Demand for INR test meters in the UK is projected at USD 186.0 million in 2026 and is expected to reach USD 353.5 million by 2036, expanding at a 6.6% CAGR. UK demand is anchored in clinical accountability for anticoagulation control, rising screening intensity for stroke risk pathways, and hospital workflows that require rapid decision support. INR monitoring remains central for patients managed on vitamin K antagonists, where dosing adjustments depend on timely readings and consistent quality controls.
INR test meters operate as decision tools, not just diagnostic devices. Stakeholders evaluate them on analytical reliability, ease of use, sample handling discipline, and the ability to reduce avoidable escalation events linked to unstable coagulation control. Hospital leaders focus on patient safety outcomes and throughput efficiency, while technology providers measure value through integration readiness, device uptime, and staff training burden. For non-expert users, the benefits are easier to understand as faster results and fewer repeat visits when monitoring routines are stable.
Operational adoption is strengthened when testing supports both facility-based services and supervised self-testing programs. Procurement teams prioritize traceability, predictable consumables supply, and device performance consistency across wards, clinics, and community pathways. Service providers supporting connected health ecosystems prioritize data capture, remote review capability, and audit-friendly reporting structures that fit NHS governance and quality assurance expectations.
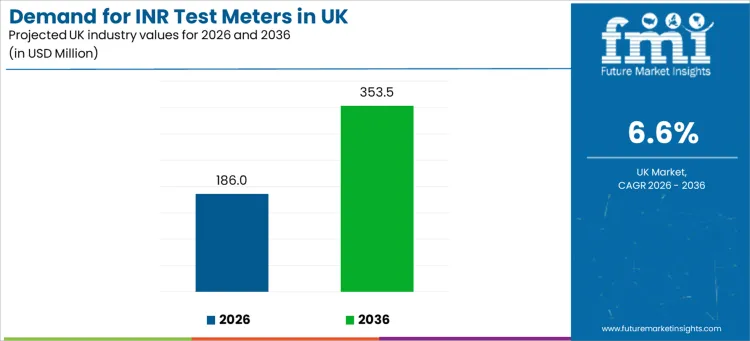
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 186.0 million |
| Industry Forecast Value (2036) | USD 353.5 million |
| Forecast CAGR (2026 to 2036) | 6.6% |
UK anticoagulation care depends on monitoring discipline, because dose decisions are anchored to INR values for patients managed on warfarin. National clinical guidance and specialist service models reinforce routine monitoring and escalation rules, which keeps testing volume steady across hospitals and community-led care. NHS-aligned pathways place strong emphasis on safety governance, making analytical confidence and repeatability critical buying criteria.
Demand strengthens when providers shift monitoring closer to the patient. Hospital trusts and community services value point-of-care tools that reduce delays between sampling and clinical action. Faster results support same-visit dose decisions, fewer call-backs, and tighter control over patients whose INR values fluctuate. This is where compact INR testing systems become operationally meaningful for clinics and ambulatory settings that balance limited appointment capacity with high patient throughput.
Self-testing and supervised home-based routines add another layer of demand. Some NHS service frameworks have recognized self-monitoring options for suitable patients, based on preference and capability. These pathways raise the importance of user-friendly meters, simple strip workflows, and error-resistant sampling processes
Innovation priorities are shifting toward connected diagnostics and clinical data continuity. Device makers that enable structured data capture and interoperable reporting gain attention from digital health stakeholders. This aligns with broader in-vitro diagnostics and point-of-care category expansion, where fast testing supports clinical decisions outside central labs. Demand themes align with point-of-care diagnostics and coagulation analyzers.
The segmentation structure in the UK reflects where INR readings are consumed at scale, how clinical accountability is managed, and which product components fit operational routines across wards, clinics, surgical pathways, and homecare support models.
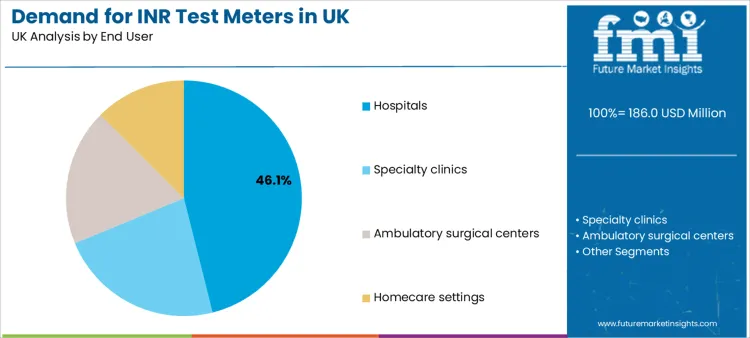
Hospitals account for a 46.1% share, making them the leading end user. Hospital environments require fast INR availability for dose correction, peri-procedure evaluation, and escalation planning when bleeding risk rises. The preference is shaped by standard operating protocols, clinical documentation requirements, and risk management frameworks that link INR stability to patient safety outcomes.
Hospitals value meters that reduce time-to-decision without compromising confidence in measurement quality. Procurement leaders evaluate device validation support, operator training pathways, consumables traceability, and availability of service support. For executives managing multi-site delivery networks, the strongest value sits in consistent performance across wards and outpatient clinics, which stabilizes clinical workflow and reduces repeat testing.
Specialty clinics capture demand where anticoagulation services are designed around repeat visits, structured monitoring, and patient education. INR test meters support faster patient flow by reducing reliance on centralized lab turnaround. Clinics measure value through appointment efficiency, fewer delayed dose changes, and smoother follow-up routines that reduce avoidable readmissions.
This segment prioritizes compact footprints, fast result displays, and consumables availability that supports scheduled care volumes. Clinics that support chronic therapy monitoring value predictable workflows that can be managed by trained nursing staff without heavy lab infrastructure.
Ambulatory surgical centers need INR confirmation when anticoagulation history influences procedural planning and bleeding risk governance. Point-of-care capability supports quick triage decisions and reduces procedural delays tied to pending lab results. Buyers focus on accuracy confidence, rapid usability, and simple quality checks that fit high-throughput environments.
When testing is embedded into pre-op workflows, meter reliability and operator consistency become major selection criteria. This makes device training, quality control routines, and supplier responsiveness key differentiators.
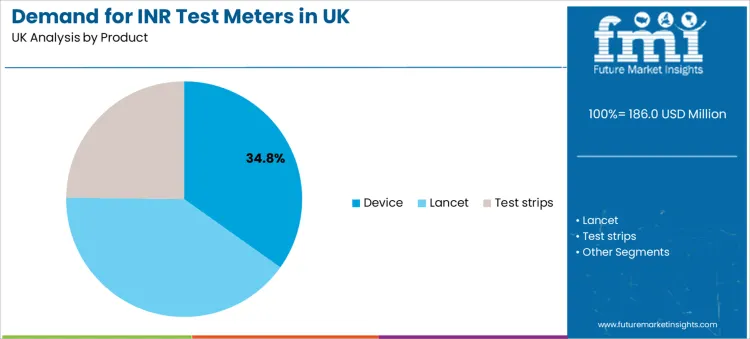
Devices hold a 34.8% share, making them the leading product category. The device sits at the center of usability, measurement stability, and operational control. Stakeholders focus on sample handling simplicity, error alerts, calibration discipline, and robustness under frequent use.
Device leadership also reflects broader investment in scalable testing infrastructure. Hospitals and service providers treat device fleets as systems that require maintenance planning, standardized training, and consistent reporting routines. Category momentum ties closely to INR testing expansion themes tracked across INR test meters.
Test strips and lancets define recurring economics and supply resilience. Consistent strip performance supports measurement repeatability, while dependable availability protects service continuity. Buyers evaluate storage stability, lot traceability, and compatibility control across device versions.
For suppliers, consumables create long-term relationships when they deliver predictable performance and reduce user errors. For hospital procurement and clinic managers, consumables define total cost discipline and operational dependability across the full monitoring lifecycle.
Demand is strengthened by INR-based anticoagulation control needs and the operational value of faster decisions in care settings. Point-of-care routines support time efficiency and reduce delays between sampling and treatment action. UK care delivery is influenced by clear device governance and compliance rules for in-vitro diagnostics, which improves purchase confidence for regulated clinical use.
Clinical governance emphasis increases the need for audit-ready testing logs, standardized operator routines, and quality assurance workflows. This favors suppliers that provide structured training models, clear QC processes, and support for service documentation.
Analytical reliability expectations are a core restraint. Procurement teams hesitate when accuracy confidence is unclear across different operators or patient conditions. Operational variability can appear when sampling technique differs across staff or when consumables handling is inconsistent.
Integration gaps can slow adoption in digitally mature providers. When reporting systems cannot capture results smoothly, clinical teams face manual documentation burden, which reduces perceived value.
Connected monitoring systems remain a key opportunity. Service providers and digital health partners can expand adoption by enabling secure result sharing, remote review workflows, and structured patient follow-up routines.
Community-based anticoagulation services offer growth potential where testing supports distributed care and reduces hospital dependency. This aligns with broader growth pathways seen across rapid coagulation testing.
Supply instability for consumables can disrupt service reliability. Budget pressure across healthcare systems can intensify price competition and extend device replacement cycles. Shifts toward alternative anticoagulants that reduce routine INR monitoring can influence long-term testing volumes for certain patient groups, placing greater emphasis on diversified clinical use cases where INR testing remains essential.
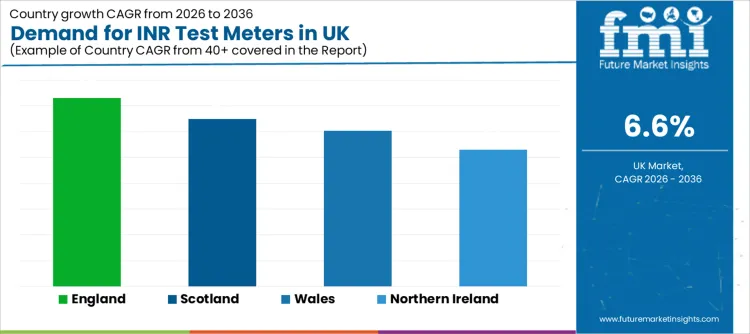
| Region | CAGR 2026 to 2036 |
|---|---|
| England | 7.3% |
| Scotland | 6.5% |
| Wales | 6.0% |
| Northern Ireland | 5.3% |
England grows at 7.3%, supported by larger hospital networks, higher patient throughput, and stronger adoption of operational tools that reduce decision delays. Demand rises when INR testing supports rapid dose adjustment workflows and standardization across multiple sites. Digital health priorities and structured service governance amplify interest in connected, audit-friendly testing routines.
Device fleets gain traction where procurement teams seek consistent performance across wards, outpatient clinics, and supervised community services. For technology service providers, England remains the most active region for integration-enabled diagnostics across larger delivery footprints.
Scotland increases at 6.5%, shaped by demand for reliable monitoring routines that fit both urban and remote care contexts. INR test meters support continuity where travel burden and appointment spacing can affect follow-up discipline.
Providers value tools that support fast testing cycles and reduce patient disruption during routine monitoring. This region favors operational simplicity, robust device handling, and dependable consumables supply to sustain steady service delivery across varied care settings.
Wales grows at 6.0%, driven by care models that prioritize service efficiency and structured patient management. INR testing supports repeatable monitoring routines that reduce delays and protect patient safety outcomes.
Stakeholders value devices that are easy to operate and support consistent measurement discipline across clinical staff teams. For suppliers, growth depends on training support, stable strip availability, and consistent performance that fits standardized monitoring schedules.
Northern Ireland rises at 5.3%, reflecting smaller-scale demand clusters and careful adoption patterns. Buyers prioritize proven device performance, dependable support services, and stable consumables availability. Growth strengthens when clinical teams confirm repeatability and when workflows integrate smoothly into existing anticoagulation service routines.
Competition is defined by measurement reliability, consumables performance, training support, and service responsiveness. Hospitals prioritize systems that support consistent results across different operators and shift patterns. Clinics and ambulatory centers value fast usability and dependable strip supply that prevents cancellations and repeat appointments.
Technology stakeholders assess the ability to support data continuity, patient monitoring workflows, and reporting discipline. Suppliers that offer structured onboarding and quality control guidance gain credibility with governance-led procurement teams.
Product differentiation is strongest when device design reduces user errors, supports clear prompts, and provides stable results under high-frequency usage. Brand confidence is reinforced when clinical service teams can rely on supplier support for validation, troubleshooting, and ongoing consumables availability. The competitive environment connects closely with broader coagulation testing categories, including special coagulation testing demand themes.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| End User | Hospitals; Specialty clinics; Ambulatory surgical centers; Homecare settings |
| Product | Device; Lancet; Test strips |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd; Lepu Medical Technology(Beijing)Co., Ltd; ACON Laboratories, Inc.; CoaguSense Inc.; Abbott |
How big is the demand for inr test meters in uk in 2026?
The demand for inr test meters in uk is estimated to be valued at USD 186.0 million in 2026.
What will be the size of inr test meters in uk in 2036?
The market size for the inr test meters in uk is projected to reach USD 353.5 million by 2036.
How much will be the demand for inr test meters in uk growth between 2026 and 2036?
The demand for inr test meters in uk is expected to grow at a 6.6% CAGR between 2026 and 2036.
What are the key product types in the inr test meters in uk?
The key product types in inr test meters in uk are device, lancet and test strips.
Which end user segment is expected to contribute significant share in the inr test meters in uk in 2026?
In terms of end user, hospitals segment is expected to command 46.1% share in the inr test meters in uk in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.