Coronary Stents Market
The coronary stents market is segmented by Product Type (Drug Eluting Stents (DES), Bare Metal Stents (BMS), Bioabsorbable Stents, Self-expanding Stents, Balloon-expandable Stents, etc.), End User (Hospitals, Cardiac Centers, Ambulatory Surgical Centers, and Other Healthcare Settings), Raw Material (Cobalt-Chromium, Stainless Steel, Platinum-Chromium, Nickel-Titanium, Polymer) and Region (North America, Latin America, Western Europe, South Asia and Pacific, East Asia, Middle East and Africa). Forecast for 2026 to 2036.
Coronary Stents Market Size, Market Forecast and Outlook By FMI
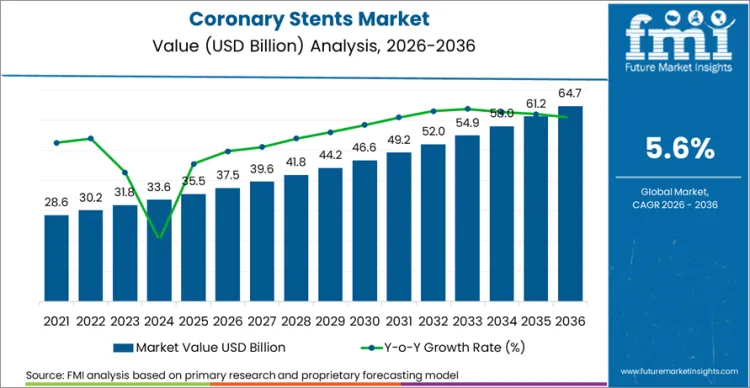
The coronary stents market is expected to expand from USD 37.5 billion in 2026 to USD 64.6 billion by 2036. The market is anticipated to register 5.6% CAGR during the forecast period as PCI procedures retain an important role in coronary artery disease treatment. Drug-eluting stents are likely to lead with 33.2% share in 2025 and hospitals are projected to account for 27.1% share in 2025.
Summary of the Coronary Stents Market
- Demand and Growth Drivers
- High cardiovascular disease incidence is expected to sustain procedure demand across catheterization laboratories and interventional cardiology departments.
- Preference for minimally invasive revascularization is anticipated to lift stent use among patients needing faster recovery after PCI.
- Clinical use of improved DES platforms is likely to reduce restenosis concerns and strengthen physician confidence in complex lesion treatment.
- Product and Segment View
- Drug-eluting stents are expected to lead product demand as medication release helps manage scar tissue formation after implantation.
- Hospitals are likely to keep the leading end user position since advanced cath labs handle complex cardiac interventions.
- Bioabsorbable stents are anticipated to gain physician attention as scaffold designs improve temporary vessel healing and long-term safety profiles.
- Geography and Competitive Outlook
- The United States is expected to keep a significant value share through premium reimbursement and advanced PCI procedure capacity.
- Italy is likely to record the fastest listed country CAGR as hospital access and cardiac procedure use improve steadily.
- Large device companies are anticipated to compete through flexible DES delivery systems and better clinical evidence for bioresorbable scaffolds.
- Analyst Opinion
- Sabyasachi Ghosh, is Principal Consultant for Healthcare at FMI and suggests, “The coronary stents market is expected to favor manufacturers with strong DES portfolios and credible scaffold data. Hospitals are likely to prefer platforms backed by evidence across complex coronary anatomy and predictable post-procedure outcomes.”
- Coronary Stents Market Value Analysis
- The coronary stents market is moving from established metallic devices toward a broader mix of DES and bioresorbable scaffolds.
- Demand is likely to increase as coronary artery disease burden keeps PCI procedures central to interventional cardiology practice.
- Adoption is expected to rise through low-profile delivery systems designed for multi-vessel disease and difficult coronary anatomy.
- Spending is anticipated to be shaped by premium reimbursement access and longer clinical validation requirements for advanced stent platforms.
Summary of Coronary Stents Market
What Is Growth Outlook for Coronary Stents Market as per Future Market Insights Projection?
Future Market Insights projects the coronary stents market to expand at a CAGR of 5.6% from 2026 to 2036, increasing from USD 37.5 Billion in 2026 to USD 64.6 Billion by 2036.
FMI Research Approach: FMI proprietary forecasting model based on PCI procedure volumes and DES/BVS adoption rates.
How Do FMI Analysts Perceive Coronary Stents Market to Evolve?
FMI analysts perceive the market evolving toward a dual-platform structure where established metallic DES systems continue as the high-volume standard while bioresorbable scaffolds gain share in select complex lesion applications.
FMI Research Approach: MicroPort Firesorb BVS launch (September 2025) and Abbott XIENCE Sierra clinical data.
Which Country Holds Largest Share in Global Coronary Stents Market?
The United States holds a significant share of the global coronary stents market by value, which is supported by the highest per-capita PCI procedural rate and premium reimbursement for next-generation DES platforms.
FMI Research Approach: FMI country-level revenue modeling by PCI procedure volumes and CMS reimbursement rates.
How Large Will Coronary Stents Market Be by 2036?
The global coronary stents market is projected to reach USD 64.6 Billion by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from WHO cardiovascular disease burden projections and PCI volume growth rates.
What Is Definition of Coronary Stents Market?
The coronary stents market includes revenue from drug-eluting stents (DES), bare-metal stents (BMS), and bioresorbable vascular scaffolds (BVS) used in percutaneous coronary intervention procedures for coronary artery disease treatment.
FMI Research Approach: FMI market taxonomy aligned with FDA and CE-mark device classifications.
What Are Globally Unique Trends Shaping Coronary Stents Market?
Globally unique trends include the re-emergence of bioresorbable scaffold technology with improved clinical outcomes, the launch of ultra-low-profile DES platforms for complex anatomies, and the prioritization of emerging markets for new stent introductions.
FMI Research Approach: MicroPort Firesorb launch (September 2025) and Abbott XIENCE Sierra India launch (May 2024).
Coronary Stents Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 37.5 Billion |
| Industry Value (2036) | USD 64.6 Billion |
| CAGR (2026 to 2036) | 5.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segment-wise Analysis
By Product
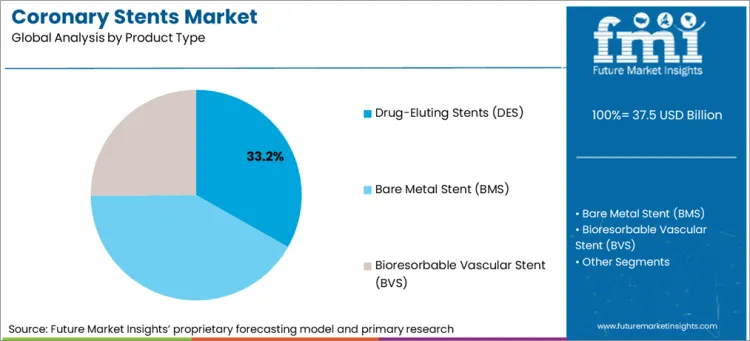
| Attributes | Details |
|---|---|
| Top Source Type | Drug-Eluting Stents (DES) |
| Market Share in 2025 | 33.2% |
Drug-eluting stents (DES) is expected to account for a leading coronary stent market share of 33.2% in 2025. The Drug Eluting Stent has the capability to prevent the risks of restenosis (the process of the arteries getting smaller again), which is known to be caused by scar tissue, and it does this by means of drugs that prevent the production of the scar tissue. The rise in coronary artery disease, which is a common health problem, has resulted in a demand for the newest stent technologies like DES. These particular types of stents perform better in challenging cases of surgery.
By End User
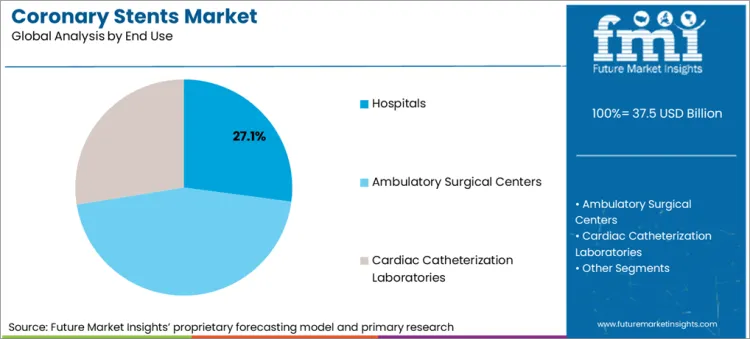
| Attributes | Details |
|---|---|
| Top Source Type | Hospitals |
| Market Share in 2025 | 27.1% |
Hospitals are poised to occupy 27.1% of the coronary stents market in 2025 due to the advanced clinical facilities. Hospitals, particularly the larger medical center types, are outfitted with the newest equipment and have specialized cardiology departments, therefore, they are the best choice for complex heart procedures.
On the other hand, the adoption of new technologies by manufacturing for stent models ensures the production of higher quality products while minimizing environmental impact, waste generation, energy consumption, and other costs. Hospitals primarily offer after-procedure care like dosage control, angiogram as a follow-up treatment, and lifestyle changes advice which together, help in the recovery of patients and also promote the stay of the stents in the bodies of the patients, especially in the healthcare systems that are good.
Key Drivers
High Prevalence of Cardiovascular Diseases Pushes the Demand for Minimally Invasive Procedures
Cardiovascular diseases (CVDs) have become more common worldwide, and the need for coronary stents has grown exponentially, throughout the years of the pandemic. The predominant risk factors are poor diet, smoking, sedentary lifestyle, and stress. According to World Health Organization (WHO) CVDs are primarily responsible for premature deaths among the population with about 18 million fatalities every year.
The coronary stent sector is seeing technological progress with items like drug-eluting stents (DES) which are capable of discharging medicine meant to obstruct restenosis, biodegradable stents that erode after a certain period of time, and state-of-the-art imaging equipment like OCT (optical coherence tomography) which comes in handy for proper stent placement.
Technological Advancements in Stent Design Improves Patient Outcomes
Technological advancements in stent design are significantly improving patient outcomes. Drug-eluting stents (DES) release medication that prevents tissue overgrowth, reducing restenosis rates compared to traditional bare-metal stents. Bioabsorbable stents, which dissolve over time, offer the advantage of eliminating long-term complications.
Additionally, advanced coatings have improved biocompatibility and minimizes the risks of stent thrombosis. Healthcare providers have turned to coronary stents due to different innovations in the sector such as Drug Eluting Stents, Biodegradable Stents and Bioresorbable Vascular scaffolds which give higher effectivity and safety.
Stringent Regulatory Approval Processes Challenges Product Launch Strategies
| Aspect | Details |
|---|---|
| Complex Regulations | Coronary stents are objects that are directly placed inside the human body. They are strictly bound to the safety and efficacy standards. From a regulatory aspect organizations such as FDA (USA), EMA (Europe), and CDSCO (India), underlines extensive clinical trials and tight testing to be conducted prior any approval. A manufacturer undergoing the approval period often faces a complex procedure that might negatively impact their market entry. |
| Cost of Compliance | Significant expenditure is involved in compliance with regulatory standards that often impacts the overall operations. It is imperative that production enterprises incur cost investments for research, clinical trials, and meet sustainability standards. The aforementioned factors may be the rationale for the high overall product price and postponement of the product launch. |
| Regional Variations | The cross-market strategies of companies are hampered by regional variances in disease prevalence, health care systems and economic factors thus rendering it harder for companies to implement the same strategies across the board. They have to tailor their products, pricing, and regulations to the requirements of every market. |
| Impact | The strict regulatory framework can slow innovation and limit the availability of new products. For smaller manufacturers, the barriers to entry can restrict competition, ultimately affecting the variety of treatment options available to patients. |
High Costs and Limited Accessibility Hinders Availability
| Aspect | Details |
|---|---|
| Pricing Challenges | The pricing of state-of-the-art coronary stents, especially drug-eluting and bioabsorbable is on the higher side as they involve advanced technology and materials. These elevated prices make the products unaffordable thus, stents are inaccessible to many parts of the population, particularly in underdeveloped countries and even some middle-income countries. The costs involved can influence patient decision to choose stent therapies. |
| Reimbursement Limitations | Reimbursement restrictions manifested in the coronary stents market due to the existence of different healthcare policies in different areas, which frequently set restrictions on the use of high-cost stents like drug-eluting and biodegradable ones. These limitations cause patients to be dependent on cheaper bare-metal stents thus, affecting the market growth for CRT and innovative technologies. |
| Health Disparities | Rural or underserved spaces often lack access to high-end cardiac care including stent implantation. The deficit of adequate resources and capital is the main reason for the increased obstructions in healthcare in these areas. Hence, the population from such areas lack access to life-saving techniques leading to fatality from cardiovascular diseases. |
| Impact | The capacity of advanced cardiac care like stenting is very low in rural and underserved parts.. The absence of necessary facilities and qualified personnel in these parts is the major reason why healthcare disparities are so acute.. |
Key Trends
The increasing acceptance of bioabsorbable stents (also referred to as bioresorbable vascular scaffolds or BVS) is a notable trend. These state-of-the-art stents outperform conventional permanent stents in terms of dissolving cells over time, which results in no permanent implant in the body.
- Biodegradability & Temporary Support: Contrary to permanent stents, bioabsorbable stents offer temporary support to the artery, allowing it to replenish itself naturally. This helps to avoid future complications such as chronic inflammation, late stent thrombosis, and vessel restenosis that could occur due to the stiff nature of traditional stents.
- Improved Patient Outcomes: Because they do not involve a permanent implant, bioabsorbable stents eliminate the risk of complications and instead foster the proper function of the artery which, in turn, accelerates the restoration of normal vascular function and flexibility.
- Technological Advancements: Progresses in materials (e.g., polylactic acid or magnesium alloy) is improving the performance of bioabsorbable stents. Drug-eluting coatings are also added to enhance effectiveness and reduce the rate of restenosis.
- Research and Development Surge: The primary companies such as Abbott Laboratories and Boston Scientific are set to provide massive sums in support for the development of next-gen bioabsorbable stents.
- Market Expansion: The rapid growth of bioabsorbable stents at a faster rate compared to other stent types is anticipated due to the increasing number of patients' requests for such devices and the approval from regulatory authorities.
Country-wise Analysis
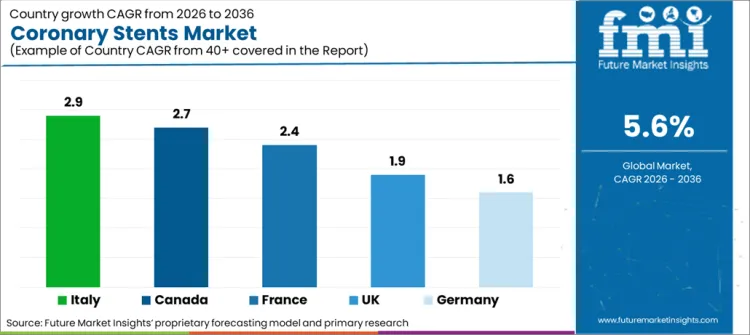
| Countries | CAGR |
|---|---|
| The USA | 1.5% |
| Canada | 2.7% |
| Germany | 1.6% |
| France | 2.4% |
| Italy | 2.9% |
| UK | 1.9% |
The USA dominates the coronary stents market with a sizeable share of global sales. Factors such as availability of an advanced medical care system and the high prevalence of cardiovascular diseases (CVDs) in the population. The popularity of drug-eluting and bioabsorbable stents is leading to high demand.
Abbott Laboratories and Boston Scientific are significantly invest in innovations in the field of high-technology drug-eluting stents, bioresorbable stents, and next-gen imaging devices, particularly. Abbott's Xience drug-eluting stent and Boston Scientific's Synergy stent are both, demonstrating the development methods for the leading-edge innovation of coronary stents.
Researchers and manufacturers are focusing on combining imaging, stent technology, and personalized medicine to improve outcomes. This includes developing advanced imaging tools for better stent placement and creating stents tailored to individual patient needs.
Europe is the fast-growing market thanks to its advanced healthcare system and a high rate of lifestyle-related health issues, Germany is leading the European coronary stents market. The government and the private companies are dedicated to improving medical devices through innovation by provide funding, resources, and incentives to support the development of new technologies in healthcare. Angioplasty and stent procedures are widely accessible through health insurance.
Competitive Landscape
The coronary stent market has made impressive strides of progress throughout the past years in factors such as new product launches, market entrants, and the changing regulatory landscape.
Elunir-Perl Drug-Eluting Stent by Medinol
Elunir-Perl was FDA approved in October 2023 and is a new stent for coronary artery disease that is designed to provide patients with a safe and effective treatment.
Xience Sierra by Abbott Laboratories
Xience Sierra was FDA approved in 2018 and is a drug-eluting stent which features a lower profile and better flexibility and is produced in small diameters and longer lengths. It's meant to assist complex percutaneous coronary interventions (PCI), thus aiding in a better patient outcome.
Resolute Onyx 2.0mm by Medtronic
Resolute Onyx 2.0mm was FDA approved in 2018. It is a tiny drug-eluting stent that is made for patients with small coronary arteries thus making it more effective in treating the problems of patients with complicated anatomy.
Recent Developments
- In September 2025, MicroPort launched Firesorb, its next-generation bioresorbable vascular scaffold, following successful multi-year clinical follow-ups in the FUTURE study series.
- In May 2024, Abbott launched its latest-generation XIENCE Sierra drug-eluting stent in India, featuring an ultra-low profile and high-flexibility delivery system for complex coronary anatomies.
- Boston Scientific reported that 2025 was an exceptional performance year with its interventional cardiology division delivering differentiated innovation that exceeded growth targets.
Emerging Startups and Innovations
Emerging startups are driving the advancement of bioresorbable stent technology; they are now targeting next-generation solutions, which are the ones that naturally dissolve in the body; this way, there is no need for permanent implants. These stents are a safe alternative to metal stents as they prevent complications like late stent thrombosis and are actually safer than traditional metal stents. A few notable companies in the field include:
- BIOTRONIK: This medical technology company is making advancements in bioresorbable vascular scaffolds, which, as per the reports of 2024, have been subjected to successful clinical trials and have also received regulatory approvals. The company desires to aid in the recovery of patients with coronary artery disease by vascular repair function while decreasing long-term problems.
- Reva Medical: The leading promoter of bioresorbable vascular scaffolds, Reva Medical, has established its Fantom scaffold in more international markets. In 2024, the company formed a partnership with the material science department at the local university to research and develop a more substantial material and better resorption rate, thereby facilitating their stent technology.
- Arterial Remodeling Technologies (ART): ART has built a considerable base on bioresorbable stent platforms and, in 2024, grew considerably after obtaining significant funding. Their mission is to polish, i.e., improve stent effectiveness, thus leading to healthier arteries in the long term.
Key Market Players
- TERUMO CORPORATION
- Boston Scientific Corporation
- Medtronic
- Vascular Concepts
- B. Braun Melsungen AG
- BIOTRONIK SE & Co. KG
- Abbott Laboratories
- Biosensors International Group, Ltd.
- HEXACATH
- STENTYS SA
Market Definition
The coronary stents market represents revenue generated from implantable coronary artery stents and scaffolds used in percutaneous coronary intervention (PCI) procedures. The market measures the value of drug-eluting stents, bare-metal stents, and bioresorbable vascular scaffolds sold to hospitals, catheterization laboratories, and interventional cardiology centers.
Inclusions cover drug-eluting stents (polymer-coated and polymer-free), bare-metal stents, bioresorbable vascular scaffolds, and dual-therapy stents. It includes balloon-expandable and self-expanding delivery systems. Dedicated PCI guidewires and inflation devices bundled with stent systems are also included.
Exclusions include peripheral vascular stents, carotid stents, structural heart devices such as TAVR valves, and standalone PCI diagnostic equipment. Coronary artery bypass graft (CABG) surgical instruments and standalone angiography systems are outside the scope.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 37.5 Billion |
| Product Type | Drug-Eluting Stents, Bare-Metal Stents, Bioresorbable Vascular Scaffolds |
| Material Type | Cobalt-Chromium, Platinum-Chromium, Stainless Steel, PLLA Bioresorbable |
| End User | Hospitals, Catheterization Laboratories, Interventional Cardiology Centers |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Key Companies Profiled | Boston Scientific, Abbott, Medtronic, MicroPort, B. Braun, Terumo |
Segmentation
By Product:
Drug Eluting Stents (DES), Bare Metal Stents (BMS), Bioabsorbable Stents, Self-expanding Stents, Balloon-expandable Stents, etc.
By End User:
Hospitals, Cardiac Centers, Ambulatory Surgical Centers, and Other Healthcare Settings.
By Raw Material:
Cobalt-Chromium, Stainless Steel, Platinum-Chromium, Nickel-Titanium, Polymer
By Region:
North America, Latin America, Western Europe, South Asia and Pacific, East Asia, Middle East and Africa.
Bibliography
- MicroPort Scientific Corporation. (2025, September). Firesorb bioresorbable vascular scaffold launch and FUTURE study results. MicroPort Press Releases.
- Abbott Laboratories. (2024, May). XIENCE Sierra drug-eluting stent India launch. Abbott Newsroom.
- Boston Scientific Corporation. (2026, February). Fourth quarter and full year 2025 results. Boston Scientific Investor Relations.
- World Health Organization. (2024). Cardiovascular diseases fact sheet. WHO.
Frequently Asked Questions
What is the current global market size for Coronary Stents?
The global market is valued at USD 37.5 Billion in 2026, driven by the persistent global cardiovascular disease burden and advancing DES and BVS technologies.
What is the projected CAGR for the market over the next 10 years?
The market is projected to grow at a CAGR of 5.6% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
Asia Pacific leads due to rapidly growing PCI procedure volumes in India and China, followed by North America driven by premium DES reimbursement and BVS clinical adoption.
What are the primary market drivers?
The re-emergence of clinically validated bioresorbable scaffolds and the launch of ultra-low-profile DES platforms for complex lesions are the primary growth catalysts.
Who are the leading suppliers in the industry?
Boston Scientific, Abbott, Medtronic, and MicroPort are key players, differentiating through next-generation DES delivery systems and bioresorbable scaffold technology.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Drug-Eluting Stents (DES)
- Bare Metal Stent (BMS)
- Bioresorbable Vascular Stent (BVS)
- Drug-Eluting Stents (DES)
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Ambulatory Surgical Centers
- Cardiac Catheterization Laboratories
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By End Use
- Competition Analysis
- Competition Deep Dive
- TERUMO CORPORATION
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Boston Scientific Corporation
- Medtronic
- Vascular Concepts
- B. Braun Melsungen AG
- TERUMO CORPORATION
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End Use
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End Use
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End Use
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End Use
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

