Solid Deodorant Sticks Market
Solid Deodorant Sticks Market Forecast and Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Solid Deodorant Sticks Market Forecast and Outlook 2026 to 2036
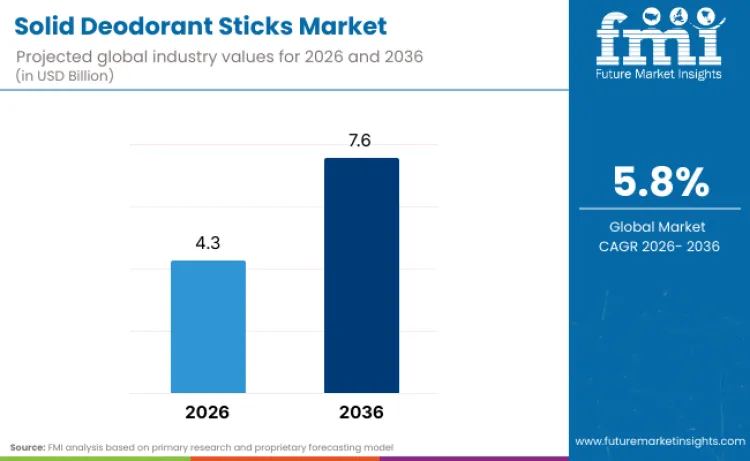
Solid deodorant sticks are projected to reach USD 4.3 billion in 2026 and continue expanding through 2036 as everyday hygiene routines shift toward formats that deliver consistent, portable, mess free application with reliable all day odor control. Growth is being reinforced by higher expectations around sweat and odor performance in urban, work centered lifestyles, which is pushing brands to improve efficacy while keeping skin feel and tolerability strong. Manufacturers are prioritizing antiperspirant stick development because this format allows controlled deposition of actives and a familiar consumer experience, while supporting differentiated positioning through fragrance systems, low residue performance, and sensitive skin variants.
Formulation investment is concentrating on balancing sweat reduction performance with comfort, using optimized antiperspirant salts, emollient structures, and glide systems that reduce drag and visible whitening while maintaining wear time. Clinical and dermatological validation is becoming a stronger commercial requirement, particularly for products claiming high efficacy or sensitive skin suitability, which is raising the importance of standardized testing and quality documentation.
Quick Stats for Solid Deodorant Sticks Market
- Solid Deodorant Sticks Industry Value (2026): USD 4.3 Billion
- Solid Deodorant Sticks Industry Forecast Value (2036): USD 7.6 Billion
- Solid Deodorant Sticks Industry Forecast CAGR: 5.8%
- Solid Deodorant Sticks Industry Leading Product Type: Antiperspirant Deodorant Sticks (38.4%)
- Solid Deodorant Sticks Industry Key Growth Regions: United States, India, Brazil
- Solid Deodorant Sticks Industry Key Players: Unilever, Procter & Gamble, Henkel, Beiersdorf, Colgate-Palmolive
Government regulation shapes both ingredients and claims. In the United States, antiperspirants are regulated as OTC drugs, which means active ingredients must align with the FDA monograph framework and products must follow drug facts labeling and manufacturing expectations for OTC products. In the European Union and UK, deodorants and antiperspirants are typically regulated as cosmetics under the EU Cosmetics Regulation (EC) 1223/2009, requiring safety assessment, responsible person obligations, and compliant labeling and claim substantiation. These compliance requirements favor brands that can support performance claims with robust testing, maintain traceable ingredient specifications, and execute consistent stick manufacturing at scale, which helps sustain category growth in both mature and emerging personal care markets.
Solid Deodorant Sticks Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 4.3 Billion |
| Industry Value (2036) | USD 7.6 Billion |
| CAGR (2026-2036) | 5.8% |
Source: Future Market Insights analysis, supported by a proprietary forecasting model and primary research
What Factors Are Driving Faster Adoption of Clinical-Strength Antiperspirant Sticks?
Clinical strength antiperspirant sticks are gaining faster adoption as consumers increasingly prioritize measurable sweat reduction, not only odor masking, which keeps aluminum salt systems central to product development and premium tier growth. Brands are improving how aluminum actives perform in solid sticks by optimizing salt type and particle characteristics, then adding stabilization steps that protect potency through shelf life and daily use. This is important for the category because clinical strength positioning relies on consistent moisture control across wear time, which supports higher value SKUs across supermarkets, hypermarkets, and drugstore channels. Formulators are also expanding usability for sensitive skin by adjusting pH, incorporating barrier supportive emollients, and using controlled delivery approaches that reduce sting, tackiness, and visible residue, helping convert consumers who previously avoided antiperspirants due to comfort concerns. Dermatology aligned education and pharmacist led guidance is reinforcing trust in aluminum based moisture control, while brands broaden assortments with stronger variants designed for heavy perspirers and hyperhidrosis adjacent needs. These shifts are making advanced antiperspirant technology a core competitive lever as companies build all day protection routines that combine sweat reduction, skin comfort, and cleaner application performance.
How Is the Solid Deodorant Sticks Market Segmented Across Key Categories?
Global landscape is intricately segmented to address diverse requirements of the personal care industry, categorizing sector by product type, sales channel, application, and consumer demographics. Structural division allows stakeholders to identify specific protection targets, such as rising demand for clinical strength formulations in medical applications or preference for natural alternatives in sensitive skin care. By analyzing these segments, manufacturers can tailor production lines to meet distinct needs of dermatological practitioners versus consumer retail brands. Segmentation also highlights shift from basic deodorant approaches to specialized antiperspirant interventions. As industry matures, granular segmentation becomes essential for understanding protection pathways and regulatory compliance, ensuring product development aligns with evolving personal care guidelines.
Why Do Antiperspirant Deodorant Sticks Maintain Market Leadership?
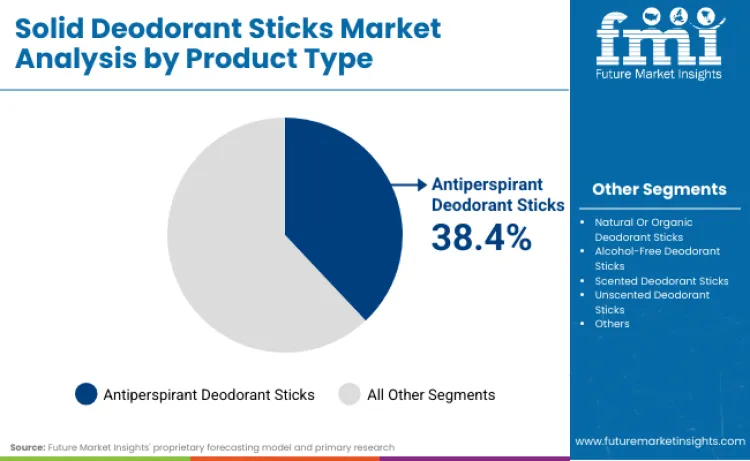
Antiperspirant deodorant sticks account for 38.4% of product type share in 2026, driven by extensive clinical evidence supporting sweat reduction benefits. Leadership position is underpinned by regulatory approvals from health authorities recognizing aluminum-based protection claims. Segment benefits from continuous formulation improvements, such as time-release technology extending protection duration. As per FMI's estimates, research focused on antiperspirant stability is renewing interest among pharmaceutical manufacturers. Critical role in managing excessive sweating further solidifies standing, as these products reduce moisture levels significantly. As dermatology guidelines prioritize antiperspirant applications, demand for clinical-grade concentrates is expected to remain robust.
Why Do Supermarkets or Hypermarkets Lead Sales Channels?
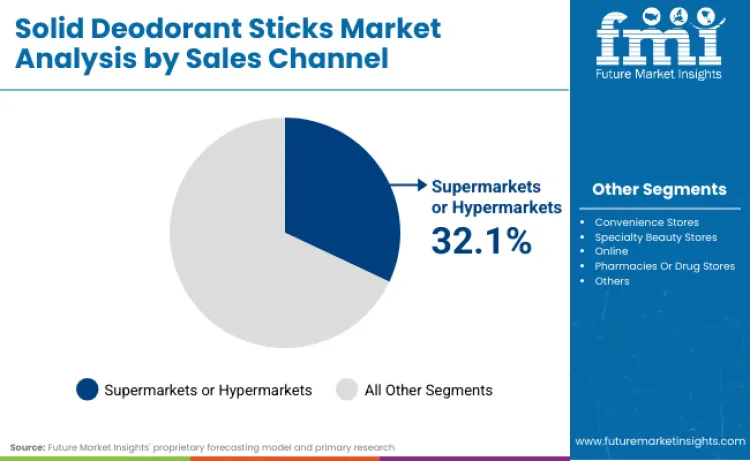
Supermarkets or hypermarkets capture 32.1% of sales channel share in 2026, reflecting consumer preference for convenient personal care shopping experiences. Dominance is linked to widespread product availability and competitive pricing strategies compared to specialty retailers. Manufacturers rely on mass retail distribution to ensure consistent product accessibility and brand visibility. These channels are essential for delivering mainstream deodorant products, driving priority status among consumer goods brands. Relentless pursuit of shelf space optimization ensures products designed for daily use remain primary choice. High consumer traffic necessitates dynamic inventory management, bolstering sustained revenue streams for retail partners.
Why Does Daily Use Lead Application Categories?
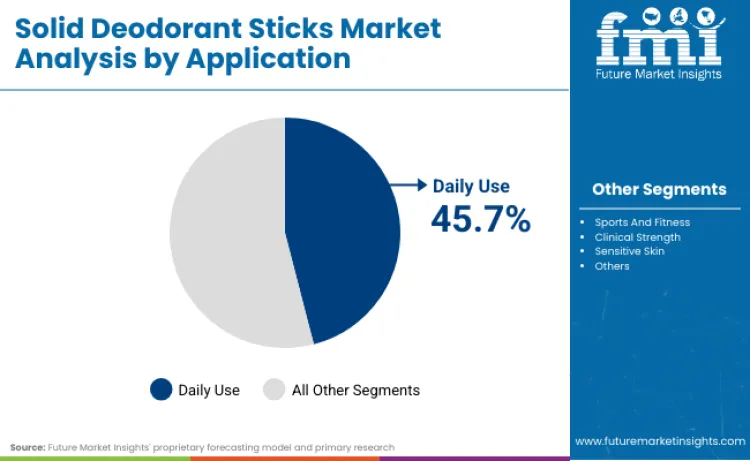
Daily use applications capture 45.7% of market share in 2026, owing to widespread consumer adoption requiring routine protection coverage. Dominance stems from lifestyle patterns demanding consistent antiperspirant application for workplace and social activities. As per FMI's projection, consumers prefer these formulations for their reliable protection and skin compatibility profiles. Usage in managing routine hygiene drives continuous volume growth among working populations. Compatibility with various clothing materials allows for versatile application across different daily scenarios. Manufacturers focusing on gentle formulations and extended protection for these routine conditions secure significant loyalty among everyday users.
How Will Gender-Specific Marketing Shape Deodorant Sales?
Gender-led positioning is still a major volume lever in deodorants because fragrance identity, benefit language, and pack cues remain strongly segmented across leading brand portfolios, while “inclusive” scent strategies are widening addressable audiences for newer lines. Recent category moves show how incumbents are actively building parallel propositions across male and female segments, including whole-body deodorant extensions rolled out under multiple brands that sit on different positioning pillars. At the same time, tighter rules around fragrance allergen disclosure in the EU are increasing formulation and label-management complexity, which directly affects how brands design, market, and refresh gender-coded scent profiles.
How Will E-commerce Integration Transform Distribution?
E-commerce is changing deodorant distribution by shifting repeat purchase from “store reminder” to “systematic replenishment,” making subscriptions and auto-ship models a practical route to stabilize demand and reduce out-of-stock churn. Direct-to-consumer deodorant brands already operationalize this through subscription programs that bundle deodorant and adjacent personal care items, reinforcing routine-based buying and supporting higher retention through predictable refill cycles. As digital sales grow, compliance and traceability expectations also rise, especially in markets where cosmetics oversight is tightening through requirements tied to safety substantiation and adverse event reporting, increasing the need for stronger product data control across digital shelves.
How Will Natural Ingredient Trends Influence Market Growth?
Natural and “clean” demand is reshaping growth by pulling consumers toward aluminum-free deodorants while simultaneously tightening the compliance bar for what brands can claim and how they manage safety documentation. In the USA, this shift is structurally important because antiperspirants are regulated as OTC drug products with defined active-ingredient frameworks, which makes “aluminum-free” positioning inherently more aligned with deodorants than antiperspirants and changes portfolio strategy by format and claim set. On the certification side, standards such as COSMOS formalize what qualifies for natural and organic certification, influencing ingredient sourcing, processing choices, and label credibility for brands competing on “natural” proof points.
How Will the Solid Deodorant Sticks Industry Evolve in Major Nations?
Global landscape for deodorant stick products is characterized by diverse cultural frameworks, influenced by climate conditions and personal care practice standards. Established markets prioritize clinical-tested formulations and dermatologist-recommended manufacturing, whereas emerging economies focus on accessible pricing and basic protection coverage. Asia-Pacific is emerging as rapid growth hub due to increasing urbanization and expanding personal care awareness. Conversely, North American sectors pivot towards premium protection and specialized antiperspirant protocols. Government initiatives in developing nations supporting hygiene education ensure sustained demand for basic deodorant products across all demographic segments.
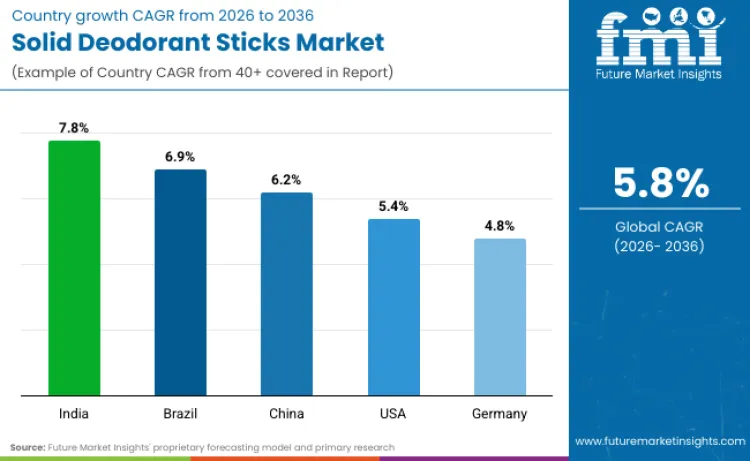
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 7.8% |
| Brazil | 6.9% |
| USA | 5.4% |
| China | 6.2% |
| Germany | 4.8% |
Source: Future Market Insights’ proprietary forecasting model and primary research
Why is the Solid Deodorant Sticks Industry in India Growing?
Sales of solid deodorant sticks in India are set to grow at 7.8% CAGR through 2036 because the category is scaling through a more formal cosmetics compliance regime that supports wider brand entry, localized manufacturing, and faster distribution expansion. India’s regulator, CDSCO, administers the Cosmetics Rules, 2020, which strengthens clarity around licensing, compliance responsibilities, and market access for cosmetics placed in India. This regulatory structure supports faster rollout of stick formats as brands expand product lines, add variants, and align labeling and quality requirements to India specific rules. Hygiene awareness programs also reinforce daily grooming routines and cleanliness messaging at a national level, supporting broader deodorant category penetration across urban consumers.
Why is the Solid Deodorant Sticks Sector in Brazil Expanding?
Demand for solid deodorant sticks in Brazil is projected to rise at 6.9% CAGR through 2036 because market access and portfolio expansion are supported by Anvisa’s structured classification and regularization system for personal hygiene products, including deodorants and antiperspirants. Anvisa’s official regulatory framework for personal hygiene products, cosmetics, and fragrances provides the compliance baseline for formulation, safety, and labeling expectations, helping organized brands scale distribution through national retail. RDC No. 7/2015 sets technical requirements for regularization and labeling and explains how products can follow registration or post market procedures depending on risk classification, which supports faster commercialization of compliant deodorant variants across channels.
Why is the Solid Deodorant Sticks Industry in USA Showing Maturity?
Solid deodorant sticks in the United States are poised to expand at 5.4% CAGR through 2036 because the market is shaped by a clear regulatory split between deodorants and antiperspirants, which drives continuous product innovation and claims based differentiation. FDA explains that deodorants that are also antiperspirants are cosmetic drug products, meaning they must meet requirements for both cosmetics and drugs. For antiperspirant products specifically, FDA has an OTC monograph and administrative order framework that sets conditions for these products to be considered generally recognized as safe and effective, which supports standardized performance positioning and consistent market supply. This structure sustains a mature market where growth comes from premiumization, skin sensitivity positioning, aluminum free alternatives, and dermatologist aligned messaging, rather than basic category formation.
Why is the Solid Deodorant Sticks Industry in China Growing?
Sales of deodorant sticks in China are set to grow at 6.2% CAGR through 2036 because the cosmetics market is operating under a stricter registration and filing system that supports broader brand participation while raising quality and safety baselines for daily use personal care. China’s NMPA has published the Provisions for Registration and Filing of Cosmetics, which operationalize the requirements under the national cosmetics supervision framework and standardize what companies must do before products can be sold. This pushes the deodorant category toward more compliant, better documented products and supports expansion of everyday grooming formats in modern retail and regulated ecommerce environments.
Why is the Solid Deodorant Sticks Sector in Germany Expanding?
Revenue from deodorant products in Germany is set to grow at 4.8% CAGR through 2036 because the market is driven by high trust in EU cosmetics compliance, plus strong consumer segmentation around ingredient safety and aluminum related preferences in antiperspirant style products. The EU Cosmetics Regulation establishes the legal requirements for cosmetic products placed on the EU market, including Germany, supporting consistent safety assessment, labeling discipline, and enforcement expectations. Germany’s BfR has published detailed reassessments and FAQs on aluminum in antiperspirants, which directly shapes consumer choice and product positioning in the deodorant aisle, including aluminum free lines and sensitive skin formats. EU level scientific review also addresses aluminum compounds in cosmetic products, reinforcing that ingredient evaluation is active and evidence led, which supports ongoing innovation and reformulation cycles.
What is the Competitive Outlook for the Solid Deodorant Sticks Market?
Competition in solid deodorant sticks is moving beyond basic odor control toward skin compatibility, format innovation, and sustainability-led repeat use. Large players are reshaping portfolios to capture premium and purpose-driven segments. Unilever’s acquisition of UK refillable brand Wild shows how seriously majors now view refillable stick systems as a scalable growth path rather than a niche eco-idea, using the brand to accelerate premium positioning and reduce plastic intensity in core markets. At the same time, Procter & Gamble has expanded deodorant positioning through its Secret, Old Spice, and Native lines by promoting broader “whole-body” usage, creating new occasions and increasing per-user consumption of stick formats.
Go-to-market strategies are also evolving. Brands are pairing mass retail visibility with direct-to-consumer refill programs, subscription packs, and sensitive-skin variants to encourage routine use and loyalty. Clinical and dermatology-friendly claims are becoming more visible on packs, reflecting rising consumer interest in aluminum alternatives and irritation-free protection.
Portfolio differences are becoming clearer: P&G emphasizes usage expansion and format versatility, while Unilever is building refillable ecosystems and premium sustainability narratives. As a result, the competitive edge is shifting toward companies that can combine dermatology credibility, refillable design, and strong distribution, turning deodorant sticks into a repeat, system-led purchase rather than a commodity hygiene item.
Key Players in Solid Deodorant Sticks Market
- Unilever
- Procter & Gamble
- Henkel
- Beiersdorf
- Colgate-Palmolive
- Revlon
- Native
- Tom's Of Maine
- Arm & Hammer
- Shiseido
- Coty Inc.
- Church & Dwight
- Kao Corporation
- L'Occitane
- Johnson & Johnson
- Edgewell Personal Care
- Godrej Consumer Products
- Wipro Consumer Care
- Marico Limited
- ITC Limited
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type Segments | Antiperspirant Deodorant Sticks; Natural Or Organic Deodorant Sticks; Alcohol-Free Deodorant Sticks; Scented Deodorant Sticks; Unscented Deodorant Sticks; Others |
| Sales Channel Categories | Supermarkets Or Hypermarkets; Convenience Stores; Specialty Beauty Stores; Online; Pharmacies Or Drug Stores; Others |
| Application Categories | Daily Use; Sports And Fitness; Clinical Strength; Sensitive Skin; Others |
| Company Categories | Multinational Corporations; Regional Brands; Natural Product Companies; Pharmaceutical Companies |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Key Countries | India, Brazil, USA, China, Germany |
| Key Companies Profiled | Unilever; Procter & Gamble; Henkel; Beiersdorf; Colgate-Palmolive; Revlon; Native; Tom's Of Maine; Arm & Hammer; Shiseido; Coty Inc.; Church & Dwight; Kao Corporation; L'Occitane; Johnson & Johnson; Edgewell Personal Care; Godrej Consumer Products; Wipro Consumer Care; Marico Limited; ITC Limited |
| Additional Attributes | Dollar sales measured for solid deodorant sticks used in daily hygiene and personal care applications, specified by active ingredient type (aluminum salts, natural minerals, baking soda, essential oils), product format, protection duration preference, application focus (antiperspirant, deodorant, clinical strength and sensitive skin), retail model (mass market vs. premium), and compliance alignment with evolving safety standards and dermatological testing regulations. |
Solid Deodorant Sticks Market by Segments
Product Type
- Antiperspirant Deodorant Sticks
- Natural Or Organic Deodorant Sticks
- Alcohol-Free Deodorant Sticks
- Scented Deodorant Sticks
- Unscented Deodorant Sticks
- Others
Sales Channel
- Supermarkets Or Hypermarkets
- Convenience Stores
- Specialty Beauty Stores
- Online
- Pharmacies Or Drug Stores
- Others
Application
- Daily Use
- Sports And Fitness
- Clinical Strength
- Sensitive Skin
- Others
Company
- Multinational Corporations
- Regional Brands
- Natural Product Companies
- Pharmaceutical Companies
Bibliography
- USA Food and Drug Administration. (2023). Antiperspirant drug products for over‑the‑counter human use; final monograph. USA Department of Health and Human Services, FDA.
- Personal Care Products Council. (2024). Strategic guidance on cosmetic ingredient safety and regulatory compliance. Personal Care Products Council.
- Organization for Economic Co‑operation and Development. (2024). Extended Producer Responsibility and consumer product sustainability policy. OECD Publishing.
Frequently Asked Questions
How big is the global solid deodorant sticks market?
The global solid deodorant sticks market is valued at USD 4.3 billion in 2026.
What is the growth outlook for the solid deodorant sticks market over the next 10 years?
The market is projected to grow at a 5.8% CAGR from 2026 to 2036, reaching USD 7.6 billion.
Which product segments or formats drive demand in this market?
Demand is driven primarily by antiperspirant deodorant sticks, supported by daily-use formats sold through mass retail channels.
How does consumer behavior differ by region?
Emerging markets such as India and Brazil prioritize affordable daily-use protection, while North America and Europe focus on clinical-strength, premium, and sensitive-skin deodorant variants.
What are the main risks and constraints affecting this market?
Key constraints include regulatory scrutiny of aluminum-based actives, rising demand for natural alternatives, claims substantiation requirements, and formulation complexity for sensitive skin.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Antiperspirant Deodorant Sticks
- Natural or Organic Deodorant Sticks
- Alcohol-Free Deodorant Sticks
- Scented Deodorant Sticks
- Unscented Deodorant Sticks
- Others
- Y-o-Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Sales Channel, 2026 to 2036
- Supermarkets or Hypermarkets
- Convenience Stores
- Specialty Beauty Stores
- Online
- Pharmacies or Drug Stores
- Others
- Y-o-Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Application, 2026 to 2036
- Daily Use
- Sports and Fitness
- Clinical Strength
- Sensitive Skin
- Others
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Company
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Company, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Company, 2026 to 2036
- Multinational Corporations
- Regional Brands
- Natural Product Companies
- Pharmaceutical Companies
- Y-o-Y Growth Trend Analysis By Company, 2021 to 2025
- Absolute $ Opportunity Analysis By Company, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Sales Channel
- By Application
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sales Channel
- By Application
- By Company
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Sales Channel
- By Application
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sales Channel
- By Application
- By Company
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Sales Channel
- By Application
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sales Channel
- By Application
- By Company
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Sales Channel
- By Application
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sales Channel
- By Application
- By Company
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Sales Channel
- By Application
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sales Channel
- By Application
- By Company
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Sales Channel
- By Application
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sales Channel
- By Application
- By Company
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Sales Channel
- By Application
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Sales Channel
- By Application
- By Company
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Sales Channel
- By Application
- By Company
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Sales Channel
- By Application
- By Company
- Competition Analysis
- Competition Deep Dive
- Unilever
- Procter & Gamble
- Henkel
- Beiersdorf
- Colgate-Palmolive
- Revlon
- Native
- Tom's Of Maine
- Arm & Hammer
- Shiseido
- Coty Inc.
- Church & Dwight
- Kao Corporation
- L'Occitane
- Johnson & Johnson
- Edgewell Personal Care
- Godrej Consumer Products
- Wipro Consumer Care
- Marico Limited
- ITC Limited
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Solid Deodorant Sticks Market Key Takeaways
- Global Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- Global Solid Deodorant Sticks Market Y-o-Y Growth (%), 2021-2036
- Global Solid Deodorant Sticks Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Global Solid Deodorant Sticks Market Pricing Analysis (USD/Unit), 2021-2036
- Global Solid Deodorant Sticks Market Size (USD Billion) By Product Type, 2021-2036
- Global Solid Deodorant Sticks Market Size (USD Billion) By Sales Channel, 2021-2036
- Global Solid Deodorant Sticks Market Size (USD Billion) By Application, 2021-2036
- Global Solid Deodorant Sticks Market Size (USD Billion) By Company, 2021-2036
- Global Solid Deodorant Sticks Market Size (USD Billion) By Region, 2021-2036
- North America Solid Deodorant Sticks Market Size (USD Billion) By Country, 2021-2036
- North America Solid Deodorant Sticks Market Size (USD Billion) By Product Type, 2021-2036
- North America Solid Deodorant Sticks Market Size (USD Billion) By Sales Channel, 2021-2036
- North America Solid Deodorant Sticks Market Size (USD Billion) By Application, 2021-2036
- North America Solid Deodorant Sticks Market Size (USD Billion) By Company, 2021-2036
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Country
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Application
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Company
- Latin America Solid Deodorant Sticks Market Size (USD Billion) By Country, 2021-2036
- Latin America Solid Deodorant Sticks Market Size (USD Billion) By Product Type, 2021-2036
- Latin America Solid Deodorant Sticks Market Size (USD Billion) By Sales Channel, 2021-2036
- Latin America Solid Deodorant Sticks Market Size (USD Billion) By Application, 2021-2036
- Latin America Solid Deodorant Sticks Market Size (USD Billion) By Company, 2021-2036
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Country
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Application
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Company
- Western Europe Solid Deodorant Sticks Market Size (USD Billion) By Country, 2021-2036
- Western Europe Solid Deodorant Sticks Market Size (USD Billion) By Product Type, 2021-2036
- Western Europe Solid Deodorant Sticks Market Size (USD Billion) By Sales Channel, 2021-2036
- Western Europe Solid Deodorant Sticks Market Size (USD Billion) By Application, 2021-2036
- Western Europe Solid Deodorant Sticks Market Size (USD Billion) By Company, 2021-2036
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Country
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Application
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Company
- Eastern Europe Solid Deodorant Sticks Market Size (USD Billion) By Country, 2021-2036
- Eastern Europe Solid Deodorant Sticks Market Size (USD Billion) By Product Type, 2021-2036
- Eastern Europe Solid Deodorant Sticks Market Size (USD Billion) By Sales Channel, 2021-2036
- Eastern Europe Solid Deodorant Sticks Market Size (USD Billion) By Application, 2021-2036
- Eastern Europe Solid Deodorant Sticks Market Size (USD Billion) By Company, 2021-2036
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Country
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Application
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Company
- East Asia Solid Deodorant Sticks Market Size (USD Billion) By Country, 2021-2036
- East Asia Solid Deodorant Sticks Market Size (USD Billion) By Product Type, 2021-2036
- East Asia Solid Deodorant Sticks Market Size (USD Billion) By Sales Channel, 2021-2036
- East Asia Solid Deodorant Sticks Market Size (USD Billion) By Application, 2021-2036
- East Asia Solid Deodorant Sticks Market Size (USD Billion) By Company, 2021-2036
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Country
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Application
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Company
- South Asia and Pacific Solid Deodorant Sticks Market Size (USD Billion) By Country, 2021-2036
- South Asia and Pacific Solid Deodorant Sticks Market Size (USD Billion) By Product Type, 2021-2036
- South Asia and Pacific Solid Deodorant Sticks Market Size (USD Billion) By Sales Channel, 2021-2036
- South Asia and Pacific Solid Deodorant Sticks Market Size (USD Billion) By Application, 2021-2036
- South Asia and Pacific Solid Deodorant Sticks Market Size (USD Billion) By Company, 2021-2036
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Country
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Application
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Company
- Middle East & Africa Solid Deodorant Sticks Market Size (USD Billion) By Country, 2021-2036
- Middle East & Africa Solid Deodorant Sticks Market Size (USD Billion) By Product Type, 2021-2036
- Middle East & Africa Solid Deodorant Sticks Market Size (USD Billion) By Sales Channel, 2021-2036
- Middle East & Africa Solid Deodorant Sticks Market Size (USD Billion) By Application, 2021-2036
- Middle East & Africa Solid Deodorant Sticks Market Size (USD Billion) By Company, 2021-2036
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Country
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Application
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Company
- USA Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- USA Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- USA Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- USA Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- USA Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Canada Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Canada Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Canada Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Canada Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Canada Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Mexico Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Mexico Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Mexico Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Mexico Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Mexico Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Brazil Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Brazil Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Brazil Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Brazil Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Brazil Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Chile Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Chile Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Chile Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Chile Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Chile Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Germany Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Germany Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Germany Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Germany Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Germany Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- UK Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- UK Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- UK Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- UK Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- UK Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Italy Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Italy Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Italy Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Italy Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Italy Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Spain Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Spain Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Spain Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Spain Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Spain Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- France Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- France Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- France Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- France Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- France Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- India Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- India Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- India Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- India Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- India Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- ASEAN Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- ASEAN Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- ASEAN Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- ASEAN Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- ASEAN Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Australia & New Zealand Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Australia & New Zealand Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Australia & New Zealand Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Australia & New Zealand Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- China Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- China Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- China Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- China Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- China Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Japan Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Japan Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Japan Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Japan Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Japan Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- South Korea Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- South Korea Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- South Korea Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- South Korea Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- South Korea Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Russia Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Russia Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Russia Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Russia Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Russia Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Poland Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Poland Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Poland Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Poland Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Poland Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Hungary Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Hungary Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Hungary Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Hungary Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Hungary Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Kingdom of Saudi Arabia Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Kingdom of Saudi Arabia Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Kingdom of Saudi Arabia Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Kingdom of Saudi Arabia Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Turkiye Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- Turkiye Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- Turkiye Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- Turkiye Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- Turkiye Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- South Africa Solid Deodorant Sticks Pricing Analysis (USD/Unit), 2021-2036
- South Africa Solid Deodorant Sticks Market Share Analysis (%) By Product Type, 2025
- South Africa Solid Deodorant Sticks Market Share Analysis (%) By Sales Channel, 2025
- South Africa Solid Deodorant Sticks Market Share Analysis (%) By Application, 2025
- South Africa Solid Deodorant Sticks Market Share Analysis (%) By Company, 2025
- Solid Deodorant Sticks Market Competition Dashboard
- Solid Deodorant Sticks Market Competition Benchmarking Analysis
- Solid Deodorant Sticks Market Share Analysis of Top Players By Regional, 2025
- Solid Deodorant Sticks Market Share Analysis of Top Players By Product Type, 2025
- Solid Deodorant Sticks Market Share Analysis of Top Players By Sales Channel, 2025
- Solid Deodorant Sticks Market Share Analysis of Top Players By Application, 2025
- Solid Deodorant Sticks Market Share Analysis of Top Players By Company, 2025
- Unilever - Company Profile
- Procter & Gamble - Company Profile
- Henkel - Company Profile
- Beiersdorf - Company Profile
- Colgate-Palmolive - Company Profile
- Revlon - Company Profile
- Native - Company Profile
- Tom's Of Maine - Company Profile
- Arm & Hammer - Company Profile
- Shiseido - Company Profile
- Coty Inc. - Company Profile
- Church & Dwight - Company Profile
- Kao Corporation - Company Profile
- L'Occitane - Company Profile
- Johnson & Johnson - Company Profile
- Edgewell Personal Care - Company Profile
- Godrej Consumer Products - Company Profile
- Wipro Consumer Care - Company Profile
- Marico Limited - Company Profile
- ITC Limited - Company Profile
- Assumptions & Acronyms Used in Solid Deodorant Sticks Market Report
List of Figures
- Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- Solid Deodorant Sticks Market Y-o-Y Growth (%), 2021-2036
- Solid Deodorant Sticks Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Solid Deodorant Sticks Market Value Chain Analysis
- Solid Deodorant Sticks Market Supply Chain Analysis
- Solid Deodorant Sticks Market Investment Feasibility Matrix
- Solid Deodorant Sticks Market PESTLE Analysis
- Solid Deodorant Sticks Market Porter's Five Forces Analysis
- Solid Deodorant Sticks Market Product Life Cycle Analysis
- Solid Deodorant Sticks Market Opportunity Map Analysis
- Solid Deodorant Sticks Market Scenario Forecast Analysis
- Solid Deodorant Sticks Market Production and Consumption Statistics
- Solid Deodorant Sticks Market Import and Export Statistics
- Global Solid Deodorant Sticks Market Share (%) By Product Type, 2025
- Global Solid Deodorant Sticks Market Share (%) By Product Type, 2036
- Global Solid Deodorant Sticks Market Share (%) By Sales Channel, 2025
- Global Solid Deodorant Sticks Market Share (%) By Sales Channel, 2036
- Global Solid Deodorant Sticks Market Share (%) By Application, 2025
- Global Solid Deodorant Sticks Market Share (%) By Application, 2036
- Global Solid Deodorant Sticks Market Share (%) By Company, 2025
- Global Solid Deodorant Sticks Market Share (%) By Company, 2036
- Global Solid Deodorant Sticks Market Share (%) By Region, 2025
- Global Solid Deodorant Sticks Market Share (%) By Region, 2036
- Global Solid Deodorant Sticks Market Attractiveness Analysis By Region
- North America Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- North America Solid Deodorant Sticks Market Share (%) By Country, 2025
- North America Solid Deodorant Sticks Market Share (%) By Country, 2036
- North America Solid Deodorant Sticks Market Share (%) By Product Type, 2025
- North America Solid Deodorant Sticks Market Share (%) By Product Type, 2036
- North America Solid Deodorant Sticks Market Share (%) By Sales Channel, 2025
- North America Solid Deodorant Sticks Market Share (%) By Sales Channel, 2036
- North America Solid Deodorant Sticks Market Share (%) By Application, 2025
- North America Solid Deodorant Sticks Market Share (%) By Application, 2036
- North America Solid Deodorant Sticks Market Share (%) By Company, 2025
- North America Solid Deodorant Sticks Market Share (%) By Company, 2036
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Country
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Application
- North America Solid Deodorant Sticks Market Attractiveness Analysis By Company
- Latin America Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- Latin America Solid Deodorant Sticks Market Share (%) By Country, 2025
- Latin America Solid Deodorant Sticks Market Share (%) By Country, 2036
- Latin America Solid Deodorant Sticks Market Share (%) By Product Type, 2025
- Latin America Solid Deodorant Sticks Market Share (%) By Product Type, 2036
- Latin America Solid Deodorant Sticks Market Share (%) By Sales Channel, 2025
- Latin America Solid Deodorant Sticks Market Share (%) By Sales Channel, 2036
- Latin America Solid Deodorant Sticks Market Share (%) By Application, 2025
- Latin America Solid Deodorant Sticks Market Share (%) By Application, 2036
- Latin America Solid Deodorant Sticks Market Share (%) By Company, 2025
- Latin America Solid Deodorant Sticks Market Share (%) By Company, 2036
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Country
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Application
- Latin America Solid Deodorant Sticks Market Attractiveness Analysis By Company
- Western Europe Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- Western Europe Solid Deodorant Sticks Market Share (%) By Country, 2025
- Western Europe Solid Deodorant Sticks Market Share (%) By Country, 2036
- Western Europe Solid Deodorant Sticks Market Share (%) By Product Type, 2025
- Western Europe Solid Deodorant Sticks Market Share (%) By Product Type, 2036
- Western Europe Solid Deodorant Sticks Market Share (%) By Sales Channel, 2025
- Western Europe Solid Deodorant Sticks Market Share (%) By Sales Channel, 2036
- Western Europe Solid Deodorant Sticks Market Share (%) By Application, 2025
- Western Europe Solid Deodorant Sticks Market Share (%) By Application, 2036
- Western Europe Solid Deodorant Sticks Market Share (%) By Company, 2025
- Western Europe Solid Deodorant Sticks Market Share (%) By Company, 2036
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Country
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Application
- Western Europe Solid Deodorant Sticks Market Attractiveness Analysis By Company
- Eastern Europe Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Country, 2025
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Country, 2036
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Product Type, 2025
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Product Type, 2036
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Sales Channel, 2025
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Sales Channel, 2036
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Application, 2025
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Application, 2036
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Company, 2025
- Eastern Europe Solid Deodorant Sticks Market Share (%) By Company, 2036
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Country
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Application
- Eastern Europe Solid Deodorant Sticks Market Attractiveness Analysis By Company
- East Asia Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- East Asia Solid Deodorant Sticks Market Share (%) By Country, 2025
- East Asia Solid Deodorant Sticks Market Share (%) By Country, 2036
- East Asia Solid Deodorant Sticks Market Share (%) By Product Type, 2025
- East Asia Solid Deodorant Sticks Market Share (%) By Product Type, 2036
- East Asia Solid Deodorant Sticks Market Share (%) By Sales Channel, 2025
- East Asia Solid Deodorant Sticks Market Share (%) By Sales Channel, 2036
- East Asia Solid Deodorant Sticks Market Share (%) By Application, 2025
- East Asia Solid Deodorant Sticks Market Share (%) By Application, 2036
- East Asia Solid Deodorant Sticks Market Share (%) By Company, 2025
- East Asia Solid Deodorant Sticks Market Share (%) By Company, 2036
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Country
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Application
- East Asia Solid Deodorant Sticks Market Attractiveness Analysis By Company
- South Asia and Pacific Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Country, 2025
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Country, 2036
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Product Type, 2025
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Product Type, 2036
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Sales Channel, 2025
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Sales Channel, 2036
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Application, 2025
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Application, 2036
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Company, 2025
- South Asia and Pacific Solid Deodorant Sticks Market Share (%) By Company, 2036
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Country
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Application
- South Asia and Pacific Solid Deodorant Sticks Market Attractiveness Analysis By Company
- Middle East & Africa Solid Deodorant Sticks Market Size (USD Billion), 2021-2036
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Country, 2025
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Country, 2036
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Product Type, 2025
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Product Type, 2036
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Sales Channel, 2025
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Sales Channel, 2036
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Application, 2025
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Application, 2036
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Company, 2025
- Middle East & Africa Solid Deodorant Sticks Market Share (%) By Company, 2036
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Country
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Product Type
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Sales Channel
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Application
- Middle East & Africa Solid Deodorant Sticks Market Attractiveness Analysis By Company
- USA Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Canada Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Mexico Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Brazil Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Chile Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Germany Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- UK Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Italy Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Spain Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- France Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- India Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- ASEAN Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- China Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Japan Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- South Korea Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Russia Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Poland Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Hungary Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Turkiye Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- South Africa Solid Deodorant Sticks Pricing Trend Analysis (USD/Unit), 2021-2036
- Solid Deodorant Sticks Market Competitive Landscape
- Solid Deodorant Sticks Market Structure Analysis
- Solid Deodorant Sticks Market Competition Dashboard
- Solid Deodorant Sticks Market Competition Benchmarking Analysis
- Solid Deodorant Sticks Market Share Analysis of Top Players By Regional
- Solid Deodorant Sticks Market Share Analysis of Top Players By Product Type
- Solid Deodorant Sticks Market Share Analysis of Top Players By Sales Channel
- Solid Deodorant Sticks Market Share Analysis of Top Players By Application
- Solid Deodorant Sticks Market Share Analysis of Top Players By Company
- Unilever - Revenue Analysis (USD Billion), 2019-2025
- Procter & Gamble - Revenue Analysis (USD Billion), 2019-2025
- Henkel - Revenue Analysis (USD Billion), 2019-2025
- Beiersdorf - Revenue Analysis (USD Billion), 2019-2025
- Colgate-Palmolive - Revenue Analysis (USD Billion), 2019-2025
- Unilever - Market Position Analysis
- Procter & Gamble - Market Position Analysis
- Henkel - Market Position Analysis
- Beiersdorf - Market Position Analysis
- Colgate-Palmolive - Market Position Analysis

