The tamper-evident pharma serialization labels market crossed a valuation of USD 4800.0 million in 2025. Industry is poised to cross USD 5068.8 million in 2026 at a CAGR of 5.60% during the forecast. Demand outlook carries the market valuation to USD 8740.7 million through 2036 as precision-timing economics reach mid-market discrete manufacturing tiers that previously deferred upgrades and regulatory bodies mandate strict unit-level physical authentication frameworks globally.
The transition from batch-level tracking to item-level authentication forces contract manufacturing organizations to overhaul their secondary packaging lines. Facilities delaying this integration lose access to high-value international supply contracts that legally require multi-layered supply chain visibility. This architectural pivot embeds permanent security requirements into everyday drug production, ensuring consistent procurement volumes regardless of broader economic conditions.
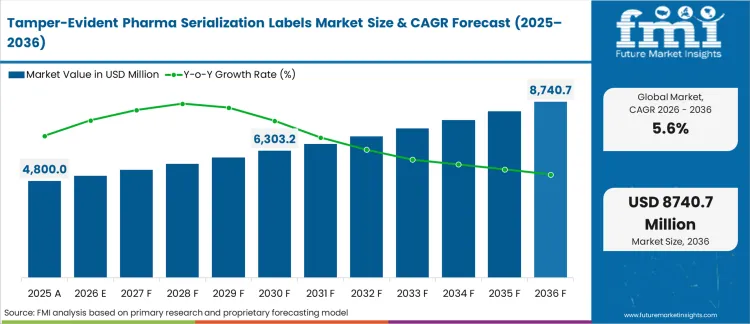
The inflection point for this industry materializes when contract manufacturing organizations abandon bolt-on compliance processes and fully integrate digital-physical authentication into native packaging workflows. Facility directors overseeing production scaling must validate cold-cure adhesives against new high-speed serialization machinery before critical commercial export windows close. Operations mastering this specific environmental resilience secure the most lucrative, high-margin biologic supply contracts across the forecast decade.
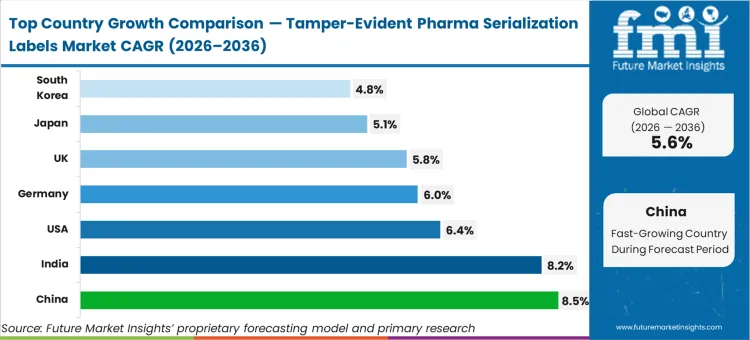
China advances at an 8.5% compound rate to 2036 as local manufacturers scale production to meet stringent global export standards. India records an 8.2% expansion, driven by immense generic drug manufacturing volumes requiring verified physical security. The United States grows at 6.4% as federal supply chain security acts reach full enforcement maturity. Germany tracks a 6.0% trajectory while the UK expands at 5.8%, reflecting established compliance ecosystems. Japan follows with a 5.1% rate, and South Korea posts a 4.8% growth profile. This geographic dispersion remains structural as diverse national health ministries independently enact parallel serialization laws.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 5068.8 million |
| Industry Value (2036) | USD 8740.7 million |
| CAGR (2026-2036) | 5.60% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Tamper-evident pharma serialization labels represent specialized physical markers applied to drug packaging that provide irreversible visual proof of interference while housing unique digital identification codes. These products fuse structural security mechanisms with variable data printing to guarantee both the authenticity and the physical integrity of medical products across the supply chain.
The market scope encompasses destructible polymer films, void-reveal adhesives, integrated RFID tags, and serialized seals designed specifically for medical containers. Components enabling secure pharmaceutical packaging solutions directly at the primary and secondary carton levels fall within the defined boundaries. Hardware-encoded NFC inlays and temperature-sensitive indicator tags linked to unique item-level identifiers are fully incorporated.
Standard commercial barcode stickers lacking physical tamper-evidence mechanisms are excluded. Purely decorative secondary packaging, unprinted sealing tapes, and software systems managing track-and-trace databases fall outside the valuation. General logistics labels not directly fulfilling regulatory pharmaceutical authentication mandates are explicitly omitted from the analysis.
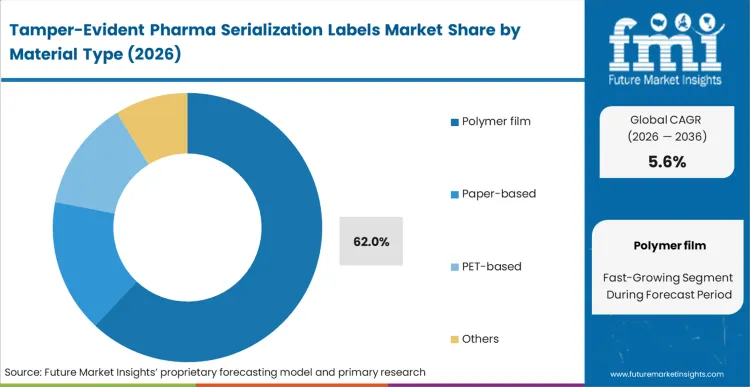
Polymer film commands a massive 62.0% share in 2026, establishing the performance baseline for modern biological supply chains. Cold-chain logistics directors transporting high-value immunotherapies require substrates that resist moisture degradation and cryogenic shattering. Based on FMI's assessment, traditional paper labels fail catastrophically under these thermal stresses, forcing immediate adoption of specialized security labels engineered from synthetic compounds. Procurement specialists evaluating packaging integrity reject materials that cannot maintain absolute physical adhesion across the entire transit lifecycle.
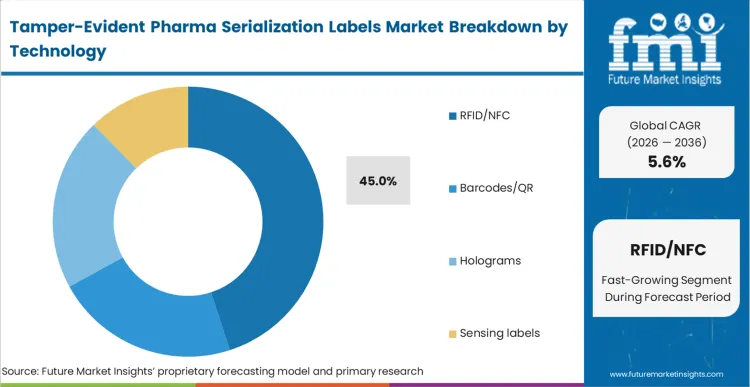
Legacy printed formats prove increasingly vulnerable to sophisticated replication, driving an organized transition toward secure digital alternatives. RFID/NFC platforms capture 45.0% share in 2026 as automated inventory validation becomes mandatory for high-throughput distribution centers. Supply chain architects implementing these technologies dramatically reduce manual scanning bottlenecks during bulk receiving operations. FMI analysts opine that integrating RFID and NFC tracking tags directly into the tamper-seal accelerates processing timelines exponentially. Pharmacy directors implementing touchless receiving protocols avoid severe labor shortages while securing critical medication.
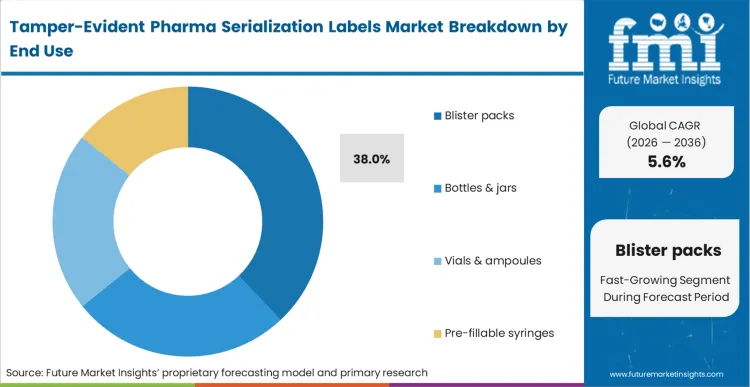
The requirement for strict unit-dose integrity has firmly established specialized packaging constraints as a critical security perimeter. Blister packs represent 38.0% share in 2026, driven by the absolute necessity of protecting individual capsules from moisture and contamination until the moment of consumption. Applying specialized void-reveal seals across these multi-cavity formats prevents undetectable pilferage in institutional healthcare settings. In FMI's view, incorporating blister pack arrays with advanced serialized lidding foil represents the ultimate standard for oral solid dose protection. Retail pharmacy operators demanding irrefutable proof of safety reject shipments lacking these integrated secondary barriers.
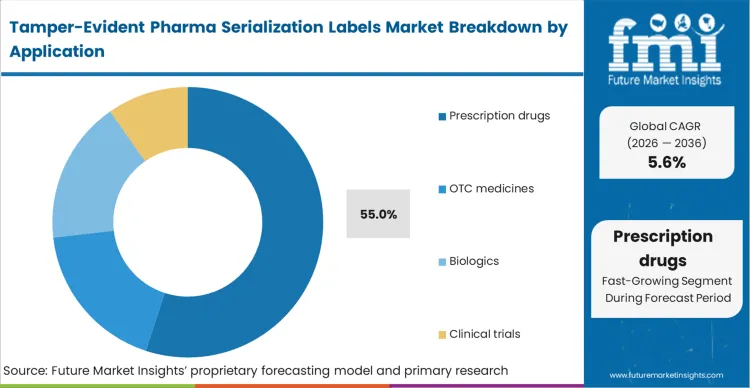
Every prescription drug manufacturer currently faces an escalating matrix of mandatory digital tracking laws to retain their distribution licenses. Prescription drugs secure 55.0% share in 2026 because compliance with national verification systems is a non-negotiable prerequisite for market access. High-value specialized therapeutics uniquely attract sophisticated counterfeit networks, necessitating advanced layered security utilizing biologics containment formats. FMI's research confirms that integrating physical destruction markers with serialized databases creates an impenetrable closed-loop authentication environment. Chief compliance officers failing to implement these synchronized safeguards trigger immediate commercial blockades from federal health agencies.
Global legislative mandates requiring complete unit-level tracking fundamentally reshape the commercial packaging environment. Pharmaceutical brand owners facing complex regulatory frameworks must deploy unique identifiers secured by physical tamper-evidence to prevent product diversion. This regulatory trigger forces packaging architects to abandon generic cartons in favor of highly specialized anti-counterfeit technologies. Manufacturers who delay this integration lose the legal right to distribute their medical products in major economies, ensuring continuous label procurement regardless of economic downturns.
Applying intricate void-reveal labels at high speeds introduces significant mechanical friction into modern production lines. Quality control directors utilizing advanced machinery face severe downtime when complex adhesives fail to cure instantly under rapid application, exposing the limits of standard barcode identification stickers. This operational gap limits throughput and elevates manufacturing costs significantly. Facility managers must constantly calibrate equipment tension and heat settings, restricting overall production velocity to accommodate delicate security materials.
Opportunities in the Tamper-Evident Pharma Serialization Labels Market
Based on the regional analysis, the Tamper-Evident Pharma Serialization Labels market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 8.5% |
| India | 8.2% |
| USA | 6.4% |
| Germany | 6.0% |
| UK | 5.8% |
| Japan | 5.1% |
| South Korea | 4.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
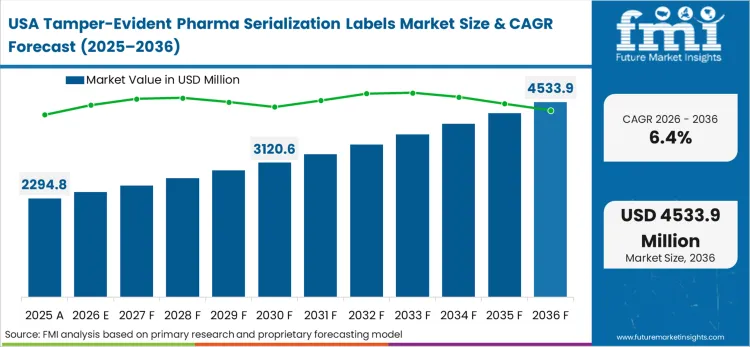
Industrial modernization mandates across North America target the systematic eradication of isolated and vulnerable operational distribution networks. Supply chain architects redesigning legacy distribution frameworks must integrate interoperable digital identifiers protected by overt physical security mechanisms. The concentration of high-value biologic and specialty drug development creates an unparalleled demand for premium, multi-functional label constructions. FMI analysts observe that this environment heavily rewards substrate converters capable of delivering extreme reliability and seamless integration with track and trace database networks.
FMI's report includes comprehensive evaluation of the Canadian and Mexican pharmaceutical packaging sectors. The integration of cross-border manufacturing requires standardized security protocols to coordinate complex regional distribution networks efficiently.
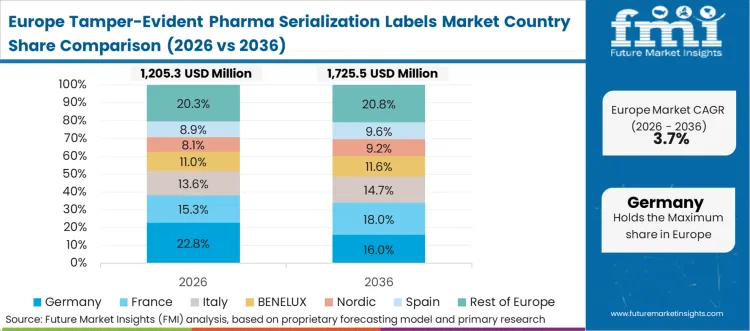
European industrial policy actively dictates the exact structural parameters of secondary packaging security across the entire continent. Pharmaceutical directors face continuous compliance audits verifying the presence and functionality of tamper-evident seals on every prescribed medication. This unified regulatory requirement forces packaging manufacturers to adopt traffic-shaping protocols guaranteeing control stability while utilizing smart label platforms. High consumer awareness regarding medication authenticity amplifies the necessity for flawless physical implementation.
FMI's report includes thorough investigation of the broader European label manufacturing ecosystem. The mandatory shift toward circular economy principles forces suppliers to invent security markers that do not disrupt the recycling streams of glass and plastic containers.
The aggressive expansion of generic pharmaceutical manufacturing capabilities defines this region's structural demand for advanced packaging security and verification frameworks. National export initiatives force local facilities to preemptively adopt the strictest international serialization standards to ensure global market access is never compromised. The necessity of meeting Western compliance frameworks accelerates the complete replacement of basic paper labels with advanced serialized holographic seals. This dynamic creates a massive, immediate requirement for scalable, high-speed security application technologies across the region.
FMI's report includes extensive analysis of the emerging Southeast Asian pharmaceutical hubs. Increasing cross-border regulatory harmonization drives coordinated adoption of serialized security formats across ASEAN nations.
The global pharmaceutical label market remains moderately consolidated among highly specialized multinational material science corporations commanding vast global networks. Supplying global pharmaceutical brands requires massive capital investment in specialized cleanroom manufacturing, strict ISO certifications, and multi-continent distribution capabilities. Companies like Avery Dennison Corporation, CCL Industries Inc., and 3M Company dominate by offering universally compliant material portfolios scaled for international deployment. Procurement teams utilize global supply reliability and demonstrated adherence to strict low-migration ink standards as the primary variables to distinguish qualified from unqualified label converters.
Leading players possess an overwhelming structural advantage through their proprietary adhesive formulations and deep integration with global medical device tracking labels hardware providers. Building a portfolio of FDA-compliant adhesives that cure instantly without bleeding into the packaging substrate requires decades of specialized chemical engineering. Deploying advanced labels tailored for prefilled syringe configurations guarantees seamless integration with automated high-speed application machinery. Challengers must invest heavily in specialized polymer research to replicate this performance, severely limiting successful market entry to exceptionally well-capitalized material science firms.
Large pharmaceutical buyers aggressively prevent vendor lock-in by rigorously qualifying multiple label suppliers for every major global drug program. Procurement teams demand open-architecture security designs that can be printed and applied using standard industry hardware across diverse global sites. This multi-vendor qualification strategy structurally limits the pricing power of dominant label manufacturers, despite the inherently high barriers to entry in developing temperature-controlled shipping formats. Top-tier converters will increasingly acquire specialized technology startups to integrate RFID and digital authentication features, shifting the competitive trajectory toward complete smart-packaging ecosystems over the forecast period.

| Metric | Value |
|---|---|
| Quantitative Units | USD 5068.8 million to USD 8740.7 million, at a CAGR of 5.60% |
| Market Definition | Tamper-evident pharma serialization labels are security markers that combine unique digital identifiers with physical destruction mechanisms to secure drug integrity. |
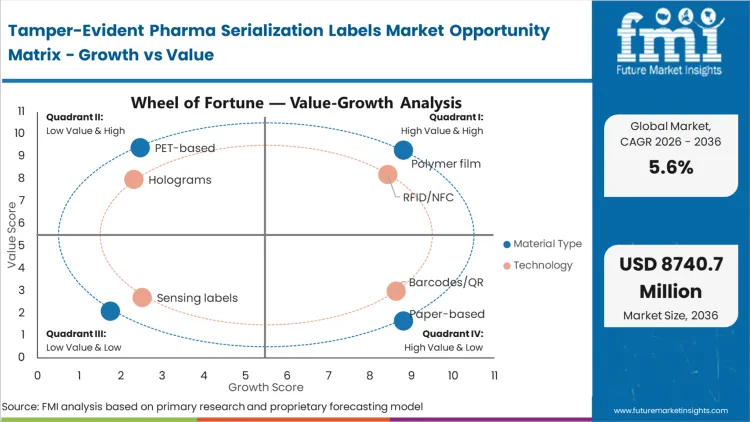
| Material Type Segmentation | Polymer film, Paper-based, PET-based, Others |
| Technology Segmentation | RFID/NFC, Barcodes/QR, Holograms, Sensing labels |
| End-use Format Segmentation | Blister packs, Bottles & jars, Vials & ampoules, Pre-fillable syringes |
| Application Segmentation | Prescription drugs, OTC medicines, Biologics, Clinical trials |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, USA, Germany, UK, Japan, South Korea, and 40 plus countries |
| Key Companies Profiled | Avery Dennison Corporation, 3M Company, CCL Industries Inc., UPM Raflatac, Schreiner Group, Brady Corporation, Flexcon Company, Inc., Essentra plc |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews with packaging qualification engineers anchor the research. The baseline value derives from a bottom-up aggregation of specialized medical label shipments, projecting adoption against regulatory enforcement dates. Segment forecasts undergo cross-validation against quarterly procurement guidance. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the demand for Tamper-Evident Pharma Serialization Labels in the global market in 2026?
Demand for Tamper-Evident Pharma Serialization Labels in the global market is estimated to be valued at USD 5068.8 million in 2026.
What will be the market size of Tamper-Evident Pharma Serialization Labels in the global market by 2036?
Market size for Tamper-Evident Pharma Serialization Labels is projected to reach USD 8740.7 million by 2036.
What is the expected demand growth for Tamper-Evident Pharma Serialization Labels in the global market between 2026 and 2036?
Demand for Tamper-Evident Pharma Serialization Labels is expected to grow at a CAGR of 5.60% between 2026 and 2036.
Which Material Type is poised to lead global sales by 2026?
Polymer film accounts for 62.0% in 2026 as cold-chain logistics directors require substrates that resist moisture degradation and cryogenic shattering.
How significant is the role of RFID/NFC in driving Tamper-Evident Pharma Serialization Labels adoption in 2026?
RFID/NFC represents 45.0% of segment share as high-throughput distribution centers shift toward automated and touchless inventory verification.
What is driving demand in China?
China's contract manufacturing sector utilizes highly automated production lines demanding robust and tear-resistant security markers to satisfy stringent export compliance requirements.
What compliance standards or regulations are referenced for Germany?
Germany's pharmaceutical packaging environment is shaped by strict compliance audits and recurring quality control requirements for high-speed application of serialized void seals.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 8.5% during 2026 to 2036.
Why is North America described as a priority region in this report?
The North American pharmaceutical sector prioritizes secure and interoperable item-level tracking systems to strengthen biologic supply chains and eliminate distribution vulnerabilities.
What type of demand dominates in North America?
Demand heavily focuses on premium multifunctional label constructions that combine overt physical tamper evidence with seamless track-and-trace integration.
What is India's growth outlook in this report?
India is projected to expand at a CAGR of 8.2% during 2026 to 2036.
Does the report cover USA in its regional analysis?
Yes, USA is included within North America under the regional scope of analysis.
What are the sources referred to for analyzing USA?
Federal supply chain security acts, public compliance programs, and pharmaceutical packaging modernization frameworks form the analytical basis.
What is the main demand theme linked to USA in its region coverage?
Strict unit-level traceability under the Drug Supply Chain Security Act forces the deployment of converged digital and physical security architectures.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe under the regional coverage framework.
What is the main Germany related demand theme in its region coverage?
Perfect high-speed application of serialized void seals on highly confined packaging surfaces defines the central demand theme in Germany.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Serialized holographic seals, tear-resistant security markers, and high-speed track-and-trace label application technologies are strategically important for Asia Pacific supply chains.
What is Tamper-Evident Pharma Serialization Labels and what is it mainly used for?
Tamper-Evident Pharma Serialization Labels are specialized security markers that combine irreversible physical tamper evidence with unique digital identifiers and are mainly used to secure drug authenticity and integrity across the supply chain.
What does Tamper-Evident Pharma Serialization Labels mean in this report?
The scope encompasses physical security labels that fuse structural tamper mechanisms with variable data printing to enable item-level pharmaceutical authentication.
What is included in the scope of this Tamper-Evident Pharma Serialization Labels report?
The market covers destructible polymer films, void-reveal adhesives, integrated RFID tags, serialized seals, hardware-encoded NFC inlays, and temperature-sensitive indicator tags designed for pharmaceutical packaging.
What is excluded from the scope of this report?
Standard commercial barcode stickers without tamper-evidence, decorative secondary packaging, unprinted sealing tapes, software track-and-trace systems, and general logistics labels are explicitly excluded.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on regulatory enforcement timelines, packaging modernization curves, and adoption of advanced pharmaceutical authentication systems for strategic planning purposes.
How does FMI build and validate the Tamper-Evident Pharma Serialization Labels forecast?
The model applies a bottom-up methodology starting with specialized medical label shipments and installed compliance hardware metrics, then cross-validates projections against capital expenditure guidance and quarterly procurement data.
What does zero reliance on speculative third-party market research mean here?
Primary interviews, public regulatory documentation, anti-counterfeiting roadmaps, and verified shipment and procurement data are used instead of unverified syndicated estimates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.