Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market
The point-of-care diagnostic analyzer accuracy and interference test equipment market is Segmented By Test Function (Accuracy Verification, Interference Testing, Precision Testing, Linearity Testing, Method Comparison), Analyzer Type (Glucose Analyzers, Blood Gas Analyzers, Electrolyte Analyzers, Coagulation Analyzers, Cardiac Analyzers), Equipment Format (Benchtop Systems, Portable Simulators, QC Stations, Comparator Readers, Data Platforms), End User (OEM Manufacturers, Hospital POCT Teams, Reference Labs, Validation Labs, Academic Centers), Sample Matrix (Whole Blood, Capillary Blood, Plasma, Serum, Control Matrix), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Size, Market Forecast and Outlook By FMI
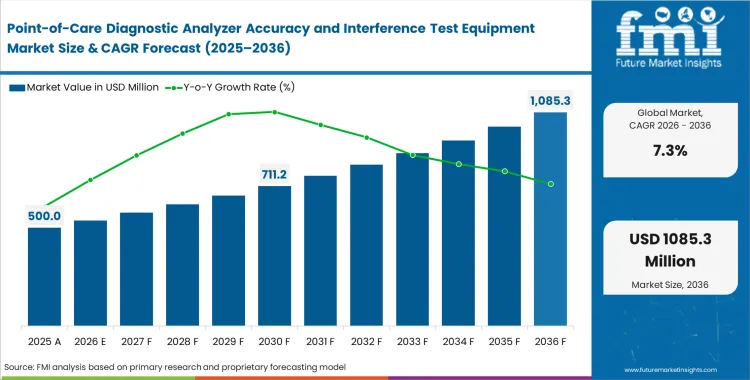
The point-of-care diagnostic analyzer accuracy and interference test equipment market was valued at USD 470 million in 2025. Sales are expected to surpass USD 500 million in 2026 at a CAGR of 7.3% during the forecast period. Industry expansion lifts total revenue valuation to USD 1,010 million through 2036 as clinical laboratories tighten validation protocols for decentralized diagnostics verification equipment.
Summary of Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market
- The market is forecast to reach USD 1,010 million by 2036.
- The market is expected to grow at a CAGR of 7.3% from 2026 to 2036.
- The market was estimated at USD 470 million in 2025.
- The forecast period represents an incremental opportunity of USD 510 million.
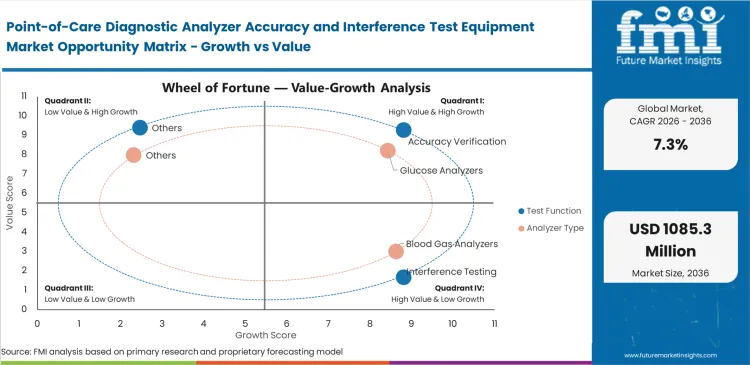
- Accuracy verification leads the test function segment with 38.0% share, driven by mandatory bias confirmation in decentralized diagnostics.
- Glucose analyzers dominate the analyzer category with 29.0% share, supported by widespread bedside usage and strict accuracy standards.
- Benchtop systems hold 31.0% share in the equipment format segment due to controlled validation requirements.
- OEM manufacturers account for 41.0% share in end use, reflecting strong involvement in premarket validation and compliance.
- Whole blood leads the sample type segment with 46.0% share, as it closely reflects real clinical testing conditions.
- Key companies in the market include Bio-Rad Laboratories, Abbott, Siemens Healthineers, Radiometer, Nova Biomedical, EKF Diagnostics, and Werfen.
Quality teams across decentralized hospital networks must prove bedside devices match central laboratory performance. Factory calibration alone no longer satisfies oversight for critical diagnostic use. Hospitals now need stricter analyzer accuracy testing to document consistency between portable systems and core lab instruments. Delays increase compliance exposure and raise the risk of treatment decisions based on inaccurate readings. This is pushing administrators toward specialized point of care diagnostics validation equipment rather than isolated device checks. Many sourcing teams still underestimate lifecycle costs, since consumables, staff training, audit preparation, and repeat verification often outweigh initial hardware spending. Buying patterns are shifting toward broader point of care diagnostics platforms that can support recurring validation across multiple sites. Facilities also need clinical diagnostics verification panels at regular intervals to maintain readiness and meet baseline CLIA verification requirements.
Digital integration between bedside analyzers and central laboratory servers removes a major validation bottleneck. Direct data transfer improves reconciliation, reduces manual transcription mistakes, and lowers labor intensity during routine quality checks. Hospitals that build automated comparator networks can reuse baseline performance data when expanding installed fleets, reducing repeated validation work. Wider hematology diagnostics deployment depends on this electronic verification loop because laboratories need a dependable way to track analyzer bias and precision across decentralized testing locations.
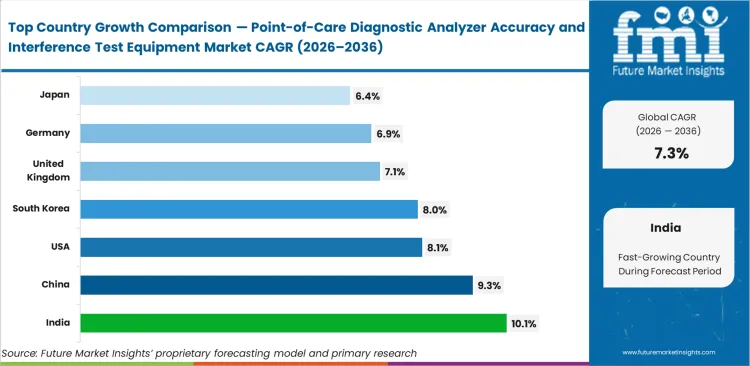
Global divergence separating these geographies depends strictly on whether localized validation guidelines require continuous daily recalibration or accept batch-based weekly verification, resulting with United States expected to project an expansion at 8.1% based on stringent regulatory enforcement of bedside testing accuracy. India predicted to lead global growth at 10.1% as private healthcare networks aggressively expand decentralized diagnostic capabilities across secondary tier cities. China likely to follow closely at an estimated 9.3% driven by aggressive national infrastructure investments targeting domestic manufacturing independence for point-of-care analyzer test equipment. South Korea is projected to register a CAGR of 8.0% through 2036. United Kingdom demand is set to rise at 7.1% CAGR through 2036. Germany is expected to grow at a CAGR of 6.9% during the study period. Japan is likely to post a CAGR of 6.4% by 2036.
Segmental Analysis
Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Analysis by Test Function
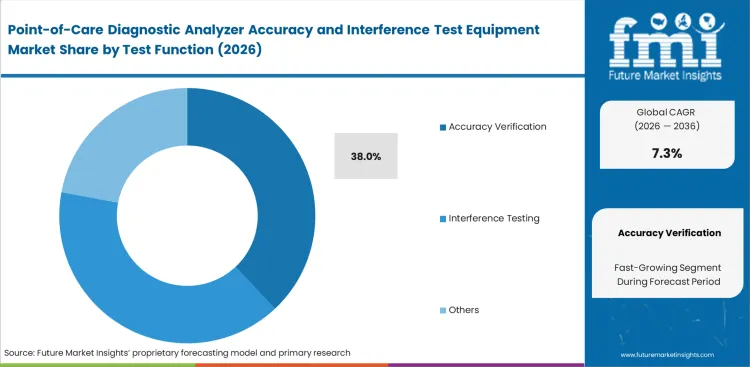
Universal regulatory mandates force hospital POCT teams to execute continuous baseline concordance checks across their clinical settings. Selecting complete verification systems provides immediate operational continuity for facilities running dozens of bedside units. Driven by the necessity to maintain daily compliance, the accuracy verification segment is estimated to account for 38.0% share in 2026 as facilities prioritize these core benchmarking protocols. Validating newly deployed devices against established blood gas analyzers keeps patient data flowing cleanly into central electronic health records. Laboratory directors often discover that frequent accuracy testing drains far more staff hours than the actual diagnostic procedures themselves, pushing them to invest heavily in automated tools to survive mandatory audits.
- Undetected Analytical Drift: Routine accuracy verification prevents silent calibration failures in decentralized units. Hospital POCT coordinators avoid disastrous diagnostic errors affecting acute patient interventions.
- Matrix Compatibility Variance: Residual risk remains concerning complex biological interferences even when baseline accuracy appears acceptable. Quality control specialists must execute separate targeted evaluations using specialized control solutions.
- Comprehensive Workflow Integration: Capturing full benefit of verification data requires automated transmission to central lab servers. IT integration officers must eliminate manual transcription steps to ensure absolute compliance traceability.
Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Analysis by Analyzer Type
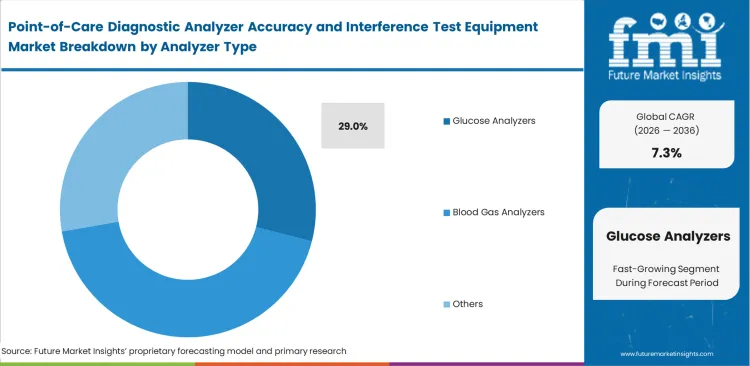
To manage massive equipment footprints, glucose analyzers are expected to hold 29.0% share in 2026 as purchasing directors prioritize validation solutions for high-volume bedside procedures. This extensive deployment forces nursing supervisors to manage thousands of individual verification events every single week. Implementing streamlined fully automatic blood gas analyzer protocols alongside routine glucose testing simplifies unit management considerably. Facilities that adopt unified blood glucose monitoring devices verification networks visibly reduce delays in critical care interventions. Experienced practitioners know that standard capillary sample protocols frequently mask severe systemic hematocrit interferences in critically ill patients, requiring specialized intensive-care verification matrices to prevent hidden hypoglycemic events.
- Central Laboratory Implementation: Core lab directors adopt unified glucose verification standards first to establish facility-wide baselines. This step creates a metrological anchor for all subsequent decentralized deployments.
- Intensive Care Unit Transition: Critical care nursing supervisors follow by demanding specialized interference validation protocols. Managing complex patient physiology requires absolute confidence in bedside glucose measurements.
- Ambulatory Clinic Integration: Outpatient facility managers reach full protocol adoption last. Strict regulatory enforcement eventually forces remote clinics to match acute-care validation standards.
Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Analysis by Equipment Format
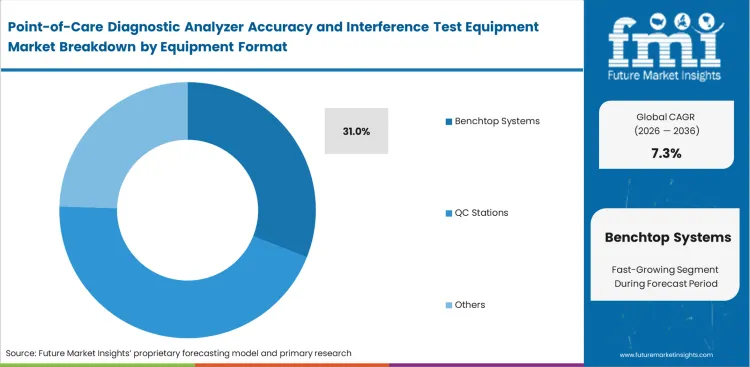
Isolated manual calibration routines simply fail to support modern hospital networks handling high-volume data integration. Central laboratory administrators depend heavily on fixed units to anchor their facility-wide quality control initiatives reliably. Setting up comprehensive glucose monitoring devices validation stations provides unmatched repeatability during routine comparative assessments. Consolidating these verification workflows onto dedicated hardware reduces reagent waste and keeps analytical variability under tighter control. Benchtop systems are expected to account for 31.0% share in 2026, supported by demand for more durable testing infrastructure, as portable simulators tend to wear out faster in demanding clinical settings. Facilities that rely on lower-cost portable units often face higher lifecycle spending over time.
- Bulk Consumable Purchasing: Centralizing verification work on benchtop systems creates clear savings in formulated control matrices. Larger standardized consumable volumes also improve pricing leverage on routine laboratory supplies.
- Hidden Maintenance Burdens: Complex benchtop architecture introduces significant preventative maintenance requirements. Clinical engineering directors must allocate specialized technician hours to maintain internal optical and fluidic stability.
- Lifecycle Comparative Metrics: Total ownership cost analysis reveals benchtop systems outperforming portable alternatives over five-year cycles. Financial administrators recognize superior return on investment despite higher initial capital outlays.
Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Analysis by End User
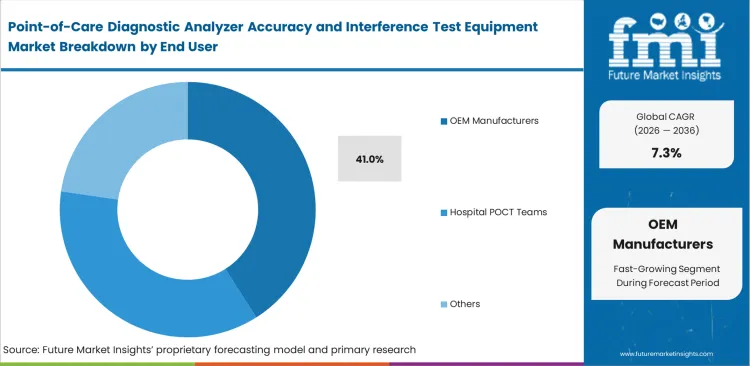
Diagnostic engineering teams must procure massive quantities of specialized evaluation hardware to satisfy strict pre-market approval requirements. These development entities utilize comprehensive interference matrices to validate new analyzer designs against hundreds of potential cross-reacting compounds. Reflecting this intensive R&D consumption, the OEM manufacturers category is likely to account for 41.0% share in 2026 across the global market. Running tests through dedicated blood sugar tester simulation equipment accelerates regulatory submission timelines considerably. Securing rapid clearance from health authorities depends entirely on proving absolute analytical specificity during early development phases, pushing formulation chemists to test interference concentrations far beyond standard physiological levels.
- Regulatory Baseline Thresholds: Standard verification matrices exceed basic accuracy requirements easily during controlled laboratory testing. R&D directors achieve rapid preliminary validation for novel diagnostic prototypes.
- Pathological Edge Failures: Significant analytical gaps appear when prototype devices encounter severe hyperlipidemic or hemolyzed sample conditions. Formulation chemists must redesign sensor architecture to mitigate these complex matrix effects.
- Clinical Qualification Mandates: Achieving commercial release demands flawless performance across exhaustive interference panels. Regulatory affairs officers must submit uncompromising data proving absolute patient safety.
Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Analysis by Sample Matrix
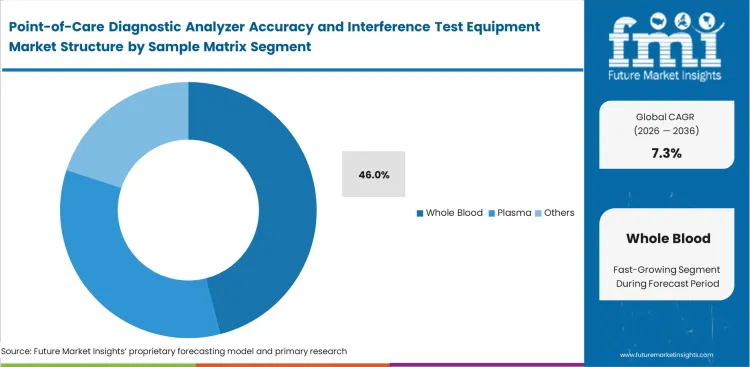
As phlebotomy supervisors prioritize exact clinical correlation in emergency scenarios, whole blood is anticipated to capture 46.0% of the market in 2026 to support critical care diagnostic workflows. Verifying analyzer performance using native blood testing equipment matrices guarantees authentic baseline readings. Moving validation protocols away from artificial aqueous controls toward true whole blood formulations dramatically improves overall diagnostic confidence for bedside interventions. Behind the scenes, logistics managers constantly battle the severe cold-chain storage requirements and short shelf lives that characterize authentic control products. Despite these handling challenges, relying exclusively on stable surrogate matrices severely compromises the validity of decentralized critical care testing.
- Initial Validation Criteria: Hospital POCT coordinators select whole blood controls prioritizing exact clinical correlation. Purchasing decisions favor matrices mimicking true patient samples perfectly.
- Routine Qualification Metrics: Ongoing daily performance checks validate lot-to-lot consistency. Laboratory technicians execute rigorous comparative runs ensuring continuous sensor stability.
- Long-Term Contract Expansion: Expanding decentralized clinical networks continue to lift consumable demand. Multi-year supply agreements are becoming more common as providers work to secure stable pricing for fragile whole blood controls.
Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Drivers, Restraints, and Opportunities
Hospital risk managers face steep financial penalties if bedside tests deliver incorrect results, forcing them to tightly control decentralized testing. Quality directors know that trusting a device's original factory calibration often leads to serious treatment mistakes and costly lawsuits. To manage this legal and clinical risk, hospitals are installing dedicated accuracy verification stations across every department. Putting off the rollout of connected in vitro diagnostics tracking systems leaves critical care teams working with dangerous blind spots. The daily operational need to check thousands of portable analyzers against central lab standards is pushing facilities to acquire this validation hardware immediately.
Mixing different brands of diagnostic hardware makes it incredibly hard to pull all verification data into one clear view. Hospital IT teams constantly struggle to force closed, proprietary data from various bedside devices onto a single quality dashboard. This roadblock exists by design. Competing analyzer makers actively fight open data standards to keep hospitals locked into buying their specific testing supplies. When facilities try to bypass this and build their own oversight networks, they run into massive custom software development costs. Failing to properly link these tools, including infectious disease diagnostics tracking systems, leaves hospital administrators unable to effectively monitor their scattered testing fleets.
- Middleware Integration Platforms: Software engineering teams develop universal translation protocols linking diverse analyzer outputs. IT directors achieve seamless quality control data consolidation across fragmented hospital networks.
- Lyophilized Matrix Stabilization: Formulation scientists create extended-shelf-life whole blood molecular diagnostics controls. Logistics managers eliminate expensive cold-chain shipping requirements for routine verification consumables.
- Automated Interference Screening: Diagnostic manufacturers embed multi-wavelength optical sensors directly into portable testing hardware. Nursing supervisors avoid administering erroneous treatments caused by severe sample hemolysis or lipemia.
Regional Analysis
Regional adoption of decentralized diagnostic validation relies heavily on local regulatory environments and healthcare infrastructure investments. Developing nations prioritize rapid expansion and localized quality control, while mature geographies focus on strict compliance and unified data integration.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 10.1% |
| China | 9.3% |
| United States | 8.1% |
| South Korea | 8.0% |
| United Kingdom | 7.1% |
| Germany | 6.9% |
| Japan | 6.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia-Pacific Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Analysis
Rapid decentralization of primary diagnostic services and aggressive state-sponsored manufacturing initiatives shape validation demands across this combined region. Hospital networks need reliable analytical baselines independent of continuous grid power, while domestic engineering teams consume specialized matrices to accelerate device clearance. Regional health administrators favor simplified visual comparator systems in remote areas, contrasting sharply with the advanced digital integration required in metropolitan hubs.
- India: Private secondary care facilities are expanding quickly across emerging urban centers, creating a strong need for localized quality control. Demand for testing hardware in India is anticipated to see sales grow at a CAGR of 10.1% over the forecast period, reflecting a surge in bedside testing fleets. Securing these regional networks creates long-term supply contracts for medical distributors.
- China: Mandatory local sourcing policies in China are pushing international diagnostic brands to work directly with domestic validation laboratories. Advanced simulation platforms are being used to meet strict clinical concordance standards for multiplex molecular diagnostics arrays, and the market in China is projected to grow at a CAGR of 9.3% during the study period. International manufacturers risk losing access to public hospitals if they do not establish these local verification partnerships.
- South Korea: Projected to witness 8.0% CAGR through 2036, South Korea relies on concentrated metropolitan healthcare systems that demand seamless digital integration. IT directors require complete middleware compatibility for new verification hardware to eliminate manual transcription errors across smart-hospital environments.
- Japan: An aging demographic accelerates the rollout of remote geriatric care networks. Adoption of localized testing validation in Japan is expected to move ahead at a CAGR of 6.4% from 2026 to 2036, as nursing home administrators seek ultra-stable formulated controls with extended ambient shelf lives. Complex cold-chain logistics simply do not work for these dispersed eldercare facilities.
Across the Asia-Pacific region, the shift toward localized healthcare delivery and domestic manufacturing independence forces both international and local diagnostic suppliers to adapt their quality control strategies to highly specific regional constraints.
North America Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Analysis
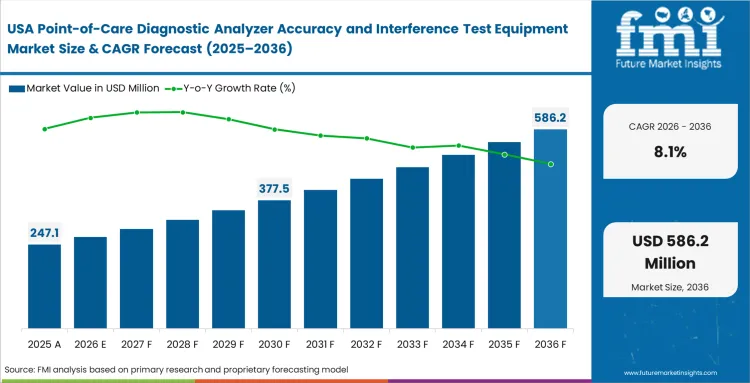
Zero-tolerance regulatory enforcement regarding decentralized testing accuracy defines the operational baseline across North American healthcare networks. Consolidated hospital systems use their immense purchasing power to force diagnostic suppliers into providing unified validation ecosystems instead of isolated proprietary tools. Aggressive standardization pushes strictly limit commercial opportunities for niche verification hardware lacking enterprise IT integration capabilities.
- United States: Stringent CLIA waiver requirements in the United States require continuous daily accuracy checks across a broad ambulatory clinic base. Tight control over consumable spending remains essential because routine verification testing creates a recurring operating cost. That cost pressure influences buying decisions just as much as instrument performance. Demand for point of care diagnostics accuracy and interference test equipment in the United States is projected to grow at a CAGR of 8.1% through 2036. Vendors that build a strong position in this tightly regulated market often gain an advantage when quality control expectations begin shaping standards in other regions.
North American laboratory directors consistently reject fragmented verification tools, opting instead for universal data management platforms that can scale across extensive enterprise IT networks.
Western Europe Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market Analysis
Unified regional diagnostic device regulations require manufacturers to prove analytical stability across diverse patient populations. Centralized national health systems place strong emphasis on long-term lifecycle value and often rely on multi-year service agreements covering both core analytical systems and specialized control matrices. Established clinical chemistry vendors with broad diagnostic portfolios remain well placed in this environment.
- United Kingdom: National Health Service priorities are centered on integrated community diagnostic centers, where consistent test quality outside traditional hospital settings remains a core requirement. Automated molecular diagnostics testing system validation protocols are being standardized to maintain uniform analytical performance across decentralized care sites. That centralized structure gives the country a concentrated buying model, which shapes vendor selection and rollout pace. Demand in the United Kingdom is expected to grow at a CAGR of 7.1% through 2036.
- Germany: Close ties between clinical research institutions and diagnostic manufacturers sustain demand for advanced interference testing. Research teams evaluating novel biomarkers against established validation matrices place added emphasis on precision, repeatability, and early analytical reliability. This gives new verification technologies a chance to build clinical credibility inside influential research settings before broader commercial adoption. The Germany market is projected to expand at a CAGR of 6.9% through 2036.
FMI analysis suggests that Western European purchasing strategies continue to favor established clinical chemistry companies with broad diagnostic portfolios and the ability to meet strict continental validation requirements. The report covers additional countries across Latin America, Eastern Europe, and the Middle East & Africa. Markets outside the main growth centers face different operating constraints, especially in maintaining cold-chain integrity for unstable biological control matrices.
Competitive Aligners for Market Players
Companies making accuracy testing equipment generally fall into two camps: those selling only the testing liquids and those selling complete testing platforms. Heavyweights like Bio-Rad Laboratories and Siemens Healthineers use their massive presence in central hospital labs to set the rules for how bedside testing should be checked. Independent lab directors, though, usually prefer third-party testing liquids. This prevents any built-in bias when they evaluate closed-system equipment like integrated clinical chemistry and immunoassay analyzer units. As buyers prefer this independence, instrument makers like Abbott have to constantly prove their internal testing methods are just as accurate as outside standards.
Established clinical chemistry companies hold a massive advantage by owning vast libraries of biological formulas that new competitors cannot easily copy. Creating stable in vitro diagnostic substrate liquids that perfectly mimic real human blood takes decades of specialized protein research. Companies like Radiometer and Werfen heavily protect their specific methods for keeping hemoglobin stable during transport and storage. When new startups try to make generic versions of these testing liquids, regulators block them almost immediately if the formulas break down under varying shipping temperatures.
Hospital buying teams are tired of dealing with mismatched equipment and fragmented software. They now demand single, unified contracts for all their diagnostic needs. Purchasing directors simply refuse to track one supply chain for the bedside analyzers and a completely different one for the testing liquids. To win these hospital contracts, suppliers like Nova Biomedical and EKF Diagnostics have to bundle their verification software, testing hardware, and control liquids into one simple price-per-test package. As validation rules get stricter, the market naturally favors vendors who can hand hospitals a fully automated, digitally connected compliance system right out of the box.
Key Players in Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment Market
- Bio-Rad Laboratories
- Abbott
- Siemens Healthineers
- Radiometer
- Nova Biomedical
- EKF Diagnostics
- Werfen
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 500 million to USD 1,010 million, at a CAGR of 7.3% |
| Market Definition | Point-of-Care Diagnostic Analyzer Accuracy and Interference Test Equipment represents specialized validation hardware and formulated reference matrices engineered strictly to quantify measurement drift and analytical error in decentralized diagnostic devices. Laboratory directors utilize these systems to confirm that bedside analyzers deliver results statistically identical to central laboratory instrumentation. |
| Segmentation | By Test Function, By Analyzer Type, By Equipment Format, By End User, By Sample Matrix, By Region |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, China, India, Germany, Japan, United Kingdom, South Korea |
| Key Companies Profiled | Bio-Rad Laboratories, Abbott, Siemens Healthineers, Radiometer, Nova Biomedical, EKF Diagnostics, Werfen |
| Forecast Period | 2026 to 2036 |
| Approach | Baseline commercial shipments of formulated control matrices and specialized simulator hardware. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
By Test Function:
- Accuracy verification
- Interference testing
- Precision testing
- Linearity testing
- Method comparison
By Analyzer Type:
- Glucose analyzers
- Blood gas analyzers
- Electrolyte analyzers
- Coagulation analyzers
- Cardiac analyzers
By Equipment Format:
- Benchtop systems
- Portable simulators
- QC stations
- Comparator readers
- Data platforms
By End User:
- OEM manufacturers
- Hospital POCT teams
- Reference labs
- Validation labs
- Academic centers
By Sample Matrix:
- Whole blood
- Capillary blood
- Plasma
- Serum
- Control matrix
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Centers for Disease Control and Prevention. (2024). Transcript: Point of care testing.
- Centers for Medicare & Medicaid Services. (2026). Clinical Laboratory Improvement Amendments (CLIA).
- Sacks, D. B., Kirkman, M. S., & Little, R. R. (2024). Point-of-care HbA1c in clinical practice: Caveats and considerations for optimal use. Diabetes Care, 47(7), 1104-1110.
- Baumstark, A., Pleus, S., Mende, J., Jendrike, N., Tesar, M., Schauer, S., Sugiyama, T., Aoki, T., Sugiura, M., Freckmann, G., & Haug, C. (2024). Investigation of the effect of 70 potential interferents on measurement results of two blood glucose monitoring systems. Journal of Diabetes Science and Technology, 19(4), 971-981.
- Koechlin, L., Boeddinghaus, J., Lopez-Ayala, P., Reber, C., Nestelberger, T., Wildi, K., Spagnuolo, C. C., Strebel, I., Glaeser, J., Bima, P., Crisanti, L., Herraiz-Recuenco, L., Dubach, E., Miró, Ò., Martin-Sanchez, F. J., Kawecki, D., Keller, D. I., Christ, M., Buser, A., … Mueller, C. (2024). Clinical and analytical performance of a novel point-of-care high-sensitivity cardiac troponin I assay. Journal of the American College of Cardiology, 84(8), 726-740.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Specific labor hours POCT coordinators allocate to routine decentralized device validation.
- Strategic procurement shifts regarding whole blood control matrices versus stable aqueous formulations.
- Financial impact of comprehensive middleware integration on massive hospital network testing fleets.
- Hidden lifecycle expenditures central laboratory directors face when purchasing portable simulation hardware.
- Regulatory bottlenecks delaying commercial clearance regarding novel diagnostic analyzer designs.
- Deep technical friction preventing seamless data reconciliation across fragmented bedside hardware platforms.
- Absolute zero-tolerance accuracy enforcement shaping adoption curves across North American ambulatory clinics.
- Aggressive state-sponsored domestic manufacturing initiatives reshaping diagnostic benchmarking standards in East Asia.
Frequently Asked Questions
What is point-of-care analyzer interference testing equipment?
This equipment includes specialized validation hardware and formulated reference matrices engineered strictly to quantify measurement drift and analytical error caused by specific substances in decentralized diagnostic devices.
How large is the POCT analyzer QC equipment market by 2036?
Demand is expected to advance to USD 1,010 million by 2036 as clinical laboratories tighten validation protocols.
What was the recorded valuation of this industry in 2025?
FMI notes the sector was valued at USD 470 million in 2025.
What is the anticipated CAGR for this sector through 2036?
Sales are expected to reach a CAGR of 7.3% during the forecast period.
Why is interference testing different from routine QC?
Interference testing specifically evaluates common chemical cross-reactivity issues in complex biological samples, whereas routine accuracy checks focus on baseline analytical concordance.
Which test function segment currently leads adoption?
Accuracy verification is projected to lead with 38.0% share in 2026.
Which analyzer types create the most validation demand?
Glucose analyzers and blood gas systems generate the highest demand due to massive installed footprints forcing nursing supervisors to manage thousands of individual verification events weekly.
What standards govern POCT analyzer accuracy studies?
Stringent CLIA waiver requirements and specific FDA guidance dictate continuous daily accuracy checks across ambulatory clinics and decentralized testing sites.
Which equipment format dominates central laboratory purchasing?
Benchtop systems are poised to garner 31.0% share in 2026.
Why do facility managers prefer benchtop systems over portable units?
Central laboratory administrators depend on robust benchtop units to anchor facility-wide quality control initiatives reliably.
Which end user segment dominates hardware acquisition?
OEM manufacturers are set to represent 41.0% share in 2026.
How do OEM validation budgets differ from hospital POCT budgets?
OEM manufacturers procure massive quantities of specialized hardware for pre-market clearance cycles, while hospital budgets focus on continuous daily operational compliance.
Which sample matrix remains the preferred validation standard?
Whole blood is likely to account for 46.0% share in 2026.
Why do POCT coordinators prefer whole blood control matrices?
Verifying analyzer performance using native whole blood matrices guarantees authentic clinical correlation during bedside emergencies.
Which countries are growing fastest in POCT validation equipment?
India leads regional growth trajectories at a CAGR of 10.1%, followed closely by China at 9.3% due to aggressive healthcare infrastructure investments.
What drives rapid growth in the Indian diagnostic sector?
Aggressive expansion of private secondary care facilities dictates localized quality control requirements across emerging urban centers.
What specific policy shapes diagnostic validation in China?
Aggressive state-sponsored initiatives targeting total domestic manufacturing independence dictate the diagnostic validation landscape here.
What is the growth projection for the United States?
USA demand across the sector is set to rise at 8.1% CAGR through 2036.
How does the United Kingdom sector perform?
United Kingdom is likely to post a CAGR of 7.1% in verification equipment by 2036.
What shapes UK procurement behavior for diagnostic validation?
National Health Service mandates prioritize the rapid deployment of integrated community diagnostic centers requiring standardized oversight.
What technological requirement defines South Korean hardware adoption?
Concentrated metropolitan healthcare systems demand seamless digital integration of all diagnostic validation workflows natively.
Which companies are active in this space?
Key suppliers include Bio-Rad Laboratories, Abbott, Siemens Healthineers, Radiometer, Nova Biomedical, EKF Diagnostics, and Werfen.
What operational friction slows validation hardware adoption globally?
Fragmented digital infrastructure across diverse diagnostic hardware brands severely restricts efficient verification data consolidation.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Function , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Function , 2026 to 2036
- Accuracy Verification
- Interference Testing
- Others
- Accuracy Verification
- Y to o to Y Growth Trend Analysis By Test Function , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Function , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Analyzer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Analyzer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Analyzer Type, 2026 to 2036
- Glucose Analyzers
- Blood Gas Analyzers
- Others
- Glucose Analyzers
- Y to o to Y Growth Trend Analysis By Analyzer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Analyzer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Equipment Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Equipment Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Equipment Format, 2026 to 2036
- Benchtop Systems
- QC Stations
- Others
- Benchtop Systems
- Y to o to Y Growth Trend Analysis By Equipment Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Equipment Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- OEM Manufacturers
- Hospital POCT Teams
- Others
- OEM Manufacturers
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sample Matrix
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sample Matrix, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sample Matrix, 2026 to 2036
- Whole Blood
- Plasma
- Others
- Whole Blood
- Y to o to Y Growth Trend Analysis By Sample Matrix, 2021 to 2025
- Absolute $ Opportunity Analysis By Sample Matrix, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Function
- By Analyzer Type
- By Equipment Format
- By End User
- By Sample Matrix
- Competition Analysis
- Competition Deep Dive
- Bio-Rad Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott
- Siemens Healthineers
- Radiometer
- Nova Biomedical
- EKF Diagnostics
- Bio-Rad Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Function , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Analyzer Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Sample Matrix, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Test Function , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Analyzer Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Sample Matrix, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Test Function , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Analyzer Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Sample Matrix, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Test Function , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Analyzer Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Sample Matrix, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Test Function , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Analyzer Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Sample Matrix, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Test Function , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Analyzer Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Sample Matrix, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Test Function , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Analyzer Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Sample Matrix, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Test Function , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Analyzer Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Sample Matrix, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Function , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Test Function , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Test Function
- Figure 6: Global Market Value Share and BPS Analysis by Analyzer Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Analyzer Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Analyzer Type
- Figure 9: Global Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Equipment Format
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Sample Matrix, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Sample Matrix, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Sample Matrix
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Test Function , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Test Function , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Test Function
- Figure 32: North America Market Value Share and BPS Analysis by Analyzer Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Analyzer Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Analyzer Type
- Figure 35: North America Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Equipment Format
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Sample Matrix, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Sample Matrix, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Sample Matrix
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Test Function , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Test Function , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Test Function
- Figure 48: Latin America Market Value Share and BPS Analysis by Analyzer Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Analyzer Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Analyzer Type
- Figure 51: Latin America Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Equipment Format
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Sample Matrix, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Sample Matrix, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Sample Matrix
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Test Function , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Test Function , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Test Function
- Figure 64: Western Europe Market Value Share and BPS Analysis by Analyzer Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Analyzer Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Analyzer Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Equipment Format
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Sample Matrix, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Sample Matrix, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Sample Matrix
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Test Function , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Test Function , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Test Function
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Analyzer Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Analyzer Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Analyzer Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Equipment Format
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Sample Matrix, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Sample Matrix, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Sample Matrix
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Test Function , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Test Function , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Test Function
- Figure 96: East Asia Market Value Share and BPS Analysis by Analyzer Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Analyzer Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Analyzer Type
- Figure 99: East Asia Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Equipment Format
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Sample Matrix, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Sample Matrix, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Sample Matrix
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Test Function , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Function , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Test Function
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Analyzer Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Analyzer Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Analyzer Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Equipment Format
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Sample Matrix, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Sample Matrix, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Sample Matrix
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Test Function , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Test Function , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Test Function
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Analyzer Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Analyzer Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Analyzer Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Equipment Format
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Sample Matrix, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Sample Matrix, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Sample Matrix
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

