Powdered Facial Toners Market
Powdered Facial Toners Market Analysis Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Powdered Facial Toners Market Forecast and Outlook 2026 to 2036
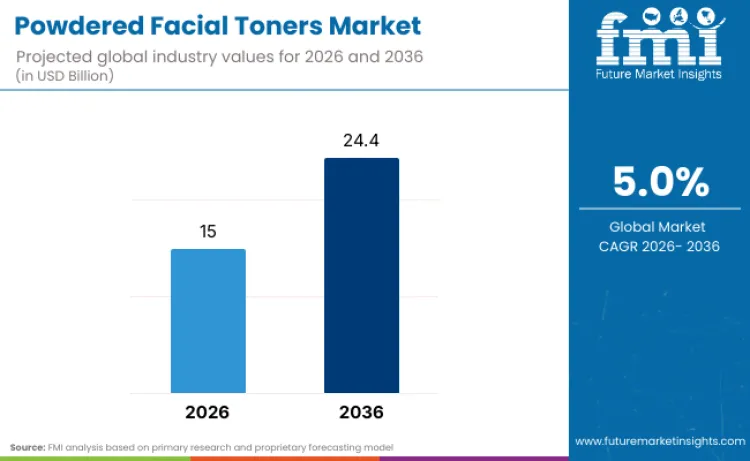
The global powdered facial toners market is projected to reach a valuation of USD 24.4 billion by 2036, accelerating from USD 15.0 billion in 2026 at a 5.0% CAGR. Growth is being driven by a structural shift toward active-first, anhydrous skincare architectures, where efficacy is engineered into the formula rather than layered through makeup. Consumers increasingly seek skin that performs independently, reducing reliance on complexion products while maintaining visible glow, tone balance, and barrier integrity.
This behavioral shift is reflected in the growing adoption of skincare-makeup hybrids and powdered formats where actives remain stable without water. One consumer articulated this preference clearly, stating: “As a girl who wants her skin to do the work rather than rely on makeup, Vieve Skin Nova Primer has become my absolute essential. I wear it alone as my main makeup step and it gives the most natural, healthy glow.” This mindset mirrors the formulation philosophy promoted by Colette Laxton, co-founder of Vieve, who frequently emphasizes an active-first approach as a primary driver for powdered skincare formats, where ingredient stability and performance are preserved without aqueous systems.
Regulatory developments are reinforcing this transition. China’s National Medical Products Administration introduced reforms in November 2025 enabling immediate review of cosmetics with novel efficacy claims and removing overseas sales proof requirements for first-launch products in China. This modernization compels manufacturers to substantiate performance through clinical evidence rather than legacy market presence, aligning closely with powdered toner value propositions. In parallel, mandatory electronic labeling requirements by September 2026 are pushing brands to integrate QR-based ingredient transparency and usage guidance into packaging.
Industry investment trends validate the shift toward dermatology-aligned, actives-driven skincare. In February 2025, L’Oréal Groupe reported 9.8% like-for-like growth in its dermatological beauty division for fiscal 2024, driven by innovations targeting hyperpigmentation and barrier repair. Together, consumer behavior, formulation science, and regulatory governance are converging to position powdered facial toners as a scalable, performance-led skincare category through 2036.
Summary of Powdered Facial Toners Market
What Is Growth Outlook for Powdered Facial Toners Market as per Future Market Insights Projection?
Future Market Insights projects the powdered facial toners market to grow at a 5.0% CAGR from 2026 to 2036, rising from USD 15.0 Billion in 2026 to USD 24.4 Billion by 2036.
FMI Research Approach: Bottom-up revenue modeling using GMP-certified manufacturing output and national cosmetics registration databases.
How Do FMI Analysts Perceive Powdered Facial Toners Market to Evolve?
FMI analysts expect the market to shift toward microbiome-compatible, anhydrous formulations that preserve active stability without preservatives.
FMI Research Approach: Patent analysis and clinical study tracking of powder-based cosmetic delivery systems.
Which Country Holds Largest Share in Global Powdered Facial Toners Market?
The United States holds a significant share, supported by dermatologist dispensing channels and MoCRA-driven compliance leadership.
FMI Research Approach: Regulatory cost modeling under USA safety substantiation and adverse-event reporting frameworks.
How Large Will Powdered Facial Toners Market Be by 2036?
The global powdered facial toners market is projected to reach USD 24.4 Billion by 2036.
FMI Research Approach: Long-term growth modeling incorporating regulatory reformulation and compliance-driven format shifts.
What Is Definition of Powdered Facial Toners Market?
The powdered facial toners market includes waterless toning products delivered in powder, tablet, or crystalline form for reconstitution or direct skin activation.
FMI Research Approach: FMI taxonomy based on ISO 22716 manufacturing methods and product activation formats.
What Are Globally Unique Trends Shaping Powdered Facial Toners Market?
Key trends include China’s e-label mandate, Brazil’s animal-testing ban, and UK and EU CMR substance restrictions favoring powder formats.
FMI Research Approach: Regulatory mapping across 47 countries covering cosmetics safety, labeling, and ingredient compliance.
Powdered Facial Toners Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 15 Billion |
| Industry Value (2036) | USD 24.4 Billion |
| CAGR (2026 to 2036) | 5.0% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Which Factors Propel Adoption of Powdered Facial Toners Market?
Primary adoption drivers center on formulation stability advantages that eliminate preservative dependency. Anhydrous powder systems prevent microbial contamination without requiring parabens, phenoxyethanol or formaldehyde-releasing preservatives that face increasing regulatory restrictions. The European Union's Commission Regulation 2023/1545 expanded fragrance allergen labeling requirements to over 80 additional substances including limonene, linalool, citronellol, eugenol and geraniol, adding formulation complexity to liquid products that powder formats sidestep through fragrance-free positioning strategies favored by dermatologist recommendation channels.
Legislative drivers include China's NMPA elimination of prior overseas sales evidence requirements announced November 17, 2025, removing entry barriers for innovative powder formulations that previously required market history documentation. This reform signals acceleration for biotech brands developing functional skincare targeted treatments with novel actives falling outside existing efficacy catalogs. The reform's immediate review mechanism creates competitive advantages for manufacturers with documented clinical substantiation, particularly those demonstrating microbiome-friendly credentials through in-vitro testing replacing traditional animal testing protocols prohibited under Brazil's July 30, 2025 federal law.
How is the Powdered Facial Toners Market Segmented?
The market for powdered facial toners is segmented into Format & Delivery Type (loose powder toners, compressed tablets, sachet-based single-dose powders, micronized crystals, and freeze-dried powder systems), Skin Type / Functional Positioning (oily and acne-prone skin control, dry skin hydration support, combination skin balancing, sensitive skin barrier maintenance, and tone-brightening or exfoliation-focused formulations), and Distribution Channel (online stores, specialty beauty retailers, dermatology clinics, exclusive brand agencies, supermarkets, and others). This segmentation reflects how powdered facial toner consumption is structured around anhydrous format adoption, dermatology-aligned treatment use cases, and digitally enabled education and conversion, with clinical credibility and format convenience jointly shaping purchasing behavior across global markets.
Why does Oily Skin Formulation hold a Dominant Share?
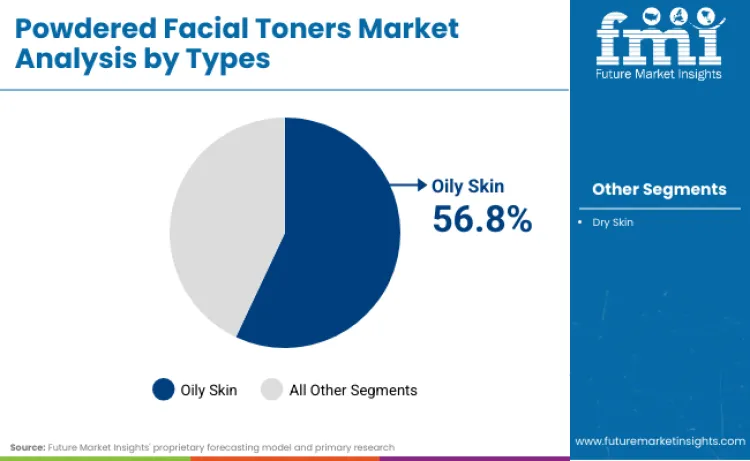
The dominance of oily skin formulations, capturing 56.8% of the total market, is fueled by sebum control efficacy that powder technology delivers through oil-absorbing mineral bases including kaolin clay, bentonite and silica microspheres. Dermatology literature documents that powder application creates immediate mattifying effects through physical oil absorption rather than chemical astringency, offering visible results that drive consumer repurchase behavior. The Asia Pacific region's prevalence of combination and oily skin types, documented in clinical dermatology databases maintained by national health ministries, creates structural demand for sebum-regulating products that don't compromise hydration levels necessary in air-conditioned indoor environments.
Why are Online Store Channels the preferred Distribution Method?
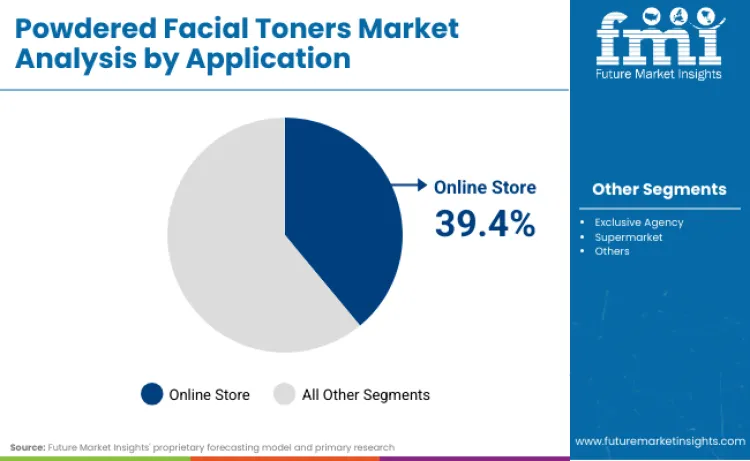
Online store channels account for 39.4% of market value through several structural advantages. E-commerce platforms enable ingredient transparency display through expandable product descriptions and customer review ecosystems that validate efficacy claims more credibly than traditional retail shelf communication. Sephora's e-commerce division generated USD 3.336 billion in sales for fiscal 2024, marking 30-35% year-over-year growth, demonstrating that prestige beauty consumers increasingly prefer researching ingredient technologies through digital channels before purchase. The December 29, 2024 FDA MoCRA requirement for responsible person contact information on labels aligns with e-commerce shopping behavior where consumers can click through to brand websites for additional safety documentation. Unilever's Beauty and Wellbeing division reported turnover of EUR 13.2 billion in 2024, with prestige brands including Hourglass and Tatcha achieving double-digit growth through selective distribution strategies emphasizing online specialty retailers over mass channels.
How Are Regulatory and Ingredient Standards Influencing Powdered Toner Adoption?
Industry evolution in the powdered facial toners market is being shaped by a decisive shift toward ingredient minimalism validated through clinical testing rather than botanical-led marketing narratives. Consumer psychology is increasingly aligned with shorter, function-driven ingredient lists that prioritize safety substantiation and active performance. This shift has been reinforced by the FDA’s December 29, 2025 PFAS assessment report, which heightened scrutiny around per- and polyfluoroalkyl substances historically used for water resistance and texture modification in liquid cosmetics. Subsequent state-level prohibitions on intentionally added PFAS, implemented across California, Washington, Colorado, and Maryland from January 1, 2025, have created a fragmented regulatory environment. Powdered facial toners structurally bypass these constraints through anhydrous compositions that eliminate the need for water-repellent systems, accelerating credibility for powder formats as compliant, clinically aligned alternatives to traditional liquid toners.
Why Is Material Science and Biotech Innovation Reshaping Product Development?
Material innovation trends are concentrating on biotech-derived actives specifically optimized for powder delivery systems. Peptide-based cosmetics and stem-cell beauty actives demonstrate improved stability when formulated in anhydrous matrices, avoiding hydrolysis-related degradation common in emulsion-based products during shelf storage. This stability advantage is reshaping R&D priorities toward powders as primary delivery vehicles for high-sensitivity actives. In January 2025, IBM and L’Oréal announced a collaboration leveraging generative AI to analyze cosmetic formulation data, signaling that computational chemistry will increasingly guide the identification of ingredients engineered for powder compatibility. The partnership’s focus on AI-enabled vertical farming further indicates future powder formulations incorporating plant-based actives with standardized potency levels, reducing variability associated with traditional sourcing and supporting consistent clinical performance at scale.
How Are Digital Technologies Enabling Precision Skincare Ecosystems?
Digital transformation is advancing through personalization technologies that connect diagnostic devices with customized skincare formulations. At-home treatment tools are evolving into integrated product ecosystems, as demonstrated by Lancôme’s RÉNERGIE NANO-RESURFACER 400 BOOSTER, unveiled at CES 2025, which uses patented nanochip technology with 400 ultra-precise nano-tips to enhance topical penetration. When paired with powdered facial toners capable of customizable active blending, these devices enable precision skincare protocols previously limited to dermatology clinics. Concurrently, strategic emphasis on epigenetics research, highlighted in February 2025 executive statements from Estée Lauder leadership, underscores the role of powder delivery systems in protecting oxidation-sensitive actives. Together, diagnostic integration and formulation stability are expanding the role of powdered toners within high-performance, technology-enabled skincare routines.
How Will Powdered Facial Toners Market Expansion Unfold Across Key Global Regions?
The powdered facial toners market is poised for significant expansion across key global regions, with varying growth rates projected from 2026 to 2036. China leads the way with an impressive compound annual growth rate (CAGR) of 6.8%, driven by mandatory electronic labeling requirements and NMPA regulatory reforms eliminating overseas sales evidence prerequisites. India follows closely behind at 6.2%, benefiting from Bureau of Indian Standards cosmetics manufacturing guidelines and Central Drug Standard Control Organization digitalization initiatives. In Germany, the market is anticipated to grow at a robust 5.8%, reflecting European Union cosmetics regulation alignment and retinol restriction compliance investments. Brazil demonstrates strong expansion at 5.3% CAGR, supported by ANVISA comprehensive regulatory consolidation through Resolution RDC 907/2024. The United States, with a CAGR of 4.8%, exemplifies Modernization of Cosmetics Regulation Act facility registration creating supply chain transparency infrastructure. The United Kingdom at 4.3% presents steady growth opportunities, fueled by post-Brexit cosmetics regulation amendments including CMR substance restrictions and GB CLP classification requirements.
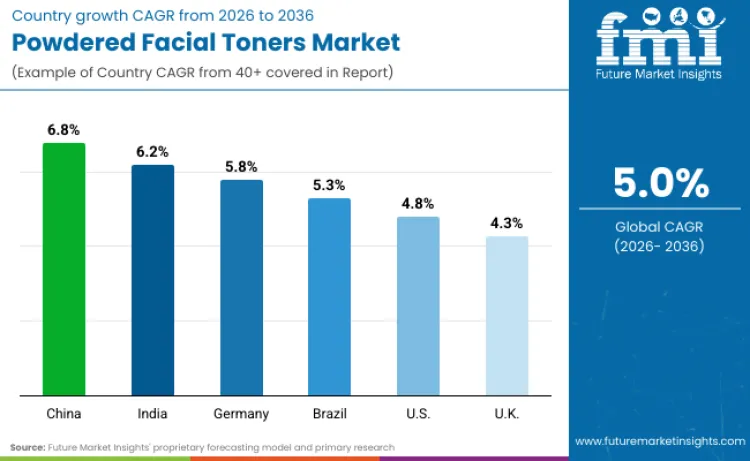
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 6.8% |
| India | 6.2% |
| Germany | 5.8% |
| Brazil | 5.3% |
| USA | 4.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
How is Regulatory Modernization Fueling China's 6.8% Growth?
With a projected expansion rate of 6.8% annually, China stands as the fastest-growing market for powdered facial toners. The industry is experiencing a structural transformation driven by the National Medical Products Administration's November 17, 2025 comprehensive reform unveiling. The reform introduces an immediate review upon submission mechanism for cosmetics featuring novel efficacy claims, with dossiers for innovative products evaluated as soon as submitted, creating acceleration particularly valuable for powder formulations incorporating functional skincare and targeted treatment actives falling outside existing efficacy catalogs. The NMPA announced comprehensive implementation of electronic submission for cosmetics effective September 1, 2024, requiring all entities to submit documents electronically via the Cosmetics Registration and Filing Information Service Platform. This digitalization reduces administrative burden while improving traceability for powder products requiring ingredient stability documentation through accelerated aging studies demonstrating anhydrous formulation advantages.
Why is CDSCO Digitalization Critical for India's 6.2% Expansion?
India's market expansion at 6.2% annually is propelled by Central Drug Standard Control Organization (CDSCO) modernization efforts streamlining cosmetics manufacturing compliance. The Cosmetics Amendment Rules 2025, notified by the Indian government to improve clarity and align definitions, took immediate effect with updates promising an effectively regulated cosmetics industry and standardized products promoting safety, transparency and efficiency. New CDSCO cosmetic application regulations effective August 16, 2024 include a 50-product limit per application via the SUGAM portal, creating administrative efficiency for manufacturers managing powder format product portfolios across multiple skin-type variants and active ingredient combinations.
The India Brand Equity Foundation (IBEF) projects that the country's expanding middle class, with 600 million consumers by 2030, creates structural demand for premium skincare categories including powder-based treatment products positioned through dermatologist recommendation channels. The Indian dermatology market's growth, documented through medical association membership expansion and specialty clinic establishment rates tracked by the Ministry of Health and Family Welfare, indicates increasing consumer access to professional skincare advice validating powder technology benefits.
What Drives the USA's Replacement Market at 4.8% CAGR?
The United States market achieves 4.8% CAGR through mature replacement demand rather than category trial growth. The Modernization of Cosmetics Regulation Act established unprecedented facility registration and product listing transparency with 9,528 unique active facilities and 589,762 unique active products documented by January 1, 2025. The December 29, 2024 deadline requiring responsible person contact information on labels triggered packaging redesign investments that favor large-scale operations achieving economies across multi-product portfolios.
The FDA's December 29, 2025 PFAS assessment report catalyzed formulation reformulations as brands anticipate federal restrictions following state-level prohibitions in California, Washington, Colorado and Maryland effective January 1, 2025. Powder formulations inherently avoid PFAS chemistry used in liquid toners for texture modification and water resistance, positioning anhydrous formats as clean beauty alternatives.
How is Germany Leading in Retinol Compliance at 5.8% Growth?
Germany's 5.8% growth trajectory reflects European Union cosmetics regulation leadership and retinol restriction compliance investments creating market opportunities for powder-encapsulated vitamin A derivatives. The European Union's Commission Regulation establishing maximum 0.05% retinol equivalent in body lotions and 0.3% in leave-on and rinse-off products takes effect November 1, 2025, with non-compliant products prohibited from market placement and required removal from sale by May 1, 2027. This three-phase implementation creates reformulation demand favoring powder encapsulation technologies that stabilize retinoids against oxidative degradation while enabling precision dosing avoiding concentration limits triggering labeling requirements. Germany's cosmetics industry, documented through the German Cosmetic, Toiletry, Perfumery and Detergent Association (IKW) manufacturing statistics, leads European Union technical innovation in active ingredient delivery systems.
Commission Regulation 2024/858 added 12 nanomaterial ingredients to prohibited substances lists with compliance required by February 2025, including colloidal copper, silver, gold and platinum commonly used in luxury skincare formulations. Powder formats avoid nanomaterial incorporation through macroscopic mineral bases including kaolin, bentonite and silica microspheres that deliver oil-absorbing benefits without triggering nanomaterial labeling requirements mandated six months before products containing novel nanomaterials enter the European Union market. The September 1, 2025 ban on 21 CMR substances under CLP Regulation amendments requires toxicological reassessment across ingredient portfolios, favoring powder formulations with shorter ingredient lists focused on active benefits rather than complex emulsion systems requiring multiple preservatives, emulsifiers and stabilizers each subject to individual safety substantiation documentation.
Why is Brazil's ANVISA Framework Boosting 5.3% Expansion?
Brazil demonstrates 5.3% annual growth supported by Agência Nacional de Vigilância Sanitária (ANVISA) comprehensive regulatory consolidation. Resolution RDC 907/2024, introduced September 23, 2024, replaces and consolidates earlier regulations governing production and sale of cosmetics, personal hygiene products and perfumes, superseding RDC 752/2022, RDC 841/2023 and Article 13 of RDC 814/2023. This regulatory simplification reduces compliance complexity for powder formulation manufacturers navigating ingredient approval processes and labeling requirements across multiple product variants targeting different skin types and active ingredient combinations. Resolution RDC 949/2024 establishes cosmetic classification into Grade 1 and Grade 2 based on formulation and intended purpose, with notification processes streamlined compared to registration requirements applied to nine specific product groups including suntan products and hair treatments.
ANVISA's reinforced labeling requirements mandate clear and accurate information including ingredients, expiration dates and usage instructions, creating communication opportunities for powder formats to differentiate through reconstitution method education and single-use sachet freshness guarantees. Resolution RDC 894/2024 established cosmetovigilance system requirements for post-market safety surveillance within national territory, enabling enterprises to fulfill responsibilities regarding product safety and ensure timely measures when necessary. This monitoring infrastructure benefits powder formulations demonstrating lower adverse event rates through preservative-free anhydrous compositions eliminating sensitizers including parabens and isothiazolinones associated with contact dermatitis documented in Brazilian Society of Dermatology clinical case reports.
How does UK Post-Brexit Regulation Influence 4.3% Growth?
The United Kingdom market at 4.3% CAGR reflects post-Brexit regulatory independence creating divergence from European Union cosmetics frameworks while maintaining alignment on core safety principles. The Cosmetic Products Restriction of Chemical Substances Regulations 2024 imposed restrictions on Butylated Hydroxytoluene (BHT) taking effect 21 days after enactment, with products placed on market before February 24, 2025 permitted for continued sale until June 24, 2025, creating short-term inventory management complexity. The Cosmetic Products Restriction of Chemical Substances (No. 2) Regulations 2024, effective January 31, 2025, introduced a three-phase ban on 64 CMR substances and restrictions on Kojic acid use, limiting the ingredient to facial and hand products at maximum 1% concentration. These substance restrictions favor powder formulations utilizing alternative tyrosinase inhibitors including niacinamide and tranexamic acid that avoid Kojic acid concentration limits while delivering hyperpigmentation treatment benefits validated through clinical endpoint photography documented in peer-
What Strategic Moves Are Defining Competitive Landscape?
Competitive dynamics center on dermatologist validation partnerships that position powder formulations as clinical intervention tools rather than cosmetic enhancement products. L'Oréal Group's dermatological beauty division achieved 9.8% like-for-like growth in fiscal 2024, with brands including La Roche-Posay, CeraVe and SkinCeuticals leveraging professional recommendation channels to justify premium pricing for anhydrous formulations incorporating unstable actives including vitamin C derivatives and growth factors. The company's March 2024 launch of Melasyl, a breakthrough molecule addressing localized pigmentation issues, demonstrates the clinical development pathway that commands dermatologist endorsement through peer-reviewed efficacy data published in specialty journals rather than traditional marketing claims substantiation through consumer perception studies. This professional validation strategy creates barriers to entry for mass-market competitors lacking research infrastructure to generate clinical endpoint documentation required by sophisticated prescriber audiences.
Manufacturing strategies prioritize ISO 22716 certification demonstrating GMP compliance for cosmetics production, control, storage and shipment. The standard covers raw material intake through final delivery, helping organizations manage quality, reduce risks and align with global standards across the 47 jurisdictions requiring cosmetics manufacturing documentation. Certification validity periods of maximum three years with recertification before expiry create ongoing compliance costs absorbed through operational scale rather than per-product basis, favoring vertically integrated manufacturers over contract manufacturing arrangements lacking direct quality oversight.
Recent Developments:
- In January 2025, L'Oréal Group announced collaboration with IBM leveraging generative AI technology to uncover insights in cosmetic formulation data for sustainable raw materials and waste reductions.
- In January 2025, Lancôme showcased RÉNERGIE NANO-RESURFACER 400 BOOSTER at CES, featuring patented nanochip technology with 400 ultra-precise nano-tips enhancing topical penetration.
- In February 2025, Estée Lauder Companies announced leadership updates under CEO Stéphane de La Faverie supporting Beauty Reimagined vision.
Key Players in Powdered Facial Toners Market
- L'Oréal S.A.
- Estée Lauder Companies Inc.
- Unilever PLC
- Shiseido Company, Limited
- Kenvue Inc.
- Avon Products Inc.
- Johnson & Johnson Consumer Inc.
- Sephora USA Inc.
- Pierre Fabre Group
- Beiersdorf AG
Market Definition
The powdered facial toners market represents revenue generated from waterless toning formulations delivered in dry formats including loose powder, compressed tablets, micronized crystals and freeze-dried sachets designed for reconstitution with water or direct application to damp facial skin. The market measures the value of products sold to end consumers through retail channels including specialty beauty stores, online e-commerce platforms, dermatology clinics, spa facilities and duty-free airport retailers.
Market Inclusion
Inclusions cover products formulated for astringent functions, pH balancing, oil control, pore minimization, exfoliation and active ingredient delivery following facial cleansing and preceding serum or moisturizer application in multi-step skincare protocols. It encompasses powder bases including kaolin clay, bentonite, silica microspheres, rice powder and tapioca starch combined with active ingredients such as niacinamide, vitamin C derivatives, alpha hydroxy acids, beta hydroxy acids, peptides and botanical extracts. Packaging formats including single-use sachets, refillable jars, compressed tablets and applicator-equipped compacts are included. Products marketed for all skin types including oily, dry, combination, sensitive and acne-prone dermotypes are covered.
Market Exclusion
Exclusions include liquid toner products regardless of active ingredient composition, micellar water cleansing products, setting powders intended for makeup application rather than skincare treatment, body powders not formulated for facial application, and baby powder products positioned for infant care rather than adult facial skincare regimens. Standard bath salts and effervescent tablet formats intended for whole-body bathing applications are outside the scope.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Format & Delivery Segments | Loose Powder Toners; Compressed Tablets; Sachet-Based Single-Dose Powders; Micronized Crystals; Freeze-Dried Powder Systems |
| Skin Type / Functional Segments | Oily & Acne-Prone Skin Control; Dry Skin Hydration Support; Combination Skin Balancing; Sensitive Skin Barrier Maintenance; Brightening & Exfoliation Formulations |
| Active Ingredient Categories | Niacinamide; Vitamin C Derivatives; Alpha Hydroxy Acids; Beta Hydroxy Acids; Peptides; Botanical Extracts |
| Distribution Channels | Online Stores; Specialty Beauty Retailers; Dermatology Clinics; Exclusive Brand Agencies; Supermarkets; Others |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia & Pacific, East Asia, Middle East & Africa |
| Key Countries | China, India, Germany, Brazil, United States, United Kingdom |
| Key Companies Profiled | L’Oréal S.A.; Estée Lauder Companies Inc.; Unilever PLC; Shiseido Company, Limited; Kenvue Inc.; Avon Products Inc.; Johnson & Johnson Consumer Inc.; Sephora USA Inc.; Pierre Fabre Group; Beiersdorf AG |
| Additional Attributes | Dollar sales measured for powdered facial toners by anhydrous format type, skin-function positioning, active ingredient system, distribution channel performance, regulatory-driven reformulation impact, compliance alignment with cosmetics safety and labeling standards, clinical substantiation trends, and country-level regulatory modernization effects on market access |
Powdered Facial Toners Market by Segments
By Skin Type:
- Oily Skin
- Dry Skin
- Combination Skin
- Sensitive Skin
- Acne-Prone Skin
By Format:
- Loose Powder
- Compressed Tablets
- Sachets
- Micronized Crystals
- Freeze-Dried Powder
By Distribution Channel:
- Online Store
- Exclusive Agency
- Supermarket
- Specialty Beauty Retailers
- Dermatology Clinics
- Others
By Active Ingredient:
- Niacinamide
- Vitamin C Derivatives
- Alpha Hydroxy Acids
- Beta Hydroxy Acids
- Peptides
- Botanical Extracts
By Region:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- National Medical Products Administration (NMPA). (2025, November). Comprehensive Cosmetics Regulation Reforms. China Government.
- Central Drug Standard Control Organization (CDSCO). (2025, January). Cosmetics Amendment Rules 2025. India Ministry of Health and Family Welfare.
- The Estée Lauder Companies Inc. (2024, August). Fiscal 2024 Results. Estée Lauder Investor Relations.
- Bureau of Indian Standards (BIS). (2004). IS 6608 Skin Creams Specification. BIS Publications.
- Cosmetic Toiletry and Perfumery Association (CTPA). (2024). UK Cosmetics Regulations Amendments. CTPA Guidance.
Frequently Asked Questions
What is the current global market size for Powdered Facial Toners?
The powdered facial toners market is valued at USD 15.0 billion in 2026, driven by rapid adoption of anhydrous, preservative-free skincare formats.
What is the projected CAGR for the Powdered Facial Toners market?
The market is projected to grow at a 5.0% CAGR from 2026 to 2036.
Which segment currently dominates the market?
Oily skin formulations lead with a 56.8% share while online stores account for 39.4% of total sales.
What are the primary drivers of market growth?
Growth is driven by regulatory pressure on preservatives, PFAS restrictions, and superior stability of powder-based skincare actives.
Who are the leading players in the Powdered Facial Toners market?
Key companies include L’Oréal S.A., Estée Lauder Companies Inc., Unilever PLC, Shiseido Company Limited, and Beiersdorf AG.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Application, 2026 to 2036
- Oily Skin
- Dry Skin
- Combination Skin
- Sensitive Skin
- Normal Skin
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Types
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Types, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Types, 2026 to 2036
- Hydrating Toner Powders
- Exfoliating Toner Powders
- Brightening Toner Powders
- Mattifying Toner Powders
- Acne-Fighting Toner Powders
- Y-o-Y Growth Trend Analysis By Types, 2021 to 2025
- Absolute $ Opportunity Analysis By Types, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Online Store
- Exclusive Agency
- Supermarket
- Others
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- By Application
- By Types
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- By Distribution Channel
- Key Takeaways
- Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- France
- Rest of Europe
- By Application
- By Types
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- By Distribution Channel
- Key Takeaways
- Asia Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- India
- Japan
- South Korea
- Rest of Asia Pacific
- By Application
- By Types
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Rest of Latin America
- By Application
- By Types
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- South Africa
- Rest of Middle East & Africa
- By Application
- By Types
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Types
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- L'Oréal
- BDF (Beiersdorf)
- Avon
- Unilever
- J&J (Johnson & Johnson)
- Shiseido
- Pierre Fabre
- Estée Lauder
- Sephora
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Powdered Facial Toners Market Key Takeaways
- Global Powdered Facial Toners Market Size (USD Billion), 2021-2036
- Global Powdered Facial Toners Market Y-o-Y Growth (%), 2021-2036
- Global Powdered Facial Toners Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Global Powdered Facial Toners Market Pricing Analysis (USD/Unit), 2021-2036
- Global Powdered Facial Toners Market Size (USD Billion) By Application, 2021-2036
- Global Powdered Facial Toners Market Size (USD Billion) By Types, 2021-2036
- Global Powdered Facial Toners Market Size (USD Billion) By Distribution Channel, 2021-2036
- Global Powdered Facial Toners Market Size (USD Billion) By Region, 2021-2036
- North America Powdered Facial Toners Market Size (USD Billion) By Country, 2021-2036
- North America Powdered Facial Toners Market Size (USD Billion) By Application, 2021-2036
- North America Powdered Facial Toners Market Size (USD Billion) By Types, 2021-2036
- North America Powdered Facial Toners Market Size (USD Billion) By Distribution Channel, 2021-2036
- North America Powdered Facial Toners Market Attractiveness Analysis By Country
- North America Powdered Facial Toners Market Attractiveness Analysis By Application
- North America Powdered Facial Toners Market Attractiveness Analysis By Types
- North America Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- Europe Powdered Facial Toners Market Size (USD Billion) By Country, 2021-2036
- Europe Powdered Facial Toners Market Size (USD Billion) By Application, 2021-2036
- Europe Powdered Facial Toners Market Size (USD Billion) By Types, 2021-2036
- Europe Powdered Facial Toners Market Size (USD Billion) By Distribution Channel, 2021-2036
- Europe Powdered Facial Toners Market Attractiveness Analysis By Country
- Europe Powdered Facial Toners Market Attractiveness Analysis By Application
- Europe Powdered Facial Toners Market Attractiveness Analysis By Types
- Europe Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- Asia Pacific Powdered Facial Toners Market Size (USD Billion) By Country, 2021-2036
- Asia Pacific Powdered Facial Toners Market Size (USD Billion) By Application, 2021-2036
- Asia Pacific Powdered Facial Toners Market Size (USD Billion) By Types, 2021-2036
- Asia Pacific Powdered Facial Toners Market Size (USD Billion) By Distribution Channel, 2021-2036
- Asia Pacific Powdered Facial Toners Market Attractiveness Analysis By Country
- Asia Pacific Powdered Facial Toners Market Attractiveness Analysis By Application
- Asia Pacific Powdered Facial Toners Market Attractiveness Analysis By Types
- Asia Pacific Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- Latin America Powdered Facial Toners Market Size (USD Billion) By Country, 2021-2036
- Latin America Powdered Facial Toners Market Size (USD Billion) By Application, 2021-2036
- Latin America Powdered Facial Toners Market Size (USD Billion) By Types, 2021-2036
- Latin America Powdered Facial Toners Market Size (USD Billion) By Distribution Channel, 2021-2036
- Latin America Powdered Facial Toners Market Attractiveness Analysis By Country
- Latin America Powdered Facial Toners Market Attractiveness Analysis By Application
- Latin America Powdered Facial Toners Market Attractiveness Analysis By Types
- Latin America Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Powdered Facial Toners Market Size (USD Billion) By Country, 2021-2036
- Middle East & Africa Powdered Facial Toners Market Size (USD Billion) By Application, 2021-2036
- Middle East & Africa Powdered Facial Toners Market Size (USD Billion) By Types, 2021-2036
- Middle East & Africa Powdered Facial Toners Market Size (USD Billion) By Distribution Channel, 2021-2036
- Middle East & Africa Powdered Facial Toners Market Attractiveness Analysis By Country
- Middle East & Africa Powdered Facial Toners Market Attractiveness Analysis By Application
- Middle East & Africa Powdered Facial Toners Market Attractiveness Analysis By Types
- Middle East & Africa Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- USA Powdered Facial Toners Pricing Analysis (USD/Unit), 2021-2036
- USA Powdered Facial Toners Market Share Analysis (%) By Application, 2025
- USA Powdered Facial Toners Market Share Analysis (%) By Types, 2025
- USA Powdered Facial Toners Market Share Analysis (%) By Distribution Channel, 2025
- China Powdered Facial Toners Pricing Analysis (USD/Unit), 2021-2036
- China Powdered Facial Toners Market Share Analysis (%) By Application, 2025
- China Powdered Facial Toners Market Share Analysis (%) By Types, 2025
- China Powdered Facial Toners Market Share Analysis (%) By Distribution Channel, 2025
- India Powdered Facial Toners Pricing Analysis (USD/Unit), 2021-2036
- India Powdered Facial Toners Market Share Analysis (%) By Application, 2025
- India Powdered Facial Toners Market Share Analysis (%) By Types, 2025
- India Powdered Facial Toners Market Share Analysis (%) By Distribution Channel, 2025
- Germany Powdered Facial Toners Pricing Analysis (USD/Unit), 2021-2036
- Germany Powdered Facial Toners Market Share Analysis (%) By Application, 2025
- Germany Powdered Facial Toners Market Share Analysis (%) By Types, 2025
- Germany Powdered Facial Toners Market Share Analysis (%) By Distribution Channel, 2025
- Brazil Powdered Facial Toners Pricing Analysis (USD/Unit), 2021-2036
- Brazil Powdered Facial Toners Market Share Analysis (%) By Application, 2025
- Brazil Powdered Facial Toners Market Share Analysis (%) By Types, 2025
- Brazil Powdered Facial Toners Market Share Analysis (%) By Distribution Channel, 2025
- UK Powdered Facial Toners Pricing Analysis (USD/Unit), 2021-2036
- UK Powdered Facial Toners Market Share Analysis (%) By Application, 2025
- UK Powdered Facial Toners Market Share Analysis (%) By Types, 2025
- UK Powdered Facial Toners Market Share Analysis (%) By Distribution Channel, 2025
- Powdered Facial Toners Market Competition Dashboard
- Powdered Facial Toners Market Competition Benchmarking Analysis
- Powdered Facial Toners Market Share Analysis of Top Players By Regional, 2025
- Powdered Facial Toners Market Share Analysis of Top Players By Application, 2025
- Powdered Facial Toners Market Share Analysis of Top Players By Types, 2025
- Powdered Facial Toners Market Share Analysis of Top Players By Distribution Channel, 2025
- L'Oréal - Company Profile
- BDF (Beiersdorf) - Company Profile
- Avon - Company Profile
- Unilever - Company Profile
- J&J (Johnson & Johnson) - Company Profile
- Shiseido - Company Profile
- Pierre Fabre - Company Profile
- Estée Lauder - Company Profile
- Sephora - Company Profile
- Assumptions & Acronyms Used in Powdered Facial Toners Market Report
List of Figures
- Powdered Facial Toners Market Size (USD Billion), 2021-2036
- Powdered Facial Toners Market Y-o-Y Growth (%), 2021-2036
- Powdered Facial Toners Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Powdered Facial Toners Market Value Chain Analysis
- Powdered Facial Toners Market Supply Chain Analysis
- Powdered Facial Toners Market Investment Feasibility Matrix
- Powdered Facial Toners Market PESTLE Analysis
- Powdered Facial Toners Market Porter's Five Forces Analysis
- Powdered Facial Toners Market Product Life Cycle Analysis
- Powdered Facial Toners Market Opportunity Map Analysis
- Powdered Facial Toners Market Scenario Forecast Analysis
- Powdered Facial Toners Market Production and Consumption Statistics
- Powdered Facial Toners Market Import and Export Statistics
- Global Powdered Facial Toners Market Share (%) By Application, 2025
- Global Powdered Facial Toners Market Share (%) By Application, 2036
- Global Powdered Facial Toners Market Share (%) By Types, 2025
- Global Powdered Facial Toners Market Share (%) By Types, 2036
- Global Powdered Facial Toners Market Share (%) By Distribution Channel, 2025
- Global Powdered Facial Toners Market Share (%) By Distribution Channel, 2036
- Global Powdered Facial Toners Market Share (%) By Region, 2025
- Global Powdered Facial Toners Market Share (%) By Region, 2036
- Global Powdered Facial Toners Market Attractiveness Analysis By Region
- North America Powdered Facial Toners Market Size (USD Billion), 2021-2036
- North America Powdered Facial Toners Market Share (%) By Country, 2025
- North America Powdered Facial Toners Market Share (%) By Country, 2036
- North America Powdered Facial Toners Market Share (%) By Application, 2025
- North America Powdered Facial Toners Market Share (%) By Application, 2036
- North America Powdered Facial Toners Market Share (%) By Types, 2025
- North America Powdered Facial Toners Market Share (%) By Types, 2036
- North America Powdered Facial Toners Market Share (%) By Distribution Channel, 2025
- North America Powdered Facial Toners Market Share (%) By Distribution Channel, 2036
- North America Powdered Facial Toners Market Attractiveness Analysis By Country
- North America Powdered Facial Toners Market Attractiveness Analysis By Application
- North America Powdered Facial Toners Market Attractiveness Analysis By Types
- North America Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- Europe Powdered Facial Toners Market Size (USD Billion), 2021-2036
- Europe Powdered Facial Toners Market Share (%) By Country, 2025
- Europe Powdered Facial Toners Market Share (%) By Country, 2036
- Europe Powdered Facial Toners Market Share (%) By Application, 2025
- Europe Powdered Facial Toners Market Share (%) By Application, 2036
- Europe Powdered Facial Toners Market Share (%) By Types, 2025
- Europe Powdered Facial Toners Market Share (%) By Types, 2036
- Europe Powdered Facial Toners Market Share (%) By Distribution Channel, 2025
- Europe Powdered Facial Toners Market Share (%) By Distribution Channel, 2036
- Europe Powdered Facial Toners Market Attractiveness Analysis By Country
- Europe Powdered Facial Toners Market Attractiveness Analysis By Application
- Europe Powdered Facial Toners Market Attractiveness Analysis By Types
- Europe Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- Asia Pacific Powdered Facial Toners Market Size (USD Billion), 2021-2036
- Asia Pacific Powdered Facial Toners Market Share (%) By Country, 2025
- Asia Pacific Powdered Facial Toners Market Share (%) By Country, 2036
- Asia Pacific Powdered Facial Toners Market Share (%) By Application, 2025
- Asia Pacific Powdered Facial Toners Market Share (%) By Application, 2036
- Asia Pacific Powdered Facial Toners Market Share (%) By Types, 2025
- Asia Pacific Powdered Facial Toners Market Share (%) By Types, 2036
- Asia Pacific Powdered Facial Toners Market Share (%) By Distribution Channel, 2025
- Asia Pacific Powdered Facial Toners Market Share (%) By Distribution Channel, 2036
- Asia Pacific Powdered Facial Toners Market Attractiveness Analysis By Country
- Asia Pacific Powdered Facial Toners Market Attractiveness Analysis By Application
- Asia Pacific Powdered Facial Toners Market Attractiveness Analysis By Types
- Asia Pacific Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- Latin America Powdered Facial Toners Market Size (USD Billion), 2021-2036
- Latin America Powdered Facial Toners Market Share (%) By Country, 2025
- Latin America Powdered Facial Toners Market Share (%) By Country, 2036
- Latin America Powdered Facial Toners Market Share (%) By Application, 2025
- Latin America Powdered Facial Toners Market Share (%) By Application, 2036
- Latin America Powdered Facial Toners Market Share (%) By Types, 2025
- Latin America Powdered Facial Toners Market Share (%) By Types, 2036
- Latin America Powdered Facial Toners Market Share (%) By Distribution Channel, 2025
- Latin America Powdered Facial Toners Market Share (%) By Distribution Channel, 2036
- Latin America Powdered Facial Toners Market Attractiveness Analysis By Country
- Latin America Powdered Facial Toners Market Attractiveness Analysis By Application
- Latin America Powdered Facial Toners Market Attractiveness Analysis By Types
- Latin America Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Powdered Facial Toners Market Size (USD Billion), 2021-2036
- Middle East & Africa Powdered Facial Toners Market Share (%) By Country, 2025
- Middle East & Africa Powdered Facial Toners Market Share (%) By Country, 2036
- Middle East & Africa Powdered Facial Toners Market Share (%) By Application, 2025
- Middle East & Africa Powdered Facial Toners Market Share (%) By Application, 2036
- Middle East & Africa Powdered Facial Toners Market Share (%) By Types, 2025
- Middle East & Africa Powdered Facial Toners Market Share (%) By Types, 2036
- Middle East & Africa Powdered Facial Toners Market Share (%) By Distribution Channel, 2025
- Middle East & Africa Powdered Facial Toners Market Share (%) By Distribution Channel, 2036
- Middle East & Africa Powdered Facial Toners Market Attractiveness Analysis By Country
- Middle East & Africa Powdered Facial Toners Market Attractiveness Analysis By Application
- Middle East & Africa Powdered Facial Toners Market Attractiveness Analysis By Types
- Middle East & Africa Powdered Facial Toners Market Attractiveness Analysis By Distribution Channel
- USA Powdered Facial Toners Pricing Trend Analysis (USD/Unit), 2021-2036
- China Powdered Facial Toners Pricing Trend Analysis (USD/Unit), 2021-2036
- India Powdered Facial Toners Pricing Trend Analysis (USD/Unit), 2021-2036
- Germany Powdered Facial Toners Pricing Trend Analysis (USD/Unit), 2021-2036
- Brazil Powdered Facial Toners Pricing Trend Analysis (USD/Unit), 2021-2036
- UK Powdered Facial Toners Pricing Trend Analysis (USD/Unit), 2021-2036
- Powdered Facial Toners Market Competitive Landscape
- Powdered Facial Toners Market Structure Analysis
- Powdered Facial Toners Market Competition Dashboard
- Powdered Facial Toners Market Competition Benchmarking Analysis
- Powdered Facial Toners Market Share Analysis of Top Players By Regional
- Powdered Facial Toners Market Share Analysis of Top Players By Application
- Powdered Facial Toners Market Share Analysis of Top Players By Types
- Powdered Facial Toners Market Share Analysis of Top Players By Distribution Channel
- L'Oréal - Revenue Analysis (USD Million), 2019-2025
- BDF (Beiersdorf) - Revenue Analysis (USD Million), 2019-2025
- Avon - Revenue Analysis (USD Million), 2019-2025
- Unilever - Revenue Analysis (USD Million), 2019-2025
- J&J (Johnson & Johnson) - Revenue Analysis (USD Million), 2019-2025
- L'Oréal - Market Position Analysis
- BDF (Beiersdorf) - Market Position Analysis
- Avon - Market Position Analysis
- Unilever - Market Position Analysis
- J&J (Johnson & Johnson) - Market Position Analysis

