Antimicrobial Copper-Infused Filament Yarns Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Antimicrobial Copper-Infused Filament Yarns Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Antimicrobial Copper-Infused Filament Yarns Market Forecast and Outlook 2026 to 2036
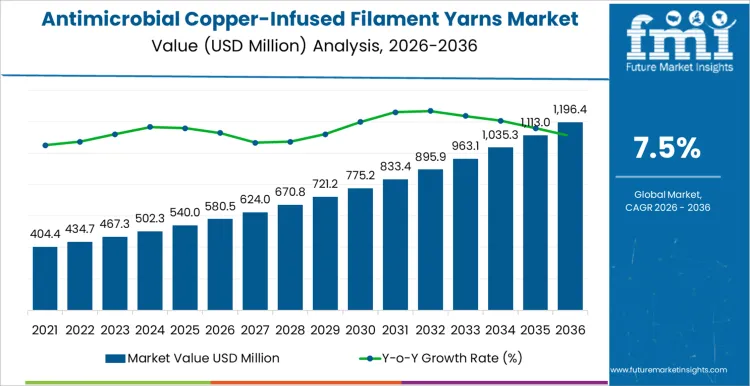
USD 580.5 million represents the opening size of the antimicrobial copper-infused filament yarns market in 2026, with the value projected to reach USD 1,196.4 million by 2036 at a 7.50% CAGR. Demand is established in medical textiles, protective apparel, and hygiene-related fabrics where microbial control forms part of product performance requirements. Material specification is set during fabric and garment development through decisions on copper loading levels, filament construction, and compatibility with finishing processes. Once a textile platform enters approved production status, subsequent yarn substitution requires renewed efficacy testing and regulatory clearance. Volume growth follows expansion of infection control programs, hospital textile procurement, and protective clothing categories.
Access to the antimicrobial copper-infused filament yarns market is governed by specification ownership, validated performance claims, and extended qualification cycles. Market participation depends on documented antimicrobial efficacy, durability through washing, and absence of adverse skin reactions. Approved material sets remain unchanged across long product lifecycles because regulatory, safety, and performance claims attach to specific constructions. Supplier replacement occurs during formal product redesign programs rather than routine sourcing cycles. Revenue development reflects the number of textile programs incorporating antimicrobial functionality. Competitive position depends on early involvement in textile design stages and sustained inclusion within standardized material systems across healthcare, protective apparel, and hygiene-related applications.
Antimicrobial Copper-Infused Filament Yarns Market Quick Stats
- Antimicrobial Copper-Infused Filament Yarns Market Value (2026): USD 580.5 million
- Antimicrobial Copper-Infused Filament Yarns Market Forecast Value (2036): USD 1,196.4 million
- Antimicrobial Copper-Infused Filament Yarns Market Forecast CAGR (2026 to 2036): 7.5%
- Leading Type in Antimicrobial Copper-Infused Filament Yarns Market: Copper-coated filaments
- Key Growth Regions in Antimicrobial Copper-Infused Filament Yarns Market: China, Brazil, USA, South Korea, Japan
- Top Players in Antimicrobial Copper-Infused Filament Yarns Market: Cupron, Noble Biomaterials, Unitika, Toray Industries, Teijin Frontier, Eastman, Wellman Advanced Materials, Toyobo, Coats Group, Ascend Performance Materials
What is the Growth Forecast for the Antimicrobial Copper Infused Filament Yarns Market through 2036?
The antimicrobial copper infused filament yarns market in 2026 stands near USD 580.5 million, anchored in textiles where surface contact and service life define risk management. Buyers specify these yarns for uniforms, healthcare fabrics, transport seating, and interior textiles where cleaning cycles alone do not control microbial load. Product selection depends on copper distribution inside the filament, wash durability, tensile retention, and compatibility with knitting and weaving speeds. Qualification work focuses on abrasion resistance, color stability, and measured activity after repeated laundering. Adoption follows program approvals rather than short purchasing windows. The expansion toward USD 1,196.4 million in 2036 aligns with a 7.50% growth profile supported by wider integration into base fabric constructions and higher yarn content per square meter in high contact surfaces.
Commercial practice shapes the spending pattern of the antimicrobial copper infused filament yarns market. Orders link to uniform contracts, fleet refresh cycles, and long production campaigns. Inventory policy favors continuity of approved grades and predictable lot behavior. Cost structure reflects copper input management, spinning control, dispersion quality, and inspection load. Product teams value repeatable performance data and stable processing more than marginal price differences. Annual values move through the upper hundreds of millions and into the low billions during the early 2030s as more textile programs convert core specifications. The rise to USD 1,196.4 million in 2036 comes from broader placement in public use textiles, thicker constructions in some end uses, and longer supply agreements that place these yarns into fixed bills of material across multiple fabric platforms.
Antimicrobial Copper-Infused Filament Yarns Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 580.5 million |
| Forecast Value (2036) | USD 1,196.4 million |
| Forecast CAGR (2026 to 2036) | 7.5% |
How Is the Antimicrobial Copper Infused Filament Yarns Market Being Used to Control Surface Hygiene and Service Life in Textile Applications?
The antimicrobial copper infused filament yarns market is specified where hygiene performance must remain stable through repeated use and cleaning cycles. Producers supplying hospital privacy curtains evaluate these yarns for wash durability and contact safety because loss of activity shows up in audit findings. Manufacturers serving medical compression garments examine tensile stability and skin contact behavior after multiple laundering steps. Programs linked to antimicrobial upholstery fabrics focus on abrasion resistance and uniform copper distribution across filaments. Teams developing odor control sportswear track long term effectiveness under sweat and heat exposure. Buyers involved in protective workwear textiles review corrosion behavior and discoloration risk. Qualification centers on controlled release behavior, filament integrity, and coating adhesion because inconsistent performance forces product downgrades and reapproval cycles.
The antimicrobial copper infused filament yarns market is managed through compliance testing and service life planning rather than through single cycle performance claims. Operations supplying public transport seating materials measure value through maintenance intervals and surface inspection results. Producers active in cleanroom furnishing textiles track particle shedding and electrostatic behavior because both affect acceptance audits. Programs connected to bedding and mattress covers link material choice to laundering tolerance and complaint rates. Storage and handling rules receive attention since oxidation and moisture exposure change surface behavior before knitting or weaving. Contracts define acceptance limits for antimicrobial persistence, color stability, and filament breakage. Demand concentrates in applications where documented hygiene performance, repeat cleaning, and inspection outcomes determine continued supply approval rather than initial product testing alone.
How do claim substantiation and service environment requirements shape demand in the Antimicrobial Copper-Infused Filament Yarns Market by copper integration and application in 2026?
In the Antimicrobial Copper-Infused Filament Yarns Market, specifications begin with efficacy retention, wash durability, and skin contact limits rather than yarn price. Programs serving healthcare textile production, workwear fabric manufacturing, and bedding and mattress textiles require constructions that keep copper available at the surface while preserving tensile and handle. By copper integration, usage spans copper-coated filaments, copper-embedded polymer yarns, copper-alloy filaments, and other methods, each showing different release profiles and abrasion response. By application, consumption covers healthcare and medical textiles, workwear and public-contact textiles, home and bedding, and other uses. Once a construction clears trials and enters technical files, substitution triggers new testing and customer approvals, which favors scale inside approved designs.
Why do copper-coated and embedded constructions dominate approved platforms?

Copper-coated filaments hold about 36% share because they place active material at the surface where contact occurs while keeping the base polymer unchanged. In healthcare textile production and hospital linen systems, this approach simplifies performance verification through surface sampling and repeated laundering. Copper-embedded polymer yarns protect the active phase from abrasion and extend service life in workwear fabric manufacturing where mechanical wear is high. Copper-alloy filaments suit programs that accept higher stiffness for long-term exposure. Other methods remain tied to narrow equipment windows. Engineers screen options through abrasion loss, ion availability, and tensile after washing. Process teams screen them through dyeing behavior and lint. Quality teams screen them through batch identity and aging. A method that clears these gates becomes standard.
Once selected, the integration method becomes embedded in spinnerets, coating heads, and finishing routes. Changing it forces new line trials, new washing matrices, and new documentation. Sampling plans and acceptance limits must be rewritten. Planning then protects the approved route with dedicated schedules. Inventory rules prevent mixing to avoid label disputes. The commercial outcome favors repeating the same construction across counts and colors. Capacity grows by adding positions rather than altering integration method. This discipline explains the persistence of surface-present and matrix-embedded approaches in platforms where verification and durability outweigh novelty.
How do care protocols and exposure patterns lock in application demand?
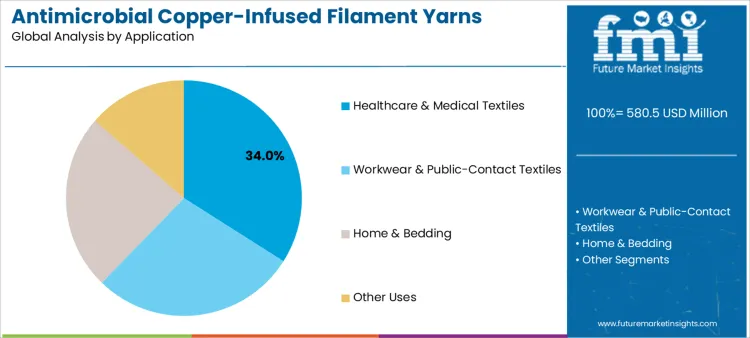
Healthcare and medical textiles represent about 34% of demand due to acceptance depends on consistent contact activity after repeated institutional laundering. In hospital linen systems, approvals link yarn behavior to wash profiles, drying temperatures, and handling cycles. Workwear and public-contact textiles follow where abrasion and sweat exposure define performance in uniform and protective apparel programs. Home and bedding use these yarns where skin contact and odor control matter in bedding and mattress textiles. Other uses remain tied to site-specific needs. Converters approve routes through knitting or weaving stability, finishing yield, and shade control. Compliance teams define sampling and record retention. Service teams define replacement intervals and storage rules. Once a route clears these checks, it becomes fixed to a narrow window.
Switching application resets trials and customer files. Tooling and kitting layouts also change. Training and inspection sheets must be updated. Supervisors then protect the approved route with dedicated schedules. Orders expand by adding shifts and widths rather than changing constructions. Demand follows applications where care protocols and exposure patterns govern performance expectations more than short purchasing cycles.
Why Are Functional Performance Demands Supporting the Antimicrobial Copper-Infused Filament Yarns Market?
Antimicrobial Copper-Infused Filament Yarns Market demand is driven by product specifications that require inherent protection against microbial presence without additional processing steps. Filament yarns infused with copper particles provide continuous contact-based antimicrobial performance in textiles for medical linens, protective apparel, sportswear, and high-traffic public fabrics. Manufacturers evaluate yarns based on copper loading uniformity, filament integrity, and retention of mechanical properties to ensure performance does not degrade over product life. Buyers prioritize consistent antimicrobial efficacy data across batches and compatibility with knitting, weaving, and finishing lines. Value is measured in reduced post-production treatment steps and predictable yarn behavior during fabrication.
What Factors Are Limiting Adoption and Scale in the Antimicrobial Copper-Infused Filament Yarns Market?
Antimicrobial Copper-Infused Filament Yarns Market growth is constrained by production cost premiums, copper dispersion challenges, and qualification cycles required by converters and end-use OEMs. Integrating copper into filament polymers affects melt flow and filament strength, which requires careful control of compounding and spinning parameters. Uniform distribution of copper within filaments is essential to maintain antimicrobial effectiveness without creating weak points that compromise mechanical performance. Higher material costs relative to untreated yarns affect adoption in price-sensitive segments such as everyday apparel and commodity textiles. Qualification time for new grades, including testing for durability after washing and processing, extends product launch timelines for converters and brand owners.
How Are Product Formats and Commercial Practices Evolving in the Antimicrobial Copper-Infused Filament Yarns Market?
Antimicrobial Copper-Infused Filament Yarns Market offerings are being structured around performance bands defined by copper content, filament denier, and processing behavior to simplify selection for converters. Suppliers provide detailed data sheets that include antimicrobial efficacy levels, mechanical properties, and recommended processing windows for knitting, weaving, and finishing lines. Portfolio alignment around common fabric applications such as medical textiles, activewear, and upholstery reduces selection uncertainty and lowers trial runs. Commercial engagement increasingly includes supply agreements that secure copper feedstocks and planned filament output to match production schedules. Technical support focuses on process mapping, defect avoidance, and maintaining yarn performance through downstream operations to help customers achieve consistent product quality.
What is the Demand for Antimicrobial Copper Infused Filament Yarns by Country?
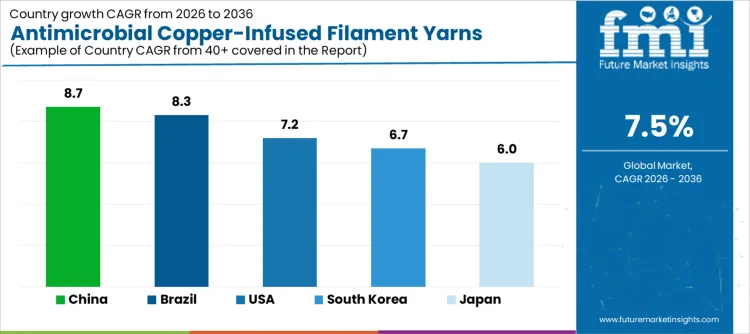
| Country | CAGR (%) |
|---|---|
| USA | 7.2% |
| Japan | 6.0% |
| South Korea | 6.7% |
| China | 8.7% |
| Brazil | 8.3% |
The demand for antimicrobial copper infused filament yarns is increasing across healthcare textiles, sportswear, and interior fabrics where odor control and surface hygiene are functional requirements. China leads at 8.7% CAGR, supported by large scale textile manufacturing and wider use of antimicrobial fabrics in medical and consumer products. Brazil follows at 8.3%, reflecting growth in functional apparel and institutional textiles. The USA grows at 7.2%, driven by adoption in healthcare linens, performance clothing, and bedding products. South Korea records 6.7%, shaped by steady demand from sportswear and technical textile producers. Japan posts 6.0%, within a mature market that emphasizes quality, durability, and controlled integration of antimicrobial features into finished textile products.
How Do Infection Control Standards Shape Procurement in the Antimicrobial Copper Infused Filament Yarns Market in the USA?
In the United States, the Antimicrobial Copper Infused Filament Yarns Market is advancing in line with a 7.2% CAGR. Procurement decisions are anchored in infection control standards that define acceptable antimicrobial efficacy, wash durability, and mechanical stability in end use textiles. Evaluation programs measure bacterial reduction rates, copper distribution uniformity, tensile retention, and color stability after repeated laundering. Once a yarn grade clears these protocols, the same approval documentation governs downstream contracts and material substitution becomes uncommon. Volume increases only when approved healthcare, institutional, or performance apparel programs expand production or introduce new product lines. Technical committees retain authority over any specification change. Supplier access depends on documentation completeness and audit outcomes. Inventory policies follow program schedules rather than spot demand. Commercial success depends on specification inclusion and consistent batch performance across multiple manufacturing sites.
How Do Manufacturing Governance Rules Determine Adoption in the Antimicrobial Copper Infused Filament Yarns Market in Japan?
Within Japanese operations, material governance is directing the Antimicrobial Copper Infused Filament Yarns Market along a path consistent with a 6% CAGR. Yarn selection is treated as a manufacturing control variable rather than a procurement preference. Any proposed change enters a formal review cycle that includes extended antimicrobial performance checks and surface stability assessments. Engineers examine copper dispersion, filament cohesion, abrasion resistance, and tolerance to processing stress. Once approved, the same yarn remains embedded in identical constructions for extended production cycles. Change requests trigger full revalidation procedures. Supplier access depends on documentation completeness and inspection outcomes. Local sources benefit from proximity to audit teams and quality oversight groups. Volume growth follows gradual portfolio expansion rather than replacement initiatives. Commercial positioning relies on procedural discipline, documentation rigor, and sustained performance verification under controlled production conditions.
How Do Platform Based Product Architectures Drive Scale in the Antimicrobial Copper Infused Filament Yarns Market in South Korea?
Platform oriented product strategy explains why the Antimicrobial Copper Infused Filament Yarns Market in South Korea is progressing in line with a 6.7% CAGR. Development organizations define standard yarn sets that apply across several product families. A single material decision can influence numerous lines without separate approvals. Evaluation centers on antimicrobial effectiveness, copper retention, tensile stability, and compatibility with existing finishing processes. When a yarn enters the platform baseline, scale follows through synchronized product introductions. Plant level changes remain limited because the platform file governs material use. Suppliers secure positions through joint testing and alignment with documentation requirements. Volume growth mirrors platform rollout schedules. Inventory planning aligns with coordinated manufacturing programs. Commercial success depends on early inclusion in platform definitions and stable execution during multi line production deployments.
How Do Central Approval Registers Control Material Choice in the Antimicrobial Copper Infused Filament Yarns Market in China?
Replication across industrial supply networks explains why the Antimicrobial Copper Infused Filament Yarns Market in China is advancing in line with an 8.7% CAGR. Material access is controlled through centralized approval systems used by large textile groups. Once a yarn performs within defined antimicrobial and processing limits in one facility, it is introduced into parallel operations with limited redesign. Reviews focus on consistency, copper content control, and tolerance to high volume production. Domestic suppliers dominate because they can follow capacity additions and regional program expansion. Imports remain concentrated in higher specification applications. Volume growth results from repeating approved constructions across sites. Commercial advantage depends on being present when a product configuration becomes a reference template rather than competing for each individual facility.
How Do Public Health Programs and Institutional Procurement Shape Demand in the Antimicrobial Copper Infused Filament Yarns Market in Brazil?
In Brazil, the Antimicrobial Copper Infused Filament Yarns Market is expanding at a rate aligned with an 8.3% CAGR. Demand is closely linked to public health programs and institutional textile procurement policies. Buyers evaluate yarns based on antimicrobial efficacy, durability under repeated washing, and mechanical stability in uniforms and bedding. Once a yarn grade is approved, it becomes part of standard supply specifications used across multiple facilities. Material changes require renewed validation. Supplier access depends on documentation quality and audit performance. Local distributors play a role in qualification support and logistics execution. Imports supply higher performance requirements. Volume growth follows program expansion and new facility commissioning. Commercial success depends on meeting institutional standards and maintaining consistent performance across high volume production and distribution networks.
How Do Suppliers Compete in the Antimicrobial Copper-Infused Filament Yarns Market?
Antimicrobial performance, fiber durability, and integration with textile processes determine adoption in the Antimicrobial Copper-Infused Filament Yarns Market. Cupron, Noble Biomaterials, Unitika, Toray Industries, Teijin Frontier, Eastman, Wellman Advanced Materials, Toyobo, Coats Group, and Ascend Performance Materials compete during pilot spinning, fiber testing, and fabric production trials. Once incorporated into medical textiles, protective apparel, or performance garments, copper distribution, wash durability, and mechanical properties anchor supplier selection. Cupron emphasizes proprietary copper complexes for consistent antimicrobial activity. Noble Biomaterials focuses on healthcare and high-contact surfaces. Unitika supplies filament yarns with controlled copper loading. Toray Industries and Teijin Frontier integrate copper fibers with high-performance polymers. Eastman provides alloy fibers through partner networks. Wellman and Toyobo target industrial and functional apparel. Coats Group focuses on coated filament yarns for sewn applications. Ascend Performance Materials delivers nylon-based fibers with embedded copper functionality.
Material consistency, process compatibility, and regulatory compliance shape competitive positioning in the Antimicrobial Copper-Infused Filament Yarns Market. Manufacturers evaluate copper ion release, fiber tensile strength, wash retention, and filament uniformity during qualification. Production teams prioritize suppliers capable of reproducible lot quality, scalable supply, and technical guidance for fabric integration. Cupron secures adoption through healthcare partnerships and antimicrobial validation data. Noble Biomaterials maintains share in medical and hygiene applications. Unitika competes in industrial and protective textiles. Toray Industries and Teijin Frontier hold positions in high-performance apparel requiring fiber-level antimicrobial properties. Eastman supports partner-based alloy fiber programs. Wellman Advanced Materials and Toyobo capture adoption in functional clothing and industrial textiles. Coats Group gains placements in sewn products requiring antimicrobial thread. Ascend Performance Materials serves technical and performance-oriented textile programs. Market positions vary by application, fiber polymer type, regulatory standards, and production scale.
Key Players in Antimicrobial Copper-Infused Filament Yarns Market
- Cupron
- Noble Biomaterials
- Unitika
- Toray Industries
- Teijin Frontier
- Eastman (alloy fibers via partners)
- Wellman Advanced Materials
- Toyobo
- Coats Group
- Ascend Performance Materials
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Copper Integration | Copper-coated filaments, copper-embedded polymer yarns, copper-alloy filaments, other copper integration methods |
| Application | Healthcare and medical textiles, workwear and public-contact textiles, home and bedding, other uses |
| Fiber Type | Polyester, polyamide, other fibers |
| Customer Segment | Healthcare and institutional, consumer brands, technical textile mills, other customers |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordics, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Cupron, Noble Biomaterials, Unitika, Toray Industries, Teijin Frontier, Eastman, Wellman Advanced Materials, Toyobo, Coats Group, Ascend Performance Materials |
| Additional Attributes | Dollar sales by copper integration, application, fiber type, and customer segment, antimicrobial efficacy retention after repeated washing, copper loading and dispersion control, abrasion resistance and color stability performance, integration into healthcare, workwear, and bedding textiles, regulatory and skin-contact compliance requirements, long qualification and reapproval cycles tied to infection control standards, and regional demand patterns driven by healthcare procurement, public-contact textiles, and functional apparel programs |
Antimicrobial Copper-Infused Filament Yarns Market Segmentation
Copper Integration:
- Copper-Coated Filaments
- Copper-Embedded Polymer Yarns
- Copper-Alloy Filaments
- Other Copper Integration Methods
Application:
- Healthcare & Medical Textiles
- Workwear & Public-Contact Textiles
- Home & Bedding
- Other Uses
Fiber Type:
- Polyester
- Polyamide
- Other Fibers
Customer Segment:
- Healthcare & Institutional
- Consumer Brands
- Technical Textile Mills
- Other Customers
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2024). Global report on infection prevention and control 2024. World Health Organization.
- International Organization for Standardization. (2019). ISO 20743: Textiles—Determination of antibacterial activity of textile products. International Organization for Standardization.
- United States Environmental Protection Agency. (2023). Copper and copper alloy surfaces: Antimicrobial efficacy and public health implications. U.S. Environmental Protection Agency.
Frequently Asked Questions
How big is the antimicrobial copper-infused filament yarns market in 2026?
The global antimicrobial copper-infused filament yarns market is estimated to be valued at USD 580.5 million in 2026.
What will be the size of antimicrobial copper-infused filament yarns market in 2036?
The market size for the antimicrobial copper-infused filament yarns market is projected to reach USD 1,196.4 million by 2036.
How much will be the antimicrobial copper-infused filament yarns market growth between 2026 and 2036?
The antimicrobial copper-infused filament yarns market is expected to grow at a 7.5% CAGR between 2026 and 2036.
What are the key product types in the antimicrobial copper-infused filament yarns market?
The key product types in antimicrobial copper-infused filament yarns market are copper-coated filaments, copper-embedded polymer yarns, copper-alloy filaments and other copper integration methods.
Which application segment to contribute significant share in the antimicrobial copper-infused filament yarns market in 2026?
In terms of application, healthcare & medical textiles segment to command 34.0% share in the antimicrobial copper-infused filament yarns market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Copper Integration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Copper Integration , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Copper Integration , 2026 to 2036
- Copper-Coated Filaments
- Copper-Embedded Polymer Yarns
- Copper-Alloy Filaments
- Other Copper Integration Methods
- Copper-Coated Filaments
- Y-o-Y Growth Trend Analysis By Copper Integration , 2021 to 2025
- Absolute $ Opportunity Analysis By Copper Integration , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Healthcare & Medical Textiles
- Workwear & Public-Contact Textiles
- Home & Bedding
- Other Uses
- Healthcare & Medical Textiles
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Fiber Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Fiber Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Fiber Type, 2026 to 2036
- Polyester
- Polyamide
- Other Fibers
- Polyester
- Y-o-Y Growth Trend Analysis By Fiber Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Fiber Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Customer Segment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Segment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Segment, 2026 to 2036
- Healthcare & Institutional
- Consumer Brands
- Technical Textile Mills
- Other Customers
- Healthcare & Institutional
- Y-o-Y Growth Trend Analysis By Customer Segment, 2021 to 2025
- Absolute $ Opportunity Analysis By Customer Segment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- By Country
- Market Attractiveness Analysis
- By Country
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- By Country
- Market Attractiveness Analysis
- By Country
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- By Country
- Market Attractiveness Analysis
- By Country
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- By Country
- Market Attractiveness Analysis
- By Country
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- By Country
- Market Attractiveness Analysis
- By Country
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- By Country
- Market Attractiveness Analysis
- By Country
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- By Country
- Market Attractiveness Analysis
- By Country
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Copper Integration
- By Application
- By Fiber Type
- By Customer Segment
- Competition Analysis
- Competition Deep Dive
- Cupron
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Noble Biomaterials
- Value (USD Million)ika
- Toray Industries
- Teijin Frontier
- Eastman (alloy fibers via partners)
- Wellman Advanced Materials
- Toyobo
- Coats Group
- Ascend Performance Materials
- Cupron
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Copper Integration , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Fiber Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Customer Segment, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Copper Integration , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Fiber Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Customer Segment, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Copper Integration , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Fiber Type, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Customer Segment, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Copper Integration , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Fiber Type, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Customer Segment, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Copper Integration , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Fiber Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Customer Segment, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Copper Integration , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Fiber Type, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Customer Segment, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Copper Integration , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Fiber Type, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Customer Segment, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Copper Integration , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Fiber Type, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Customer Segment, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Copper Integration , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Copper Integration , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Copper Integration
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Fiber Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Fiber Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Fiber Type
- Figure 12: Global Market Value Share and BPS Analysis by Customer Segment, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Customer Segment, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Customer Segment
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Copper Integration , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Copper Integration , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Copper Integration
- Figure 29: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Application
- Figure 32: North America Market Value Share and BPS Analysis by Fiber Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Fiber Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Fiber Type
- Figure 35: North America Market Value Share and BPS Analysis by Customer Segment, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Customer Segment, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Customer Segment
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Copper Integration , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Copper Integration , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Copper Integration
- Figure 42: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Application
- Figure 45: Latin America Market Value Share and BPS Analysis by Fiber Type, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Fiber Type, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Fiber Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Customer Segment, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Customer Segment, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Customer Segment
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Copper Integration , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Copper Integration , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Copper Integration
- Figure 55: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Application
- Figure 58: Western Europe Market Value Share and BPS Analysis by Fiber Type, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Fiber Type, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Fiber Type
- Figure 61: Western Europe Market Value Share and BPS Analysis by Customer Segment, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Customer Segment, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Customer Segment
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Copper Integration , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Copper Integration , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Copper Integration
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Application
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Fiber Type, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Fiber Type, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Fiber Type
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Customer Segment, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Customer Segment, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Customer Segment
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Copper Integration , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Copper Integration , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Copper Integration
- Figure 81: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Application
- Figure 84: East Asia Market Value Share and BPS Analysis by Fiber Type, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Fiber Type, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Fiber Type
- Figure 87: East Asia Market Value Share and BPS Analysis by Customer Segment, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Customer Segment, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Customer Segment
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Copper Integration , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Copper Integration , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Copper Integration
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Fiber Type, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Fiber Type, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Fiber Type
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Customer Segment, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Customer Segment, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Customer Segment
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Copper Integration , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Copper Integration , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Copper Integration
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Fiber Type, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Fiber Type, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Fiber Type
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Customer Segment, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Customer Segment, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Customer Segment
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

