The Healing Skin Care Products Market is projected to expand from USD 18.6 Billion in 2026 to USD 36.9 Billion by 2036, at a 7.1% CAGR. The decade is being shaped by dermatology-led scale strategies that turn skin repair into a repeat purchase platform across pharmacies, e-commerce, and clinic-adjacent channels. L’Oréal’s Dermatological Beauty reporting shows the division is operating through a professional network effect, anchored by healthcare professional partnerships and prescription influence that supports repair-positioned skincare conversion at scale. In the same disclosure, Myriam Cohen-Welgryn, President of Dermatological Beauty, states: “We lead skincare innovation, partnering with 310,000 healthcare professionals, to provide life-changing products and services for all.”
Portfolio renovation cadence is also visible in European dermo-skincare. Beiersdorf’s 2024 annual reporting highlights reinvestment in eczema and repair platforms, including the overhaul of Eucerin AtopiControl and performance claims tied to moisturisation duration, reinforcing that barrier repair is being treated as a structured franchise rather than seasonal demand. On the pure-play dermatology side, Galderma’s 2024 annual report positions Dermatological Skincare as a core growth engine with Cetaphil framed as a flagship sensitive-skin platform, underscoring category confidence in repair-led mass-premium scaling.
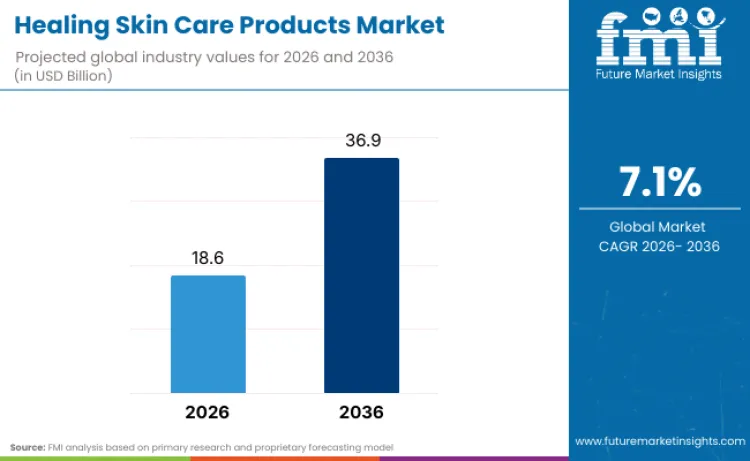
It is projected to be USD 18.6 Billion in 2026 and USD 36.9 Billion by 2036, expanding at a 7.1% CAGR.
FMI Research Approach: Modeled using repair-led skincare consumption, channel throughput, and compliance-driven portfolio refresh cycles.
Creams and ointments lead with a 34.7% share in 2026, supported by high trust in occlusive, barrier-first formats that scale cleanly through pharmacy and clinician-adjacent recommendation loops.
FMI Research Approach: Share mapped to format suitability for barrier repair routines and repeat purchase behaviour.
Wound healing leads with a 31.8% share in 2026, signalling demand concentration around everyday repair needs where OTC and dermo-cosmetic positioning can convert without prescription access barriers.
FMI Research Approach: Application mix estimated using consumer self-care routines and pharmacy-led conversion patterns.
Pharmacies and drug stores lead with a 36.5% share in 2026, reflecting trust-led purchasing and pharmacist recommendation pathways for repair and sensitive-skin regimens.
FMI Research Approach: Channel split sized using healthcare-adjacent retail throughput and replenishment cadence.
| Items | Values |
|---|---|
| Market Size (2026) | USD 18.6 Billion |
| Forecast Value (2036) | USD 36.9 Billion |
| CAGR (2026 to 2036) | 7.1% |
Growth is being driven by the industrialisation of dermatology-partnered selling models and the tightening of cosmetics governance, which increases the premium on compliant, clinically anchored repair claims. China is reinforcing compliance-led category expansion through cosmetics registration and filing rules designed to regulate product safety and quality, which advantages scaled brands that can sustain ingredient documentation and safety substantiation for repair claims. In the United States, FDA guidance on cosmetic facility registration and product listing under MoCRA clarifies submission responsibilities, required information, and timing, raising documentation discipline and favouring incumbents with mature regulatory and quality systems. These mechanisms collectively expand shelf access and consumer trust for healing and repair skincare, while pushing competitive advantage toward operators that can run fast renovation cycles without compliance disruption.
The market for Healing Skin Care Products is segmented into Product Type (creams and ointments, lotions, gels, serums, and cleansers), Ingredient Type (botanical extracts, vitamins and minerals, peptides and proteins, natural oils and butters, and synthetic actives positioned within cosmetics or OTC-permitted scope), Application (wound healing, burn care, acne and scar repair, eczema and dermatitis support, and anti-inflammatory skin repair), and Distribution Channel (pharmacies and drug stores, dermatology clinic retail counters, online retail, supermarkets and hypermarkets, and specialty stores). This segmentation reflects how consumers adopt repair routines through familiar topical formats, how healing and barrier-restoration claims are structured under tightening cosmetics governance, how demand concentrates around recurring compromised-skin use cases, and how trust-led channels convert higher-intent purchases in dermo-cosmetic and pharmacy-adjacent settings.
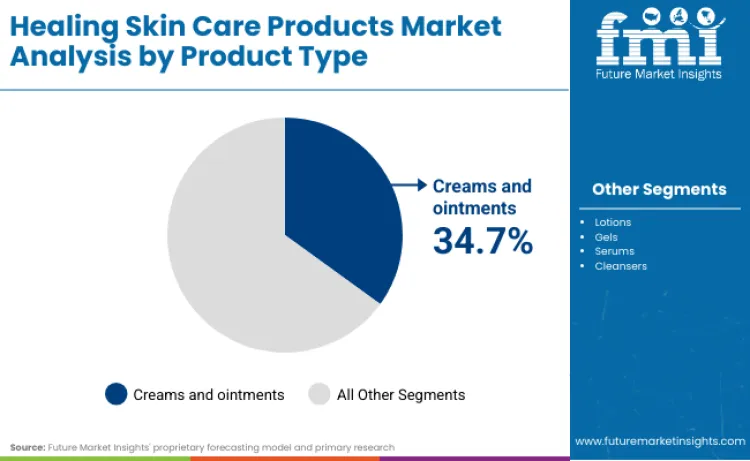
Creams and ointments lead with a 34.7% share in 2026 because the category’s core purchase driver is barrier restoration that consumers can feel quickly and repurchase predictably, and these formats deliver that outcome with minimal behavioural change. Large operators are scaling repair-led skincare through clinically anchored brand systems rather than one-off hero launches. Beiersdorf’s 2024 reporting shows the same structural logic in Europe, highlighting the overhaul of Eucerin AtopiControl for atopic dermatitis and performance framing around moisturisation duration, which supports repeat purchase for repair needs that are chronic rather than occasional. These repeated portfolio actions make creams and ointments the most scalable format inside healing skincare because they align with pharmacy recommendation behaviour, preserve claims discipline under cosmetics rules, and translate across climates and skin conditions with low adoption friction.
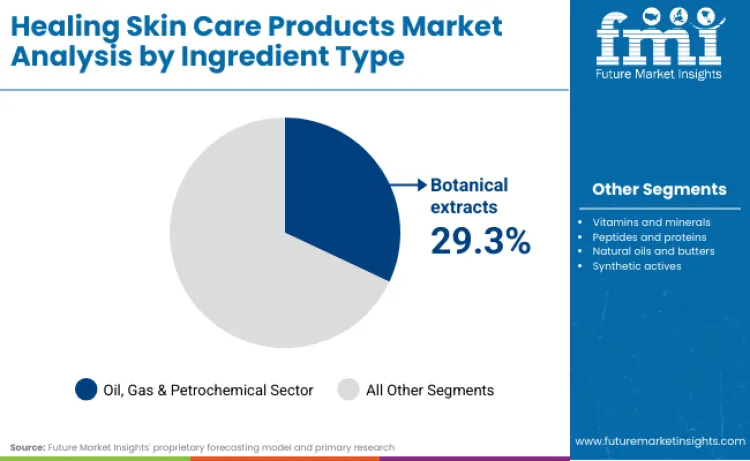
Botanical extracts lead with a 29.3% share in 2026 because they enable repair narratives that scale across markets without forcing drug-like claims, and they remain easier to document and globalise within cosmetics compliance boundaries than higher-risk actives. China’s Provisions for Registration and Filing of Cosmetics are explicitly designed to regulate registration and filing while ensuring quality and safety, and they require registrants and filing persons to select compliant ingredients and clarify ingredient source and safety information through official information platforms. This raises the value of ingredient systems that can be substantiated consistently across SKUs, which structurally benefits botanical platforms inside repair skincare. The same dynamic appears in the United States where FDA guidance on registration and listing clarifies who must submit and what information must be included, increasing operating friction for smaller brands and advantaging scaled portfolios with established documentation systems. In practice, botanical extracts become the most commercially resilient ingredient lane for healing skincare because they support sensitive-skin positioning, tolerate reformulation cycles, and can be integrated across creams, lotions, and cleansers without elevating regulatory exposure.
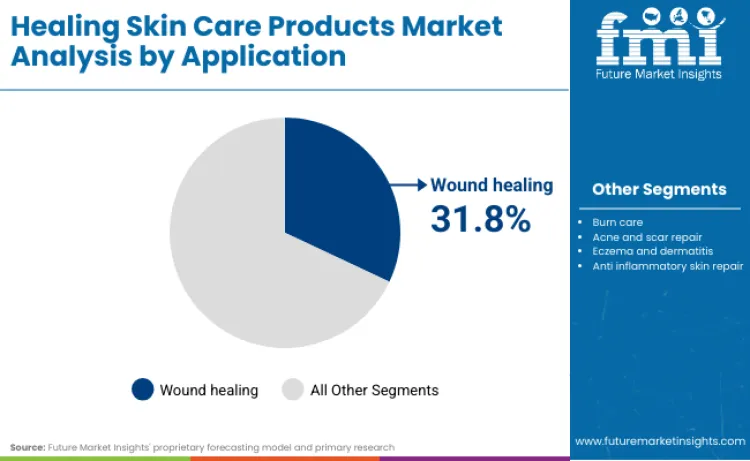
Wound healing leads with a 31.8% share in 2026 because the most frequent repair need in household skincare is practical, everyday restoration of compromised skin, which converts through self-care routines and pharmacy trust without requiring specialist access. The category behaves as a high-intent purchase where consumers seek risk reduction and predictable outcomes, and this concentrates demand in wound and repair use cases that can be positioned inside compliant cosmetics or OTC-permitted frameworks. Compliance systems are also strengthening the market’s bias toward trusted repair claims. FDA’s MoCRA-linked guidance clarifies the statutory requirement to submit facility registrations and product listings for cosmetics, which increases accountability signals for brands selling repair-positioned skincare at scale. In Europe, Beiersdorf’s reporting on Eucerin AtopiControl renovation demonstrates how major portfolios treat compromised-skin needs as a structured franchise, reinforcing adjacent demand for wound and repair routines that consumers interpret as protection and recovery.
A key trend is the conversion of dermatological skincare into a scaled repair platform via professional networks and flagship sensitive-skin brands. Galderma’s 2024 annual report positions Dermatological Skincare as a foundational business and frames Cetaphil as a flagship sensitive-skin brand, reinforcing that pure-play dermatology portfolios are scaling repair and sensitivity claims through mass-premium expansion. The operational implication is that brands are investing in medical detailing, pharmacy relationships, and regimen logic to defend pricing and sustain replenishment.
A key restraint is rising compliance and product substantiation load, which increases time-to-market friction and raises operating costs for smaller brands attempting to scale healing claims across borders. FDA’s guidance clarifies who must submit cosmetic facility registrations and product listings, what information must be included, how submissions are made, and when they are required, tightening documentation discipline for products sold in the United States. In China, the cosmetics registration and filing provisions require ingredient safety responsibility and disclosure of ingredient source and safety information through official systems, increasing the governance burden for brands with complex supply chains. The effect is a competitive tilt toward incumbents with established regulatory systems and quality infrastructure.
Future Market Insights identifies China (CAGR 9.6%), India (8.9%), Germany (8.2%), Brazil (7.5%), the United States (6.7%), the United Kingdom (6.0%), and Japan (5.3%) as the principal country-level growth engines shaping the global Healing Skin Care Products market through 2036. China leads expansion as tighter cosmetics registration, filing, and in-market surveillance frameworks reward compliant, clinically substantiated repair portfolios that can scale through pharmacy and e-commerce channels. India’s growth is driven by regulatory formalisation under Cosmetics Rules and rising trust in organised pharmacy-led dermo-cosmetic brands addressing wound care, eczema, and sensitive skin needs. The United States benefits from MoCRA-driven governance that strengthens documentation discipline and favours incumbents capable of sustaining high-volume repair assortments across pharmacies and online retail. Germany’s demand reflects a mature but expanding dermo-skincare culture supported by EU-wide cosmetics compliance and strong pharmacy credibility. Brazil’s growth is shaped by ANVISA-led electronic regularisation that concentrates demand toward compliant branded portfolios. The UK shows steady expansion under structured product notification requirements, while Japan’s slower growth reflects disciplined cosmetics standards and mature consumption patterns that prioritise renovation cadence over aggressive claim escalation. FMI emphasizes that across regions, healing skincare demand is anchored in compliance-led trust, pharmacy conversion, and repeat repair routines rather than discretionary beauty cycles.
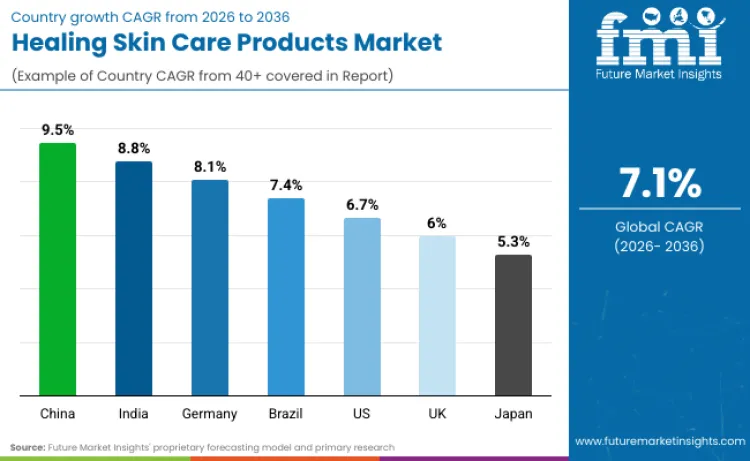
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 9.6% |
| India | 8.9% |
| Germany | 8.2% |
| Brazil | 7.5% |
| USA | 6.7% |
| UK | 6.0% |
| Japan | 5.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
China grows at a 9.6% CAGR because the market is being formalised through cosmetics registration and filing rules that tighten ingredient accountability and strengthen in-market control, which favours scaled brands able to sustain documentation discipline for repair claims. The Provisions for Registration and Filing of Cosmetics are framed to regulate registration and filing while ensuring quality and safety, and they assign responsibility to registrants and filing persons for ingredient safety, including clarifying ingredient source and safety information via official platforms. For healing skincare, this acts as a market-access filter: brands with mature regulatory workflows can expand repair assortments faster across channels, while weaker operators face higher friction when updating formulations, claims, or suppliers. The outcome is accelerated growth led by compliant portfolios that can keep pharmacy and e-commerce listings stable while executing rapid renovation cycles.
India expands at a 8.9% CAGR because the market is moving toward structured compliance and organised retail execution, which benefits established brands in repair-led skincare. CDSCO states that no cosmetic shall be imported into India unless the product is registered under the rules by the Central Licensing Authority, and that cosmetics are required to be registered along with pack size, variants, and manufacturing premises before import. This mechanism favours scaled operators with documentation capability and supports wider availability of compliant healing skincare across pharmacies and modern trade. It also reduces fragmentation that can dilute consumer trust in sensitive-skin and repair claims, enabling higher repeat purchase in barrier and wound-repair adjacent routines. The result is growth driven by formalisation and distribution deepening rather than short-lived trend adoption.
Germany grows at a 8.2% CAGR because demand is shaped by a regulated cosmetics environment that rewards brands with disciplined safety substantiation and continuous portfolio renovation in clinically adjacent skincare. EU Regulation (EC) No 1223/2009 harmonises cosmetics rules to support an internal market while ensuring a high level of protection of human health, reinforcing the value of robust compliance systems for repair-positioned products. In parallel, Beiersdorf’s 2024 reporting highlights ongoing innovation and renovation within its dermatological skincare portfolio, including Eucerin AtopiControl updates for eczema-prone skin, which supports predictable replenishment in repair routines and keeps pharmacy credibility high. These forces anchor Germany’s growth in regulated trust, pharmacy conversion, and consistent product renovation rather than discretionary beauty cycles.
Brazil advances at a 7.5% CAGR because ANVISA’s regularisation framework formalises how personal care products, cosmetics and perfumes are placed and maintained in market, strengthening compliance discipline and shifting advantage to scaled operators. For healing skincare, this supports growth by improving product reliability and channel confidence, particularly in supermarkets and pharmacies that prefer compliant, traceable portfolios. It also raises friction for smaller brands that struggle with documentation and timely updates, which concentrates category scaling into incumbents that can industrialise submissions and reformulation cycles.
The United States grows at a 6.7% CAGR because MoCRA-linked compliance is raising documentation expectations for cosmetics facilities and products, favouring incumbents that can sustain high-volume repair portfolios across pharmacies, mass retail, and e-commerce. FDA guidance states it provides recommendations and instructions for submitting cosmetic facility registrations and product listings, including statutory requirements, who is responsible, what information to include, how to submit, and when to submit. This increases the value of mature quality systems and reduces disruption risk for high-velocity repair skincare, sustaining category expansion through trust and availability rather than speculative claim escalation.
The UK expands at a 6.0% CAGR because market access for cosmetics in Great Britain requires notification through the Submit cosmetic product notifications service before products can be placed on the GB market, reinforcing responsible person accountability and structured compliance. For healing skincare, this strengthens channel confidence in established brands that can manage notifications, labelling responsibility, and SKU updates without disruption, supporting stable growth through pharmacies and specialty retail. The market’s high online penetration supports replenishment, but compliance-led discipline keeps growth anchored in reliable portfolio execution.
Japan grows at a 5.3% CAGR because the market operates under defined cosmetics standards that constrain ingredient selection and keep category expansion disciplined. The Ministry of Health, Labour and Welfare’s Standards for Cosmetic Products set requirements that cosmetics shall not contain medical drug ingredients except specified cases, reinforcing a clear boundary that shapes how healing and repair claims are positioned. This structure supports trust and quality but limits aggressive claim escalation, so growth depends on renovation cadence and brand equity more than rapid category disruption. In Japan, global leadership does not automatically translate into category leadership because domestic operators with established distribution and compliance infrastructure retain structural advantage.
Competition is led by scaled beauty and consumer health operators that can run compliant claim architectures, sustain pharmacy credibility, and refresh repair portfolios at high cadence. Scope includes creams and ointments, lotions, gels, serums, and cleansers positioned for healing and skin repair across wound healing, burn care, acne and scar repair, eczema and dermatitis support, and anti-inflammatory skin repair sold as finished consumer products. Scope excludes prescription dermatology drugs, medical devices and advanced wound dressings, and in-clinic procedures. Asia’s competitive structure is governance-led, with China’s cosmetics registration and filing provisions reinforcing ingredient accountability and in-market control, favouring scaled brands that can maintain documentation and stable supply. Japan remains structurally distinct due to cosmetics standards that constrain medical-drug ingredient inclusion, supporting incumbents with local compliance and distribution strength rather than automatic translation of global leadership.
The Healing Skin Care Products Market covers finished topical consumer products positioned for skin repair, recovery, and barrier support across compromised-skin use cases. It includes creams and ointments, lotions, gels, serums, and cleansers marketed for wound healing support, burn care, acne and scar repair, eczema and dermatitis support, and anti-inflammatory skin repair when sold as cosmetic or OTC-permitted consumer skincare. It focuses on products purchased for repeat repair routines through retail, pharmacy, and clinic-adjacent channels. It excludes prescription dermatology treatments, advanced wound dressings regulated as medical devices, and in-clinic procedures.
Included revenues cover branded healing skincare sold through pharmacies and drug stores, dermatology clinics retail counters, online retail, supermarkets and hypermarkets, and specialty stores. Included also covers dermo-cosmetic franchises and dermatological skincare brands scaled through healthcare professional partnerships, where the end product is a packaged consumer skincare item. L’Oréal’s Dermatological Beauty platform and its disclosed scale of healthcare professional partnerships is included as a structural signal of category expansion through clinical adjacency. Included ingredient systems span botanicals, vitamins and minerals, peptides and proteins, natural oils and butters, and synthetic actives, where positioned within cosmetics rules and documented through compliant supply chains.
Excluded revenues cover prescription medicines for dermatological conditions, products whose primary regulatory classification is a drug, advanced wound care dressings and devices, and hospital-use consumables not sold as consumer skincare. Excluded also covers supplements, ingestible beauty products, and aesthetic injectables. Bulk ingredient sales are excluded unless attributable to finished consumer product sell-through. Advisory and regulatory services are excluded. In markets where cosmetics registration and filing rules place ingredient accountability on registrants and filing persons, only the finished compliant consumer products are in scope, not the regulatory process itself.
| Items | Values |
|---|---|
| Quantitative Units | USD 18.6 Billion |
| Product Type Segments | Creams & Ointments; Lotions; Gels; Serums; Cleansers |
| Ingredient Type Segments | Botanical Extracts; Vitamins & Minerals; Peptides & Proteins; Natural Oils & Butters; Synthetic Actives |
| Application Categories | Wound Healing; Burn Care; Acne & Scar Repair; Eczema & Dermatitis; Anti-Inflammatory Skin Repair |
| Distribution Channels | Pharmacies & Drug Stores; Online Retail; Supermarkets & Hypermarkets; Dermatology Clinics; Specialty Stores |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia & Pacific, East Asia, Middle East & Africa |
| Key Countries | China, India, Germany, United States, Brazil, United Kingdom, Japan |
| Key Companies Profiled | L’Oréal; Johnson & Johnson; Beiersdorf; Procter & Gamble; Unilever; Galderma; Kao Corporation; Himalaya Wellness; Kenvue; Bayer |
| Additional Attributes | Dollar sales measured for healing skincare across product type, ingredient platform, application focus and channel performance; pharmacy-led conversion and clinician-adjacent recommendation dynamics; compliance and substantiation impact under U.S. MoCRA registration and listing, China cosmetics filing and ingredient accountability frameworks, UK product notification systems, and country-level regularisation models; premiumisation and dermo-cosmetic portfolio scaling effects; renovation cadence of repair franchises by leading companies; channel mix shifts between pharmacy, e-commerce and clinic retail environments |
What is the global market size of Healing Skin Care Products in 2026 and 2036?
The healing skin care products market is valued at USD 18.6 billion in 2026 and is projected to reach USD 36.9 billion by 2036.
What is the projected CAGR for the Healing Skin Care Products Market through 2036? The market is expected to grow at a 7.1% CAGR from 2026 to 2036.
The market is expected to grow at a 7.1% CAGR from 2026 to 2036.
Which product type dominates the Healing Skin Care Products Market in 2026?
Creams and ointments lead with a 34.7% share due to their barrier-repair effectiveness and strong pharmacy-led repeat purchase behavior.
Why do pharmacies and drug stores remain the leading distribution channel in 2026?
Pharmacies dominate with a 36.5% share because healing skincare purchases rely on trust, clinical adjacency, and pharmacist recommendation pathways.
What is the primary factor driving faster growth in China compared to mature markets?
China leads growth with a 9.6% CAGR due to compliance-led formalisation that favours scalable, well-documented repair skincare portfolios.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.