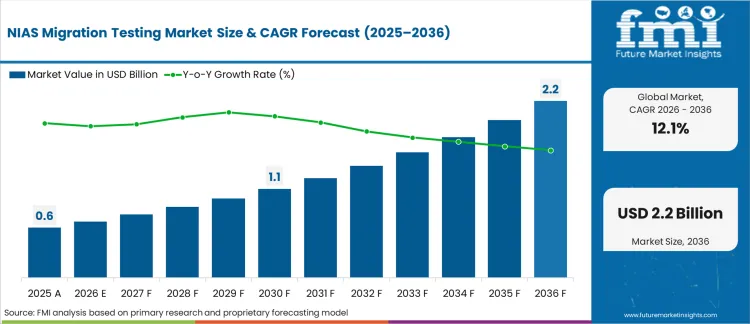
The NIAS migration testing market was valued at USD 0.6 billion in 2025. Industry is poised to cross USD 0.7 billion in 2026 at a CAGR of 12.1% during the forecast period. Demand outlook carries the valuation to USD 2.2 billion through 2036 as migration limits compel packaging converters to adopt non-targeted analytical screening for unknown chemical degradation products.
The transition from simple compliance checklists to rigorous chemical profiling forces global food packaging converters to invest heavily in non-targeted analytical methodologies. Converters who delay this transition lose critical supplier contracts with multinational food and beverage brands that refuse to absorb downstream recall liabilities. Most migration failures do not originate from the virgin raw materials themselves, but rather from unpredictable reaction intermediates formed during high-temperature converting processes or secondary recycling loops. This structural shift elevates the importance of food contact safety verification beyond a mere quality control step into a fundamental market access requirement, particularly within the heavily scrutinized FMCG packaging sector.
When regulatory bodies transition the 10 ppb threshold for unidentified substances from an advisory guideline to a legally enforced hard cap, the baseline requirements for market participation permanently change. Global packaging converters must implement continuous non-targeted batch screening across their entire production output within a strict 18-month compliance window. Facilities that successfully integrate these protocols secure their supply chain position, while non-compliant operators face immediate exclusion from premium retail distribution channels.
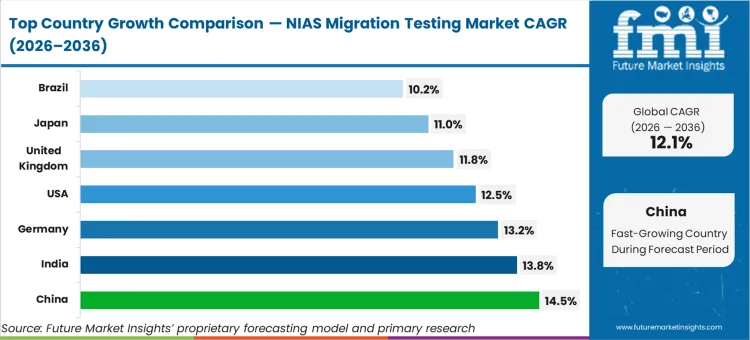
China sets the pace with a 14.5% CAGR, advancing rapidly as the domestic rollout of the updated GB 9685 national standard strictly governs food contact materials. India records a 13.8% rate, driven by expanding export-oriented packaging manufacturing requiring strict international compliance. Germany tracks at 13.2% as robust mechanical recycling infrastructures demand stringent toxicological verification of secondary polymers. The United States follows at 12.5%, responding to heightened FDA scrutiny on complex multi-layer barrier structures. The United Kingdom expands at 11.8%, while Japan reaches an 11.0% trajectory. Brazil posts a 10.2% growth rate. This geographic spread reflects the varying velocities at which national authorities are codifying trace chemical migration thresholds into absolute and enforceable law.
The NIAS migration testing market encompasses specialized analytical services and toxicological risk assessments designed to identify and quantify non-intentionally added substances transferring from packaging into food. These substances include impurities, degradation chemicals, and reaction by-products formed during material manufacturing or recycling processes. The market relies heavily on advanced mass spectrometry techniques and computational toxicology to ensure compliance with international food safety frameworks.
The market scope includes non-targeted chemical screening, specific migration limit (SML) testing, and exposure assessment modeling for complex food contact materials. Services utilizing advanced chromatographic detection methods such as GC-MS and LC-MS to identify volatile and semi-volatile compounds down to trace levels are fully incorporated. Risk assessment services applying the Threshold of Toxicological Concern (TTC) approach to evaluate the safety of unidentified migrants, and analytical procedures used to ensure the safety of smart labels and active materials, also fall strictly within these boundaries.
Standard microbiological pathogen testing and basic nutritional profiling of food products are explicitly excluded from this valuation. Standalone physical stress testing, shelf-life integrity evaluations, and packaging aesthetic assessments are omitted. Routine testing for intentionally added substances (IAS) that does not incorporate dedicated screening protocols for unknown degradation products or reaction intermediates falls completely outside the defined parameters of this specialized analytical market.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.7 billion |
| Industry Value (2036) | USD 2.2 billion |
| CAGR (2026-2036) | 12.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
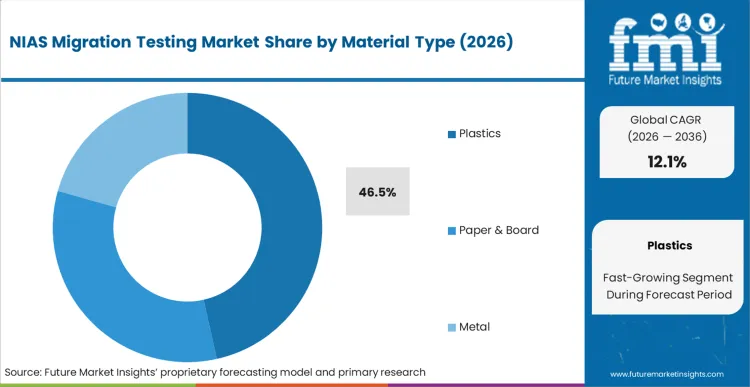
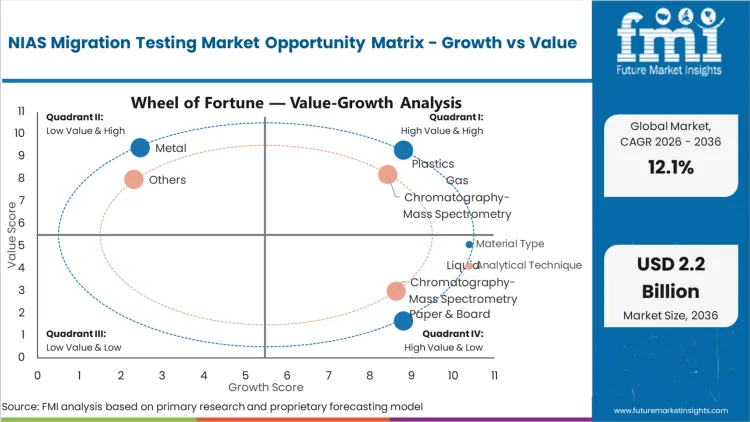
Plastics represent the dominant material segment, capturing 46.5% of the total share in 2026. This concentration is driven directly by the inherent chemical complexity of polymer manufacturing and the aggressive expansion of post-consumer recycled (PCR) content mandates. Global food packaging converters must validate that their recycled polymer resins do not introduce hazardous impurities or breakdown products into the food supply chain. Facilities that fail to implement rigorous screening protocols for these plastic substrates risk catastrophic recall events and permanent damage to their vendor qualification status. According to FMI's estimates, the integration of advanced mass spectrometry into routine quality control allows converters to detect trace oligomers and photoinitiators long before the packaging reaches retail shelves. Quality assurance directors specifying testing protocols for plastic food contact materials must demonstrate compliance with strict specific migration limits to secure brand owner approval.
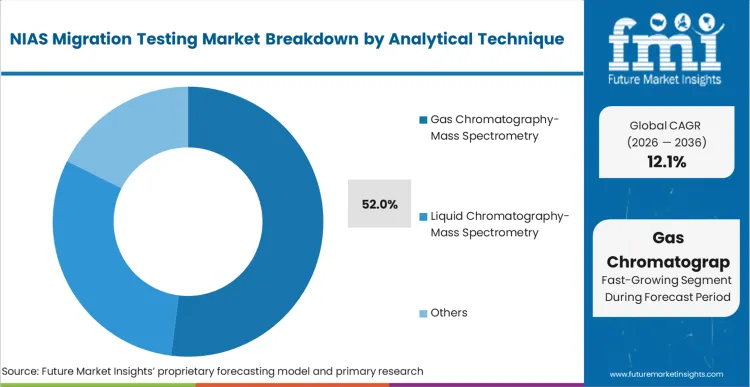
Gas Chromatography-Mass Spectrometry (GC-MS) leads the analytical technique segment, securing a 52.0% share in 2026 due to its unparalleled efficacy in detecting volatile and semi-volatile unknown compounds. Contract laboratory directors invest heavily in this specific instrumentation to meet the 10 ppb (micrograms/kg) screening threshold demanded by European regulators. FMI analysts opine that the expansion of proprietary spectral libraries directly enhances the capability of these systems to confidently identify complex degradation products. Testing laboratories that fail to upgrade their GC-MS capabilities find themselves unable to offer the comprehensive chemical hazard screening required by tier-one packaging suppliers. Without this technical foundation, analytical providers simply cannot execute the rigorous non-targeted methodologies necessary for complete toxicological risk assessments.
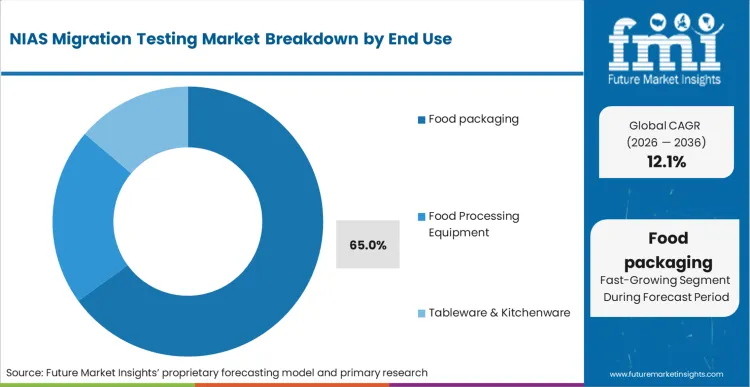
Food packaging emerges as the primary end use, commanding a 65.0% share in 2026 as brand owners intensify their scrutiny of multi-layer barrier films and sustainable paperboard liners. Multinational food and beverage corporations enforce strict vendor qualification standards that require comprehensive documentation of all potential chemical migrants. Based on FMI's assessment, the shift away from legacy single-use plastics toward novel bio-based materials introduces entirely new profiles of non-intentionally added substances that require extensive baseline testing. Packaging procurement leads must present verifiable toxicological risk assessments for every new substrate introduced into their supply chain. Failure to provide this critical analytical data results in immediate disqualification from major commercial tenders.
The strict enforcement of Commission Regulation (EU) No 10/2011 and Article 3 of Framework Regulation (EC) 1935/2004 forces global food packaging converters to fundamentally alter their quality assurance frameworks. This regulatory mandate drives a massive expansion in outsourced analytical screening, as manufacturers must prove that their materials transfer no hazardous constituents into food products. Converters upgrading their production lines face an absolute requirement to document the safety of all chemical species, including those not intentionally added. Facilities that attempt to bypass these comprehensive evaluations face severe product seizures and immediate exclusion from the lucrative metal packaging sector and the broader European consumer market.
The extreme analytical complexity required to identify unknown reaction intermediates creates significant operational bottlenecks for testing laboratories. Translating complex chromatograms into actionable toxicological risk assessments demands highly specialized scientific expertise that remains in critically short supply globally. While laboratories deploy advanced software algorithms to accelerate spectral matching, the final hazard evaluation relies heavily on manual toxicological review, strictly limiting the scalability of high-throughput screening operations and keeping service turnaround times extended.
Based on the regional analysis, the NIAS Migration Testing market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 14.5% |
| India | 13.8% |
| Germany | 13.2% |
| United States | 12.5% |
| United Kingdom | 11.8% |
| Japan | 11.0% |
| Brazil | 10.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Aggressive regulatory modernization programs across the Asia Pacific region fundamentally reshape the compliance obligations for regional packaging manufacturers. Governments overhauling their food safety standards mandate rigorous analytical evaluation of all contact materials, effectively eliminating the historical reliance on self-certification. As per FMI's projection, this sweeping legislative shift forces domestic producers to partner with accredited third-party laboratories to validate their export readiness. The resulting surge in demand for specialized migration screening protocols rapidly expands the regional footprint of international testing conglomerates.
FMI's report includes comprehensive evaluation of the broader regional deterministic frameworks encompassing South Korea, Indonesia, and the ASEAN economic bloc. A prevailing dynamic across these supplementary markets involves the rapid localization of specialized analytical capacity, driven by the unsustainable costs of shipping complex physical samples to centralized testing hubs.
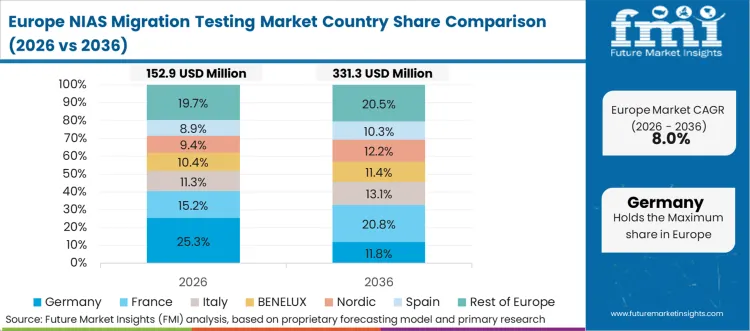
Europe's transition toward a fully circular packaging economy introduces extreme chemical complexity into the material recovery infrastructure. The aggressive mandate to incorporate high percentages of post-consumer recycled content directly conflicts with the absolute requirement for zero hazardous chemical migration. In FMI's view, this structural tension forces recycling facility operators and packaging converters to implement continuous, high-frequency analytical screening. Validating that mechanical and chemical recycling processes effectively strip out non-intentionally added substances requires highly sophisticated contaminant detection networks operating at the very edge of analytical capability.
FMI's report encompasses detailed intelligence tracking the Nordic and Benelux regions, highlighting intense corporate sustainability pledges that push testing demands far beyond basic regulatory baselines. The shift from synthetic fossil-derived materials toward biologically sourced alternatives is creating entirely new demands for unknown substance quantification across these environmentally conscious nations.
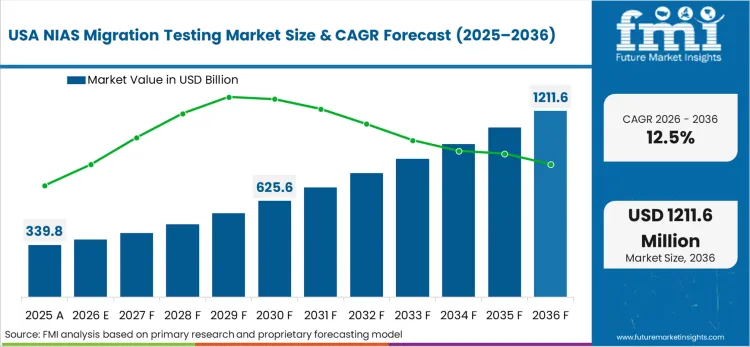
Heightened litigation risks and intense consumer advocacy pressure in the Americas force major food brands to adopt highly defensive procurement strategies. Multinational corporations operating across these jurisdictions increasingly reject the baseline regulatory minimums, instead demanding proactive, non-targeted screening of all supplier materials. FMI analysts opine that this aggressive risk mitigation approach forces packaging converters to execute extensive exposure assessments before a new product ever reaches the commercialization phase. By demanding comprehensive protective packaging validation, these massive buyers effectively set private safety standards that far exceed federal requirements.
FMI's coverage evaluates the expansive North American supply network, assessing Canada and Mexico's structural roles in supporting multinational brand compliance. Increased integration of cross-border packaging ecosystems forces regional converters to harmonize their toxicological screening protocols to ensure uninterrupted continental product distribution.
The global NIAS migration testing market operates as a highly concentrated landscape dominated by a select group of multinational testing, inspection, and certification (TIC) conglomerates. This consolidated structure exists because the capital intensity required to outfit facilities with state-of-the-art mass spectrometry equipment, combined with the difficulty of recruiting specialized toxicologists, creates an immense barrier to entry. Industry heavyweights such as Eurofins Scientific SE, Intertek Group plc, and SGS SA leverage their massive global laboratory networks to secure exclusive, multi-year compliance contracts with top-tier food and beverage brands. Buyers primarily evaluate these vendors based on the proprietary breadth of their chemical spectral libraries and their established credibility with regional regulatory authorities like EFSA and the FDA.
Established market leaders maintain a profound structural advantage through their accumulated historical databases of known reaction intermediates and polymer degradation profiles. A regional challenger attempting to disrupt this hierarchy must specifically build a comprehensive, digitally integrated toxicological modeling platform capable of matching the rapid turnaround times of the incumbents. The ability to seamlessly integrate analytical reporting data directly into a client's supply chain management software represents a critical differentiator. Challengers that fail to invest in predictive in silico capabilities will remain relegated to low-margin, targeted analytical work, entirely locked out of the lucrative non-targeted screening sector.
To counteract the pricing leverage held by these massive testing conglomerates, large packaging converters actively split their compliance portfolios across multiple accredited vendors. This structural tension between buyers seeking cost-efficiency and laboratories demanding premium rates for specialized toxicological assessments naturally limits extreme pricing power, even within a highly concentrated market. Furthermore, leading packaging manufacturers are increasingly investing in their own internal baseline screening capabilities to pre-qualify materials before submitting them for formal third-party certification. Moving toward 2036, the market is projected to undergo moderate fragmentation at the regional level, as agile, highly specialized boutique laboratories emerge to service the specific analytical needs of niche bio-based compostable adhesives startups.

| Metric | Value |
|---|---|
| Quantitative Units | USD 0.7 billion to USD 2.2 billion, at a CAGR of 12.1% |
| Market Definition | The NIAS migration testing market encompasses specialized analytical services and toxicological risk assessments designed to identify and quantify non-intentionally added substances transferring from packaging into food. |
| Material Type Segmentation | Plastics, Paper & Board, Metal, Glass & Ceramics, Multi-material/Others |
| Analytical Technique Segmentation | Gas Chromatography-Mass Spectrometry (GC-MS), Liquid Chromatography-Mass Spectrometry (LC-MS), Inductively Coupled Plasma (ICP), Others |
| End Use Segmentation | Food Packaging, Food Processing Equipment, Tableware & Kitchenware |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, United States, United Kingdom, Japan, Brazil, and 40 plus countries |
| Key Companies Profiled | Intertek Group plc, Smithers, Eurofins Scientific SE, SGS SA, TÜV SÜD, Bureau Veritas SA, Mérieux NutriSciences |
| Forecast Period | 2026 to 2036 |
| Approach | The baseline value derives from a bottom-up aggregation of commercial laboratory testing volumes, applying region-specific regulatory enforcement curves to project the future adoption velocity. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the NIAS migration testing in 2026?
The NIAS migration testing market is estimated to be valued at USD 0.7 billion in 2026 as stringent migration limits force packaging converters to abandon targeted legacy assays.
What will it be valued at by 2036?
Market size for NIAS migration testing is projected to reach USD 2.2 billion by 2036 as continuous non-targeted batch screening becomes a universal supply chain requirement.
What CAGR is projected?
Demand for NIAS migration testing is expected to grow at a CAGR of 12.1% between 2026 and 2036, driven by the structural necessity of identifying unpredictable reaction intermediates.
Which material type segment leads?
Plastics leads with a 46.5% share in 2026 as intense chemical complexity and recycled content integration demand massive contamination baseline testing.
Which analytical technique segment leads?
Gas Chromatography-Mass Spectrometry (GC-MS) leads because its detection precision is structurally required to isolate and identify semi-volatile compounds down to 10 ppb thresholds.
Which end use segment leads?
Food Packaging dominates due to massive global production volumes and intense brand owner scrutiny of multi-layer barrier films.
What drives rapid growth?
The strict enforcement of Commission Regulation (EU) No 10/2011 forces global food packaging converters to fundamentally alter their quality assurance frameworks and invest in advanced screening.
What is the primary restraint?
Translating complex chromatograms into actionable toxicological risk assessments demands highly specialized scientific expertise that remains in critically short supply globally.
Which country grows fastest?
China expands at 14.5% compound rate, advancing rapidly as the domestic rollout of the updated GB 9685 national standard strictly governs food contact materials.
How does Framework Regulation (EC) 1935/2004 affect testing volumes?
Article 3 mandates that all migrating substances must be safe, structurally requiring laboratories to execute comprehensive hazard evaluations for previously ignored degradation products.
Why is non-targeted analysis replacing legacy methods?
Targeted chemical analysis only finds known substances, forcing the industry to adopt non-targeted screening methods capable of detecting entirely unknown degradation and reaction products.
How do the top TIC players compete for enterprise contracts?
Incumbents fiercely protect their market position by heavily investing in the proprietary breadth of their mass spectral libraries and highly specialized computational toxicology platforms.
How does India navigate shifting regulations?
Expanding export-oriented manufacturing in India demands strict adherence to European food safety frameworks, driving intense localized laboratory capital expansion.
What creates pressure in the German market?
Germany's extensive mechanical recycling infrastructure demands rigorous chemical verification to clear secondary polymers for direct food contact.
Why is the 10 ppb threshold significant?
It represents the critical analytical sensitivity point where undetected non-intentionally added substances begin to trigger massive regulatory rejection events.
What role does blockchain play in compliance?
Digital traceability integration connects specific physical test results directly to individual polymer batches, granting manufacturers absolute proof of compliance during audits.
How do USA buyers influence vendor standards?
Intense consumer advocacy pressure forces major food brands to adopt defensive procurement strategies, setting private safety standards that far exceed federal requirements.
Why are recycled resins driving testing demand?
Secondary plastics carry significant risk of legacy contamination, requiring continuous batch-level exposure assessments before being cleared for consumer contact.
What defines the United Kingdom's adoption velocity?
Post-Brexit regulatory divergence forces packaging manufacturers to navigate dual compliance pathways, multiplying the total volume of necessary toxicological evaluations.
How does Brazil integrate mass spectrometry?
The massive agricultural export sector requires absolute confidence in bulk transit packaging, triggering heavy capital investment in modern analytical laboratories.
What falls outside the scope of this report?
Standard microbiological pathogen testing and routine evaluations for intentionally added substances that lack unknown degradation screening are explicitly excluded.
How was this forecast validated?
Projections underwent rigorous cross-validation against publicly reported capital expenditure guidance from the world's leading testing, inspection, and certification conglomerates.
What is the core strategic takeaway for packaging manufacturers?
Facilities must implement continuous non-targeted batch screening protocols to prevent catastrophic product recalls before regional enforcement deadlines permanently shut them out of retail channels.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.