminian, A., Mokhtari, K., Bahaeddini, M., Tabrizian, P., Farahani, N., Nabavi, N., & Hashemi, M. (2024). Unraveling the molecular landscape of osteoarthritis: A comprehensive review focused on the role of non-coding RNAs. Pathology - Research and Practice, 260, 155446.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Specific workflow integration challenges faced by orthopedic departments adopting in vivo biological interventions.
- Manufacturing capacity bottlenecks restricting global viral vector supply chains.
- Details driving any osteoarthritis gene therapy market report tracking purchasing constraints.
- Clinical trial design shifts required to prove cartilage regeneration to regulators.
- Neutralizing antibody prevalence rates impacting patient selection criteria across different regions.
- Cold-chain distribution risks inherent in supplying localized specialty clinics.
- Variables distinguishing related interventions such as native septic arthritis treatment algorithms.
- Diagnostics requirements bridging therapies with advanced juvenile idiopathic arthritis diagnostics.
Frequently Asked Questions
What is osteoarthritis gene therapy?
Direct genetic modification of joint tissues utilizes specialized delivery vectors to halt degradation. Treatments introduce specific nucleic acids to alter local cellular behavior and arrest cartilage deterioration.
How large is the osteoarthritis gene therapy market in 2026?
Valuation reaches USD 9.4 billion in 2026. Early commercialization of localized intra-articular treatments establishes this baseline, proving initial clinical viability and defining subsequent payer engagement frameworks.
What is the projected market value by 2036?
Revenue is expected to cross USD 20.1 billion by 2036. This figure signals a transition from palliative pain management toward definite disease modification protocols across major orthopedic centers.
Why is in vivo gene therapy the leading segment?
Direct intra-articular administration avoids complex cellular extraction and reimplantation procedures. Orthopedic surgeons favor this modality due to straightforward integration into existing joint injection workflows.
Which end users drive the most revenue in this market?
Advanced biological therapeutics require specific handling infrastructure and rigorous cold-chain maintenance. Hospitals possess necessary biosafety cabinets and specialized pharmacy personnel to manage these complex localized treatments safely.
Which countries are growing fastest in osteoarthritis gene therapy?
Massive state-backed cellular biology investments accelerate domestic clinical translation in China, yielding a 10.2% CAGR. India tracks closely at 9.4% due to expanding clinical trial capacity.
Which companies are closest to commercialization?
Entities like Pacira BioSciences and Kolon TissueGene lead clinical advancement by securing reliable vector production capacity. Their detailed chemistry, manufacturing, and controls documentation separates them from early-stage academic spin-offs.
How does osteoarthritis gene therapy differ from stem cell therapy?
Genetic interventions utilize engineered vectors to modify host cellular machinery directly. Stem cell approaches require complex harvesting, expansion, and reimplantation of living cells, introducing massive logistical and regulatory hurdles.
What are the main commercialization risks for OA gene therapy?
Strict regulatory demands for proving actual cartilage thickening extend clinical timelines. Demonstrating modification requires multi-year observation periods, adding massive costs to every development program.
Why are viral vectors leading the current pipeline?
Adeno-associated viruses deliver proven transfection efficiency across dense avascular joint tissues. Chief scientific officers rely on this established durability to ensure long-term therapeutic protein expression within synovial boundaries.
Can you summarize the osteoarthritis gene therapy market outlook to 2036?
Sustained investment carries cumulative valuation to USD 20.1 billion through 2036. The shift involves transitioning from basic symptom management to measurable cartilage regeneration metrics evaluated by hospital formulary committees.
Can you explain the growth drivers for osteoarthritis gene therapy demand?
Rising arthroplasty revision rates force orthopedic directors to seek earlier interventions. Payers require long-term cost containment strategies to offset escalating joint replacement expenditures, favoring curative intent therapies.
What defines the osteoarthritis gene therapy industry outlook?
Value-based contracting requires developers to guarantee efficacy, altering traditional upfront pharmaceutical payment models. This dynamic forces biotechnology firms to prove definitive long-term cost offsets.
How do payer contracts shape commercial strategy?
Large buyer networks demand risk-sharing agreements tied to definitive patient outcomes. Value-based contracting requires developers to guarantee efficacy, altering traditional upfront pharmaceutical payment models.
Why do academic spin-offs struggle commercially?
Small research groups often discover superior genetic constructs but lack required production scale. Securing early chemistry, manufacturing, and controls documentation remains a primary barrier to translating academic success into viable products.
How does neutralizing immunity impact vector selection?
Pre-existing antibodies destroy viral delivery vehicles before transfection occurs. Clinical coordinators must implement rigorous pre-screening protocols, driving developers to engineer novel capsids that evade natural immune surveillance.
What slows broader commercialization globally?
Strict regulatory demands for proving actual cartilage thickening extend clinical timelines. Demonstrating modification requires multi-year observation periods, adding massive costs to every development program.
How are non-viral vectors altering the sector?
Lipid nanoparticle advancements bypass severe viral manufacturing bottlenecks. R&D directors utilize these emerging platforms to initiate clinical trials faster and reduce overall production expenses.
What operational risk do pharmacy directors face?
Strict temperature controls govern advanced biologic distribution. Failing to maintain perfect cold-chain integrity during storage and localized transport results in massive inventory spoilage and immediate financial loss.
How does reimbursement coding complicate adoption?
Novel gene therapies lack established outpatient billing structures. Hospital administrators face prolonged prior authorization battles and must negotiate entirely new payment pathways for these high-cost single-dose interventions.
What is the primary constraint for viral vector production?
Global specialized bioreactor capacity remains severely limited. Supply chain managers face competition securing contract manufacturing slots, dictating commercial rollout speeds regardless of clinical success.
Why are biomarkers critical for future trials?
Identifying reliable early-response indicators shortens clinical evaluation significantly. Chief scientific officers use these surrogate endpoints to prove therapeutic efficacy years before definitive radiological changes manifest.
What defines success for incumbent manufacturers?
Established teams maintain extensive regulatory experience and validated assay protocols. This infrastructure allows rapid lot-release testing, providing a massive advantage over challenger biotechnology firms.
Table of Content
- Executive Summary
- Global Market Outlook
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy Type , 2026 to 2036
- In vivo gene Therapy
- Ex vivo gene Therapy
- In vivo gene Therapy
- Y to o to Y Growth Trend Analysis By Therapy Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Specialty Clinics
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Vector Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Vector Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Vector Type, 2026 to 2036
- Viral Vectors
- Non-Viral Vectors
- Others
- Viral Vectors
- Y to o to Y Growth Trend Analysis By Vector Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Vector Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Therapy Type
- By End User
- By Vector Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- By End User
- By Vector Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Therapy Type
- By End User
- By Vector Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- By End User
- By Vector Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Therapy Type
- By End User
- By Vector Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- By End User
- By Vector Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Therapy Type
- By End User
- By Vector Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- By End User
- By Vector Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Therapy Type
- By End User
- By Vector Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- By End User
- By Vector Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Therapy Type
- By End User
- By Vector Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- By End User
- By Vector Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Therapy Type
- By End User
- By Vector Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- By End User
- By Vector Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Therapy Type
- By End User
- By Vector Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Therapy Type
- By End User
- By Vector Type
- Competition Analysis
- Competition Deep Dive
- Pacira BioSciences
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Kolon TissueGene
- Genascence
- Xalud Therapeutics
- Belief BioMed
- Pacira BioSciences
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Therapy Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Vector Type, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Therapy Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Vector Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Therapy Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Vector Type, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Therapy Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Vector Type, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Therapy Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Vector Type, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Therapy Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Vector Type, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Vector Type, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Therapy Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Vector Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Therapy Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Therapy Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Therapy Type
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value Share and BPS Analysis by Vector Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Vector Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Vector Type
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Therapy Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Therapy Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Therapy Type
- Figure 26: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End User
- Figure 29: North America Market Value Share and BPS Analysis by Vector Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Vector Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Vector Type
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Therapy Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Therapy Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Therapy Type
- Figure 36: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by End User
- Figure 39: Latin America Market Value Share and BPS Analysis by Vector Type, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Vector Type, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Vector Type
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Therapy Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Therapy Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Therapy Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by End User
- Figure 49: Western Europe Market Value Share and BPS Analysis by Vector Type, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Vector Type, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Vector Type
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Therapy Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Therapy Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Therapy Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by End User
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Vector Type, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Vector Type, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Vector Type
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Therapy Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Therapy Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Therapy Type
- Figure 66: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by End User
- Figure 69: East Asia Market Value Share and BPS Analysis by Vector Type, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Vector Type, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Vector Type
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Therapy Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapy Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Therapy Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Vector Type, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Vector Type, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Vector Type
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Therapy Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Therapy Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Therapy Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Vector Type, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Vector Type, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Vector Type
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

