Insulin resistance improvement drug market is anticipated to reach USD 10.3 billion in 2026, driven by an escalating global burden of metabolic disorders. With a CAGR of 8.4% extending through 2036, total industry revenue is poised to surpass USD 23.1 billion by the end of this forecast duration. Chronic lifestyle conditions and rising obesity rates act as primary accelerators for this financial trajectory. Pharmaceutical entities are channeling substantial funds into novel therapeutics, aiming to mitigate long-term complications associated with uncontrolled glucose levels.
A shift from palliative care toward curative mechanisms is redefining investment strategies across major economic zones. Patients are increasingly seeking effective management solutions, pushing manufacturers to innovate beyond traditional therapies. This upward trend reflects a broader necessity for advanced pharmacological interventions in managing metabolic health effectively over the coming decade.
Rising awareness regarding prediabetes management significantly influences industry expansion. Healthcare providers are emphasizing early intervention, utilizing various therapeutic classes to enhance insulin sensitivity before full-blown diabetes onset. Clinical trials exploring dual agonists and novel molecules are yielding promising results, fostering confidence among investors and medical practitioners.
Adoption of sedentary lifestyles continues to fuel incidence rates of metabolic syndrome, creating sustained demand for efficacious medical treatments. Government initiatives aimed at curbing chronic disease burdens are facilitating faster approval processes for next-generation drugs. Consequently, market participants are witnessing robust opportunities in developing regions where healthcare infrastructure is rapidly modernizing to address these critical public health challenges.
Innovations in drug delivery systems are transforming patient compliance and therapeutic outcomes. Researchers are integrating insulin management system technologies with pharmaceutical regimens to offer holistic care. Development of non-injectable insulin options is gaining traction, addressing needle phobia barriers that often hinder adherence. Focus is also shifting towards cardiometabolic drugs that offer dual benefits of glycemic control and cardiovascular protection. Advancements in biotechnology allow for precise targeting of metabolic pathways, enhancing efficacy of anti-hyperglycemic agents. Such technological strides are pivotal in shaping future treatment landscapes, ensuring that diverse patient needs are met with precision and safety.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 10.3 Billion |
| Industry Value (2036) | USD 23.1 Billion |
| CAGR (2026 to 2036) | 8.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Emergence of multi-functional peptides is serving as a significant catalyst for broader adoption of metabolic therapies. Recent breakthroughs in glp-1 receptor agonist formulations have revolutionized treatment protocols by offering weight loss benefits alongside glucose regulation. This dual-action approach appeals strongly to healthcare providers managing patients with obesity diabetes drugs requirements. Additionally, clinical success of sglt2 inhibitors in reducing heart failure risks has expanded prescribing potential beyond simple glucose management.
Continuous education programs regarding long-term risks of untreated insulin resistance are also driving patient proactivity. Pharmaceutical companies are capitalizing on these trends by launching combination therapies that simplify dosing regimens, thereby improving medication adherence rates. Such multifaceted drivers ensure that demand for advanced resistance-improving drugs remains on a steep upward curve.
The insulin resistance improvement drug market is segmented across several key categories based on various criteria, including drug class, application, distribution channel, and geography. Drug class segmentation typically includes categories such as biguanides, thiazolidinediones, GLP-1 receptor agonists, and SGLT2 inhibitors, each with unique mechanisms of action for managing insulin resistance. Application segmentation may focus on specific conditions associated with insulin resistance, such as Type 2 diabetes and polycystic ovary syndrome (PCOS). Distribution channels can range from hospital pharmacies and retail pharmacies to online platforms, reflecting shifts in consumer purchasing behavior.
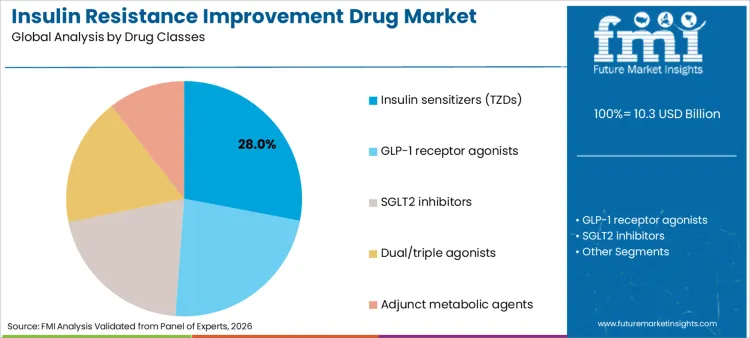
Insulin sensitizers (TZDs) account for 28% of share in 2026, driven by their direct mechanism of action on PPAR-gamma receptors to improve insulin sensitivity. Dominance is underpinned by clinical guidelines that continue to recommend TZDs as effective add-on therapies for patients uncontrolled on metformin. This segment benefits significantly from cost advantages compared to newer biologics, making them accessible in cost-sensitive regions. Ongoing research into reducing side effects associated with this class is renewing interest among prescribers. Furthermore, their role in managing hyperglycemia treatment protocols remains vital for specific patient phenotypes. As healthcare systems strive for cost-efficient disease management, demand for proven, affordable sensitizers is expected to remain robust, sustaining their leadership position throughout the forecast period.
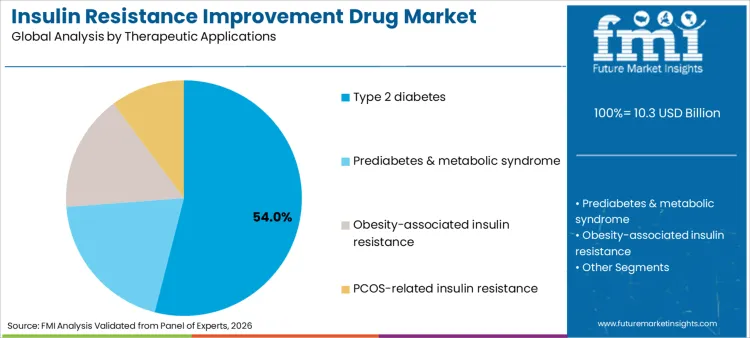
Type 2 diabetes applications command a 54% share of the sector in 2026, reflecting the massive global prevalence of this chronic condition. Dominance is inextricably linked to the pathophysiology of the disease, where insulin resistance is a core defect requiring targeted pharmacological intervention. Sector relies heavily on alpha-glucosidase inhibitors and other agents to manage postprandial spikes effectively. These therapies are essential for preventing microvascular and macrovascular complications, which drive healthcare costs. Consequently, relentless drive to manage diabetes progression ensures that drugs designed to ameliorate resistance remain highest priority for funding and procurement in both public health programs and private insurance formularies.
Biosimilars are set to fundamentally reshape sales landscapes by introducing cost-competitive alternatives to branded biologics. As patents for blockbuster drugs expire, entry of insulin biosimilars is creating price erosion, forcing originators to innovate or lower costs. This shift towards affordable biologics is democratizing access to high-quality treatments in emerging economies. Manufacturers who fail to adapt their pricing strategies risk losing share to agile competitors offering bioequivalent products. Furthermore, availability of lower-cost options drives consumption volume, as healthcare systems can treat more patients within fixed budgets. Rise of biosimilars also stimulates innovation in insulin delivery pens, as companies seek to differentiate their commodity products through superior device engineering and user-friendly features.
Regulatory bodies globally are increasingly advocating for cardiovascular outcome trials (CVOTs) for all new metabolic drugs, directly impacting development timelines and costs. This regulatory shift forces pharmaceutical companies to generate robust safety data, increasing barriers to entry for smaller players. However, it also introduces opportunities for differentiation; agents that demonstrate superior cardiovascular safety gain rapid formulary inclusion. Strict pharmacovigilance standards are impacting production of sglt2 inhibitors treatment options. Manufacturers must now ensure rigorous post-marketing surveillance to monitor rare adverse events. While these regulations drive development of safer drugs, they also impose higher compliance costs. Companies that proactively align with FDA and EMA guidelines regarding comprehensive safety profiles will gain a competitive edge, positioning their products as gold standards.
Europe is projected to demonstrate significant expansion from 2026 to 2036, while emerging economies like India and China are demonstrating even faster acceleration. Regional evolution is characterized by distinct growth drivers: established nations in the West are pivoting towards novel biologics and weight-management indications, whereas Asian nations are expanding access to generic sensitizers and improving diagnosis rates. Disparity in growth rates highlights a shifting center of gravity, where huge diabetic populations in Asia-Pacific are creating massive volume demand. Conversely, North American and European sectors are focusing on innovation and specialized applications, such as fatty liver treatment overlaps. Government grants and private funding in these major nations are heavily slanted towards combating metabolic syndrome, ensuring sustained capital influx.
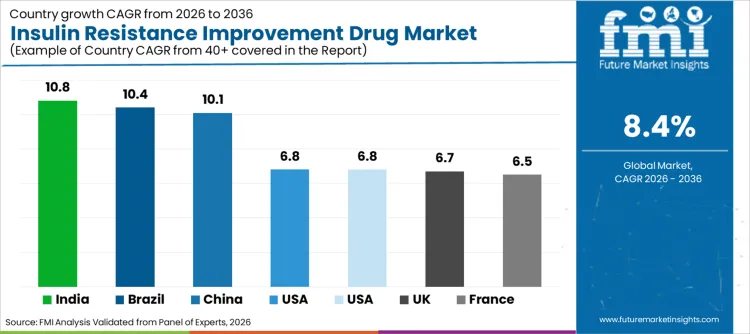
| Region | CAGR (2026 to 2036) |
|---|---|
| USA | 6.8% |
| Brazil | 10.4% |
| China | 10.1% |
| India | 10.8% |
| Germany | 6.8% |
| France | 6.5% |
| UK | 6.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Sales of insulin resistance improvement drugs in the USA are set to grow at a CAGR of 6.8% through 2036. This expansion is underpinned by high obesity rates and a healthcare system that rapidly adopts premium therapeutic innovations. Growth is fueled by substantial insurance coverage for novel drug classes, which translates into high revenue generation per prescription. Furthermore, rapid adoption of ozempic and similar agents for weight management has expanded the addressable patient pool significantly. Presence of key industry players headquartered within the country also streamlines supply chains, ensuring rapid access to latest launches. Emphasis on preventive cardiology within diabetes care continues to be a primary driver, maintaining USA position as a critical revenue generator.
Demand for insulin sensitizers in China is projected to rise at a 10.1% CAGR during the assessment period. "Healthy China 2030" initiatives emphasize management of chronic diseases, fostering a favorable environment for domestic and international pharmaceutical manufacturers. Increasing investments in local biotechnology firms are creating massive supply of cost-effective antidiabetics to support vast patient populations. Rising prevalence of lifestyle-related conditions in urban centers is prompting local hospitals to adopt advanced pharmacological protocols. This shift is further supported by government efforts to negotiate lower drug prices, thereby accelerating uptake of essential medicines across provinces and fueling sector expansion.
Insulin resistance improvement drug industry in India is poised to expand at a CAGR of 10.8%, representing highest growth rate among analyzed nations. Surge is largely attributed to country's status as "Diabetes Capital of the World," necessitating urgent and widespread access to affordable medications. Government's focus on generic drug initiatives is providing essential medications to rural demographics, upgrading care from basic dietary management to pharmacological intervention. Furthermore, increasing number of partnerships between Indian pharma giants and global innovators necessitates production of high-quality formulations. Cost advantage of manufacturing in India, combined with improving healthcare access schemes, is attracting significant investment into the domestic metabolic health ecosystem.
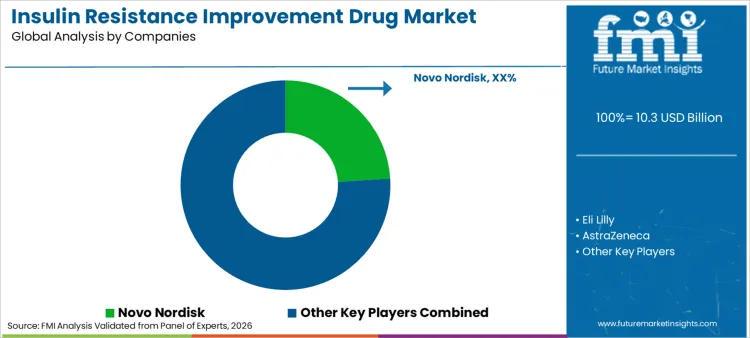
Competition in this industry is intensifying as major players leverage mergers and acquisitions to consolidate their portfolios and expand geographic footprints. Leading companies like Novo Nordisk and Eli Lilly are actively developing multi-agonist peptides to integrate weight loss efficacy into diabetes care.
Strategies are increasingly focused on bundling therapeutics with digital health solutions to offer comprehensive disease management platforms. "Beyond the pill" business models ensure long-term patient engagement and retention. Additionally, strategic partnerships with academic centers allow companies to co-develop molecules that address specific metabolic pathways. Competitive landscape is also witnessing a rise in direct-to-consumer marketing, where manufacturers are differentiating themselves by offering patient support programs and copay assistance to appeal to cost-conscious consumers.
| Items | Values |
|---|---|
| Quantitative Units | USD Million, USD Billion |
| Drug Classes | Insulin sensitizers (TZDs), GLP-1 receptor agonists, SGLT2 inhibitors, Dual/triple agonists, Adjunct metabolic agents |
| Therapeutic Applications | Type 2 diabetes, Prediabetes & metabolic syndrome, Obesity-associated insulin resistance, PCOS-related insulin resistance |
| Distribution Channels | Hospital pharmacies, Retail pharmacies, Online pharmacies, Specialty clinics |
| Regions | North America, Europe, East Asia, South Asia, Latin America, MEA |
How large is the demand for Insulin Resistance Improvement Drugs in the global market in 2026?
The demand for Insulin Resistance Improvement Drugs in the global market is estimated to be valued at USD 10.3 billion in 2026.
What will be the market size of Insulin Resistance Improvement Drugs in the global market by 2036?
The market size for Insulin Resistance Improvement Drugs in the global market is projected to reach USD 23.1 billion by 2036.
What is the expected demand growth for Insulin Resistance Improvement Drugs in the global market between 2026 and 2036?
The demand for Insulin Resistance Improvement Drugs in the global market is expected to grow at a CAGR of 8.4% between 2026 and 2036.
Which drug class in the Insulin Resistance Improvement Drug market is poised to lead global sales by 2026?
Insulin sensitizers (TZDs) are expected to be the dominant drug class, capturing approximately 28% of the global market share in 2026 due to widespread clinical use.
How significant is the role of Retail Pharmacies in driving Insulin Resistance Improvement Drug sales in 2026?
Retail pharmacies represent a critical distribution channel, projected to hold a substantial 38% share of the total market in 2026 as patient accessibility remains a key priority.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.