Real World Evidence Linkage Services Market
The Real World Evidence Linkage Services Market Is Segmented By Data Source (Claims Data, EHR Data, Registry Data, Trial Data, And Lab Data), Linkage Method (Tokenization, Deterministic Matching, Probabilistic Matching, Hybrid Matching, And Graph Linkage), Delivery Model (Managed Services, Project-Based, Platform-Enabled, And Embedded Linkage), Use Case (Regulatory Studies, Safety Studies, HEOR Studies, Synthetic Controls, And Trial Follow-Up), End User (Biopharma, Cros, Medtech, Payers, And Researchers), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Real World Evidence Linkage Services Market Size, Market Forecast and Outlook By FMI
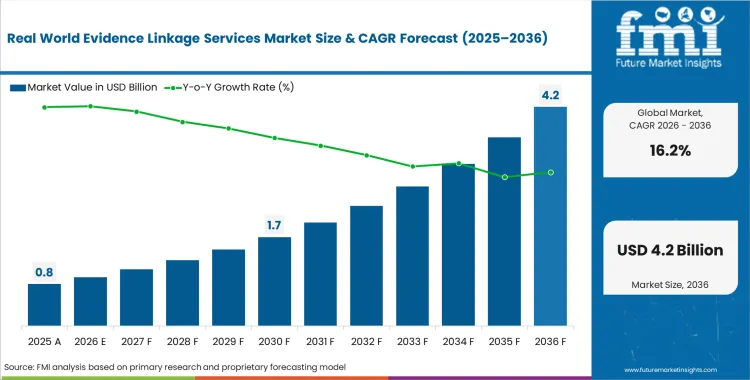
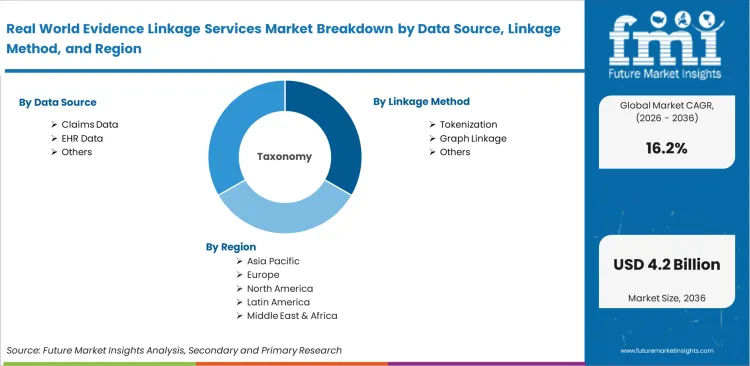
The real world evidence linkage services market fulfilled the estimated USD 0.7 billion in 2025. This industry, as it steadily progresses is now anticipated to cross USD 0.8 billion in 2026 at a CAGR of 16.2% during the forecast period. Revenue is expected to reach USD 3.6 billion by 2036 as life sciences organizations connect fragmented patient data into unified longitudinal records to meet more detailed regulatory safety requirements.
Sourcing directors at top-tier pharmaceutical companies face a fundamental shift in how longitudinal patient records are constructed for post-market surveillance. Procurement priorities are moving away from simple data acquisition toward high-fidelity connection services that bridge the gap between clinical trial results and real world evidence solutions. Delaying the integration of fragmented claims and clinical records no longer just slows down research; it creates significant liabilities in drug safety monitoring and market access negotiations. The cost of healthcare data linkage services now frequently exceeds the cost of the underlying data licenses, because the ability to match patient identities across different EHR systems has become the primary factor in whether a study meets regulatory standards.
Summary of Real World Evidence Linkage Services Market
- Real World Evidence Linkage Services Market Definition
- This market encompasses the specialized services required to link fragmented patient data across different healthcare settings while maintaining strict privacy standards. It utilizes advanced matching algorithms and tokenization to enable comprehensive longitudinal studies for regulatory and commercial purposes.
- Demand Drivers in the Market
- Regulatory pressure from the FDA and EMA forces safety officers to provide evidence of drug efficacy in diverse, real-world populations beyond controlled trial environments.
- The rise of high-cost cell and gene therapies compels payers to demand longitudinal outcome data before finalizing value-based reimbursement agreements.
- Increased adoption of decentralized clinical trials pushes R&D directors to link remote monitoring data with existing medical history to ensure patient safety.
- Key Segments Analyzed in the FMI Report
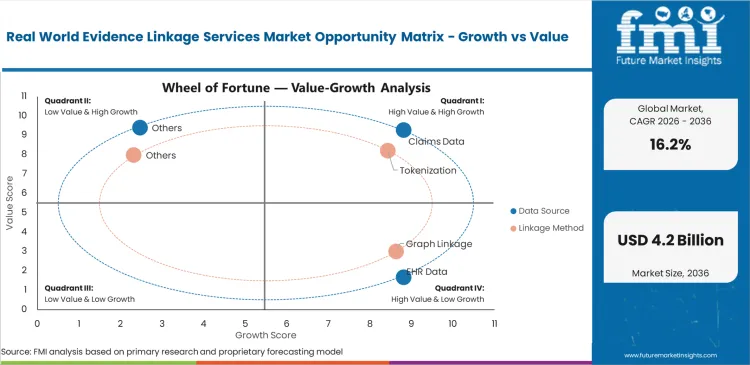
- Claims Data: 34.0% share in 2026, providing the foundational cost and utilization baseline required for insurance-linked outcomes research.
- Tokenization: 39.0% share in 2026, as privacy-preserving protocols become the non-negotiable standard for cross-institutional data sharing.
- Biopharma: 43.0% share in 2026, driven by the structural requirement to validate long-term safety profiles for novel biological products.
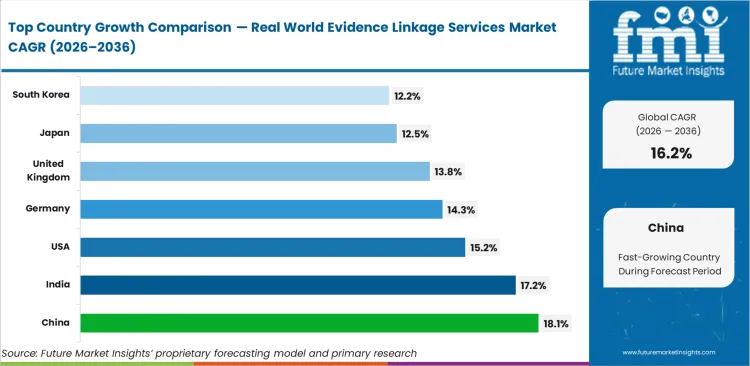
- China: 18.1% CAGR, reflecting massive investments in healthcare digitization and a centralized push for nationwide patient data standardization.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Analyst, Healthcare, at FMI, points out, " Our latest assessment shows that regulators are placing greater weight on real-world evidence in both drug approvals and post-market review. RWE linkage services help pharmaceutical companies understand how treatments perform across patients with different ages, health conditions, and care settings. Companies that keep their data accurate, meet privacy standards, and use validated linkage methods are in a stronger position to support regulatory submissions."
- Strategic Implications / Executive Takeaways
- HEOR directors must prioritize the validation of linkage algorithms over data volume to ensure reimbursement submissions withstand payer scrutiny.
- Data strategy leads face a choice between building in-house tokenization capabilities or partnering with managed service providers to offload compliance risks.
- R&D heads at mid-size biotechs must implement linkage early in Phase II trials to create a seamless transition into post-market surveillance activities.
- Methodology
- FMI's approach focuses on the structural shift from manual data cleaning to automated clinical data provenance management to ensure forecast accuracy. The model integrates primary insights from data privacy officers with hard data on API transaction volumes to map the true adoption of linkage services.
Data privacy officers at major healthcare networks act as the primary triggers for broader adoption by moving from manual consent-based matching to automated healthcare interoperability solutions. This transition represents a critical structural gate where within this market, privacy-preserving record linkage is moving from an occasional research tool to a standard operational requirement. Once this threshold is crossed, the friction of multi-institutional data sharing dissipates, allowing for the rapid scaling of federated research networks that preserve patient anonymity while delivering unprecedented clinical depth.
China leads growth at an 18.1% CAGR, where rapid hospital digitization has created large pools of EHR data that have not yet been connected, followed by India at 17.2%, which is growing its role as a clinical trial hub. The United States follows at a 15.2% CAGR, supported by detailed HEOR reporting requirements. Germany is at 14.3% and the UK at 13.8%, as researchers in both countries work through multi-country privacy regulations to share data across borders. Japan linked healthcare data services at 12.5% and the South Korea RWD linkage market at 12.2% remain key growth areas, showing a clear split between markets centered on primary data creation and those focused on connecting historical data for regulatory compliance.
Real World Evidence Linkage Services Market Definition
Identity resolution in this sector refers to the technical and operational bridge connecting disparate patient datasets, such as EHRs, insurance claims, and genomic profiles, into a single longitudinal record without compromising privacy. Structural boundaries of this market focus on the service layer that manages data cleaning, normalization, and secure tokenization. It is analytically distinct from general data brokerage because it prioritizes the mechanism of connection rather than the content of the data itself.
Real World Evidence Linkage Services Market Inclusions
Inclusions focus on patient data tokenization services, privacy-preserving record linkage (PPRL) protocols, and deterministic or probabilistic matching architectures. Scope extends to healthcare API integrations that facilitate real-time data streaming and specialized consulting for data governance. Services designed specifically for health technology assessment (HTA) and synthetic control arm data services also fall within these operational boundaries.
Real World Evidence Linkage Services Market Exclusions
General data storage, cloud infrastructure, and the sale of raw, unlinked medical records are excluded from this analysis. Software used solely for internal hospital administration or billing that does not facilitate cross-institutional research linkage is outside the scope. The third-party analytics services that do not perform the underlying identity resolution or tokenization tasks are further omitted to maintain focus on the linkage mechanism.
Real World Evidence Linkage Services Market Research Methodology
- Primary Research: Interviews with HEOR directors, biopharma data strategy leads, and clinical trial operations managers at Tier-1 pharmaceutical firms.
- Desk Research: Analysis of FDA/EMA real-world data guidance, HIPAA/GDPR compliance frameworks, clinical trial registries, and patent filings for PPRL technologies.
- Market-Sizing and Forecasting: Baseline anchored by the cumulative volume of tokenized patient records and annual contract values for managed linkage services.
- Data Validation and Update Cycle: Forecasts cross-validated against independent life sciences R&D spend reports and data interoperability adoption rates across major EHR vendors.
Segmental Analysis
Real World Evidence Linkage Services Market Analysis By Data Source
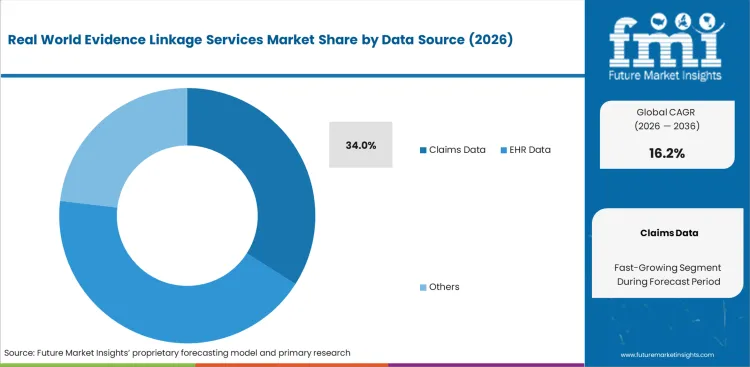
This sub-segment holds 34.0% share in 2026, as claims data represents the foundational layer of longitudinal research because it provides a continuous, multi-year record of patient interactions regardless of where care was delivered. FMI's assessment is that the structural permanence of claims as a linkage anchor is often underestimated by generalists focusing solely on clinical depth. While EHR data offers clinical precision, procurement directors at major life science firms recognize that insurance records provide the necessary 'glue' to follow patients as they move between different health systems or providers. The operational reality for a health economics researcher is that without EHR claims linkage services, the total cost of care and long-term medication adherence remain speculative. Moving from basic ICD-10 matching to more complex clinical trial data linkages is the difference between a standard safety study and one that meets regulatory submission requirements.
- Economic Baseline Anchor: Claims records provide the only reliable mechanism for tracking total medical expenditure across fragmented provider networks. Sourcing managers at payers use linked claims data to validate value-based care outcomes, ensuring that reimbursement reflects actual clinical results rather than isolated procedure codes.
- Adherence Tracking Logic: Linking pharmacy claims with clinical records allows medical directors to identify the specific point of treatment failure. R&D directors at pharmaceutical firms gain the ability to distinguish between a drug's lack of efficacy and a patient's failure to fill prescriptions at the retail level.
- Regulatory Safety Reporting: Federal safety officers require claims-linked longitudinally to detect rare adverse events that may not appear in short-term trial datasets. Procurement teams at linked real world data providers must qualify suppliers based on their ability to merge high-volume claims with patient registry software to meet these rigorous post-market reporting standards.
Real World Evidence Linkage Services Market Analysis By Linkage Method
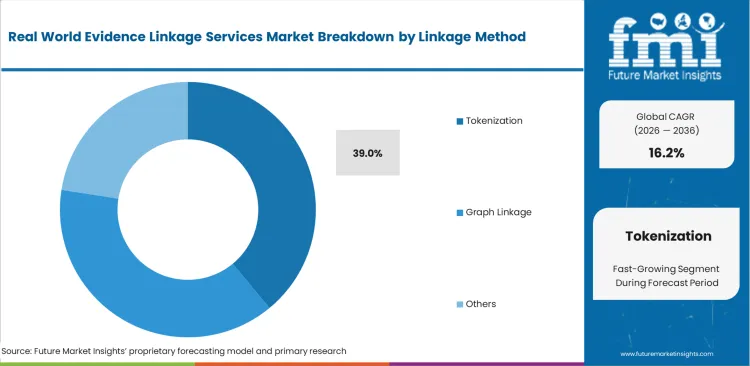
Patient data tokenization services often perform better in procurement reviews because they align more closely with the legal risk standards set by data privacy officers. This shift is also shaping buying behavior among R&D directors at mid-sized companies, many of whom now see privacy-preserving record linkage as a factor that can increase dependence on a single vendor. Once a clinical researcher validates a drug's safety profile inside a specific tokenized geometry, re-qualifying for a different clinical trials support software solutions protocol restarts the full compliance clock. Tokenization holds 39.0% of the market by linkage method. Researchers often overlook the fact that while tokenization scales well for structured data, it performs poorly on unstructured clinical notes, which limits its usefulness in studies that rely on free-text records.
- Privacy Risk Mitigation: Tokenization eliminates the need for moving personally identifiable information (PII) across institutional boundaries during the research process. Data privacy officers at hospital networks approve these services because they minimize the risk of re-identification while maintaining high-fidelity healthcare business intelligence capabilities.
- Multi-Source Interoperability: Standardized token formats allow for the seamless connection of genomic, clinical, and socio-economic data from entirely different vendors. Sourcing leads at biopharma companies prioritize token-compatible datasets to avoid the high costs associated with manual deterministic vs probabilistic matching healthcare evaluation projects.
- Longitudinal Record Stability: Maintaining a consistent token across multiple years of data updates ensures that the patient trajectory remains accurate without repeated consent requirements. Clinical operations managers rely on this stability to build robust synthetic control arms that mirror current standard-of-care patient profiles.
Real World Evidence Linkage Services Market Analysis By Delivery Model
Outsourced patient matching services dominate because most life sciences organizations do not have the internal infrastructure to run large-scale probabilistic matching. This segment holds 46.0% share, as FMI analysts note that maintaining healthcare analytics pipelines makes outsourced linkage a structural necessity rather than an operational choice. Procurement directors face a 12-to-18-month lead time to build internal linkage teams, forcing them to rely on established RWE services vendors who already possess pre-negotiated data rights with major EHR networks. Managed services often process data inside a closed system where the buyer never sees the underlying personal identifiers. This setup can actually speed up regulatory approval for multi-center studies because the risk of re-identification is lower.
- Compliance Offloading: Outsourcing identity resolution to a third-party managed service provider shifts the burden of HIPAA and GDPR compliance to a specialized entity. Legal counsel at biotech startups mandates de-identified data linkage services to reduce the liability associated with handling sensitive life science analytics data internally.
- Infrastructure Scaling: Managed linkage services provide the elastic compute power needed to process billions of records during peak study periods. IT directors at global pharmaceutical companies avoid the capital expenditure of building proprietary linkage engines by utilizing specialized service providers on a subscription basis.
- Expertise Gap Bridging: Specialized linkage providers possess the domain knowledge to handle messy, non-standardized EHR data from diverse global regions. Research leads at healthcare contract research organization firms use these services to ensure that international data is correctly normalized for global regulatory submissions.
Real World Evidence Linkage Services Market Drivers, Restraints, and Opportunities
Commercial stakes for regulatory-grade linkage services have moved from "nice-to-have" research projects to a critical forcing condition for drug approval. HEOR data linkage services directors at Tier-1 biopharma firms now face a scenario where payers refuse to reimburse novel therapies without linked evidence of long-term patient benefit. This pressure is compounded by the speed at which competitors are using clinical research organization services to build synthetic control arms, potentially shortening trial timelines by months. Organizations that do not implement linkage protocols early may lose ground to smaller companies that can demonstrate real-world clinical results faster than incumbents can complete traditional Phase IV studies.
The main barrier to adoption is that patient data is fragmented across EHR systems that use different definitions and data formats. Even when tokenization is applied, the underlying clinical definitions, such as what constitutes a "disease progression" event, vary wildly between hospital systems. This misalignment forces researchers to spend up to 70.0% of their project timeline on manual data normalization and mapping, creating an operational bottleneck that digital transformation in healthcare tools have yet to fully automate. While vendors promise "plug-and-play" linkage, practitioners find that the specific clinical nuances of each specialty require bespoke mapping, a reality that keeps costs high and adoption rates moderate despite strong institutional desire for unified patient data.
- Genomic-Clinical Convergence: Connecting global digital health records with genomic sequencing data allows researchers to identify specific patient sub-groups that respond best to precision medicine.
- Automated Trial Recruitment: Utilizing linked claims and EHR datasets enables artificial intelligence in healthcare models to identify eligible trial participants in real-time, significantly reducing enrollment costs.
- Synthetic Control Arm Construction: Providing high-fidelity linked records for rare diseases allows biotech companies to avoid the high cost and ethical challenges of recruiting a placebo arm in small patient populations.
Regional Analysis
Countries across this market are at different stages of moving from fragmented patient data systems toward unified, tokenized records, and each operates under different privacy regulations. Global adoption curves are currently shaped by the varying speeds of hospital digitization and the localized maturity of healthcare data linkage services.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 18.1% |
| India | 17.2% |
| United States | 15.2% |
| Germany | 14.3% |
| United Kingdom | 13.8% |
| Japan | 12.5% |
| South Korea | 12.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Real World Evidence Linkage Services Market Analysis
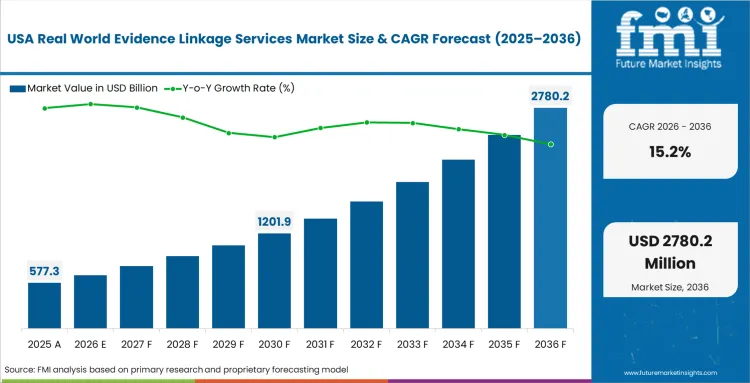
Policy mandates in the United States have moved from simple electronic record-keeping to a structural requirement for "meaningful use" of patient-generated health data. The trajectory of this region is defined by the integration of patient portals with centralized research networks, forcing providers to adopt standardized API protocols. FMI observes that the maturity of the USA insurance market makes it the global leader in EHR claims linkage services.
- United States: Regulatory guidelines from the FDA regarding the use of real-world data in regulatory submissions have created a standardized pathway for drug approvals using linked evidence. The market is expected to grow at a 15.2% CAGR as USA biopharma firms increasingly work with patient tokenization vendors to build longitudinal records that meet these new requirements. This creates a significant commercial advantage for early adopters who can submit more robust safety profiles to federal agencies.
FMI’s report includes additional countries like Canada. This market is characterized by a high degree of regional health data centralization, which facilitates large-scale longitudinal studies while maintaining strict adherence to provincial privacy standards.
Asia-Pacific Real World Evidence Linkage Services Market Analysis
Infrastructure-led dynamics define the adoption curve in Asia-Pacific, where the rapid construction of massive centralized hospital databases provides an ideal environment for large-scale real world evidence data integration services. The structural condition here is the leapfrogging of legacy paper systems directly into cloud-based patient engagement platforms in emerging markets.
- China: China is growing at 18.1%, as healthcare administrators use tokenization to connect clinical records with genomic research. This is supported by government programs to standardize EHR data across provincial hospitals. This systemic push has created the world’s largest pool of connectable patient records, positioning the country as a primary destination for global precision medicine research.
- India: Clinical research leads are leveraging the expansion of the National Digital Health Mission to link trial data with local pharmacy records, resulting in a 17.2% CAGR that significantly improves the operational outcome of long-term safety studies. By creating a unified health ID system, the mission simplifies identity resolution across public and private sectors for the India real world data linkage market.
- Japan: Aging population demographics are forcing the Ministry of Health to adopt linked RWE for drug pricing and reimbursement negotiations, with a forecast 12.5% CAGR through the next decade. Researchers are currently integrating clinical decision support app data with geriatric registries to optimize long-term care outcomes.
- South Korea: South Korea is growing at 12.2%, as technology directors incorporate patient-reported outcome linkage into digital health platform workflows. High levels of smartphone penetration and digital health adoption in the South Korea RWD linkage market have led to a surge in trial follow-up efficiency.
FMI’s report includes additional countries like Australia and Singapore. These markets demonstrate a structural trajectory toward high-trust, cross-border data linkage for regional health security.
Europe Real World Evidence Linkage Services Market Analysis
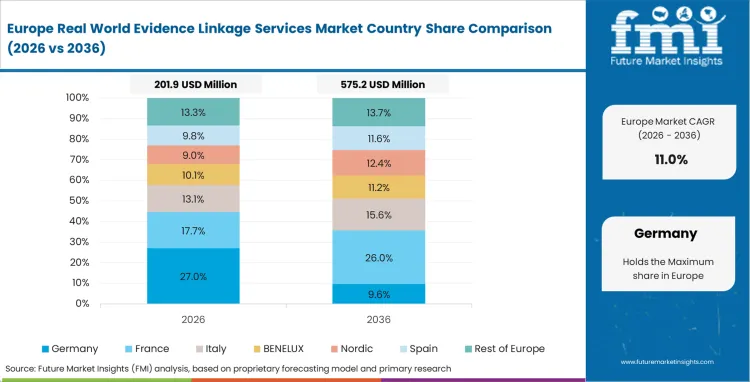
The European landscape is increasingly shaped by the shift toward high-trust, cross-border research frameworks such as the European Health Data Space (EHDS). Researchers in this region are navigating complex multi-national privacy regulations while maintaining the clinical utility of de-identified data linkage services. FMI notes that the region’s focus on regulatory-grade linkage services is a direct response to stringent EMA safety monitoring requirements.
- Germany: Germany’s patient data linkage services are expanding at a 14.3% CAGR as hospital networks adopt privacy-preserving record linkage standards to comply with the Hospital Future Act. This digital transformation allows researchers to connect inpatient records with long-term outpatient follow-up data while strictly adhering to local data protection laws.
- United Kingdom: In the United Kingdom, RWE linkage services are projected to grow at a 13.8% CAGR, supported by NHS infrastructure that enables longitudinal patient journey linkage at scale. Evidence teams are increasingly stitching trial IDs with insurance records to monitor patient outcomes after formal study periods end, ensuring study validity for reimbursement dossiers.
FMI’s report includes additional countries like France and Italy. These markets are currently focusing on the development of centralized national health data hubs to facilitate secure identity resolution for large-scale epidemiological research.
Competitive Aligners for Market Players
The competitive dynamic in the RWE linkage services market is defined by the tension between established data aggregators and specialized privacy-tech entrants. Companies like Datavant hold a strong position by building a neutral network that lets different parties link data without exposing the underlying proprietary clinical assets. Buyers at Oracle Life Sciences and IQVIA choose Datavant specifically because it acts as a neutral intermediary that does not compete with their own data products. Fragmentation remains high at the service tier because the specific cleaning requirements for oncology linked data providers differ fundamentally from the claims-heavy focus of Veradigm.
Incumbents possess a critical capability that challengers cannot replicate quickly, a massive library of pre-validated data connectors and established legal DUA (Data Use Agreement) templates with thousands of healthcare provider sites. Building a tokenization engine is the easy part; the structural barrier is the decade-long process of qualifying as a trusted data processor within the firewall of major health systems like TriNetX. Challengers must focus on building superior clinical trials support software solutions that can handle unstructured data linkage through natural language processing, a capability that incumbents are currently scrambling to acquire through partnership rather than internal development.
Large biopharma buyers are actively resisting vendor lock-in by mandating the use of "open" tokenization standards that allow them to switch linkage providers without losing their longitudinal historical data. This buyer power is forcing the market toward a bifurcated structure where identity resolution becomes a commodity service, while the high-margin competitive battleground shifts to the analytical interpretation of the linked results. By 2036, the structural trajectory of competition will be defined by who controls the "master patient index" at the global level, a position that requires both technical scale and the highest level of multi-national regulatory trust.
Key Players in Real World Evidence Linkage Services Market
- Datavant
- IQVIA
- TriNetX
- Oracle Life Sciences
- Veradigm
- OM1
- Flatiron Health
- HealthVerity
- Komodo Health
- Tempus
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.8 billion to USD 3.6 billion, at a CAGR of 16.2% |
| Market Definition | Specialized services for the secure, privacy-preserving connection of disparate patient datasets, including EHRs, insurance claims, and genomic data, to create unified longitudinal records for clinical and regulatory analysis. |
| Segmentation | Data Source (Claims, EHR, Registry, Trial, Lab), Linkage Method (Tokenization, Matching Types), Delivery Model, Use Case, End User, Region. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa. |
| Countries Covered | United States, Canada, Brazil, Mexico, Germany, UK, France, China, India, Japan, South Korea. |
| Key Companies Profiled | Datavant, IQVIA, TriNetX, Oracle Life Sciences, Veradigm, OM1, Flatiron Health. |
| Forecast Period | 2026 to 2036 |
| Approach | Proprietary forecasting model based on tokenized record volume, life sciences R&D spend, and primary research with HEOR and data privacy leads. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Real World Evidence Linkage Services Market Segments
Data Source
- Claims Data
- EHR Data
- Registry Data
- Trial Data
- Lab data
Linkage Method
- Tokenization
- Deterministic Matching
- Probabilistic Matching
- Hybrid Matching
- Graph Linkage
Delivery Model
- Managed Services
- Project-based
- Platform-enabled
- Embedded Linkage
Use Case
- Regulatory Studies
- Safety Studies
- HEOR Studies
- Synthetic Controls
- Trial Follow-up
End User
- Biopharma
- CROs
- Medtech
- Payers
- Researchers
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- USA Food and Drug Administration. (2024, July). Real-world data: Assessing electronic health records and medical claims data to support regulatory decision-making for drug and biological products.
- European Medicines Agency. (2024). Real-world evidence provided by EMA: Support for regulatory decision-making.
- Centers for Medicare & Medicaid Services. (2025). Research identifiable file data use agreement policies.
- Tyagi, K., & Willis, S. J. (2025). Accuracy of privacy preserving record linkage for real world data in the United States: A systemic review. JAMIA Open, 8(1), ooaf002.
- Datavant. (2025, April). Datavant Analysis: 2025 Trends in Clinical Trial Tokenization and Real-World Data Linkage.
- Gavin, K. M., Sundermann, M. L., & Wieland, A. (2025). Leveraging real-world data for safety signal detection and risk management in pre- and post-market settings. Frontiers in Drug Safety and Regulation, 5, 1626822.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Why Claims Data remains the dominant structural anchor for longitudinal record construction.
- The role of Tokenization as a primary driver for multi-institutional research collaboration.
- How Managed Services provide the necessary infrastructure for scalable identity resolution.
- The impact of FDA and EMA guidances on the adoption of linkage protocols.
- Competitive dynamics between data aggregators and neutral privacy-tech platforms.
- Strategic implications for HEOR and R&D directors in building longitudinal patient profiles.
- Regional growth disparities driven by hospital digitization and regulatory frameworks.
- The shift toward synthetic control arms as a mechanism for reducing trial costs.
Frequently Asked Questions
How big is the real world evidence linkage services market?
The real world evidence linkage services market is valued at USD 0.8 billion in 2026, representing a critical segment of the broader life sciences data sector.
What are the main growth drivers for RWE linkage services?
Revenue is expected to expand at a CAGR of 16.2%, reaching a total valuation of USD 3.6 billion by 2036 as manual matching shifts to automated tokenization.
What data sources are used in real world evidence linkage?
Claims Data holds a 34.0% share because it provides the most consistent longitudinal record of patient behavior across different providers.
How does privacy-preserving record linkage work in healthcare?
Identity resolution replaces sensitive patient identifiers with unique, encrypted keys to allow for record linkage without moving personally identifiable information.
How are EHR, claims, and registry records linked for RWE studies?
The best way to link EHR and claims data for regulatory-grade RWE involves automated tokenization that maps disparate patient IDs into a unified longitudinal patient journey linkage.
Which industries buy real world evidence linkage services?
Biopharma RWE linkage services account for 43.0% of the market share, using linkage to build long-term safety profiles and synthetic control arms.
Can clinical trial data be linked to claims databases?
Yes, clinical trial to RWD linkage is increasingly used to track post-trial patient safety and long-term health utilization by stitching trial IDs with insurance records.
Why is EHR and claims linkage important for RWE?
Linked datasets prevent gaps in patient-level longitudinal data services that are required for regulatory and reimbursement submissions.
How accurate is privacy-preserving patient matching?
Modern tokenized patient matching for RWE achieves high precision by using multi-source validation and advanced probabilistic algorithms to ensure data is suitable for regulatory-grade linkage services.
What is tokenization in healthcare data linkage?
Tokenization is the process of de-identifying data by replacing sensitive fields with non-sensitive digital equivalents to allow secure connection without exposing real identities.
Which vendors are most active in healthcare data linkage?
Prominent linked real world data providers include Datavant, IQVIA, and TriNetX, which provide the infrastructure to bridge fragmented datasets while maintaining privacy.
How are linked datasets used in regulatory studies?
Linked records allow pharmaceutical companies to provide the FDA and EMA with evidence of drug performance in broad, real-world populations for safety signal detection.
What are the main barriers to scaling patient-level linkage?
Persistent data fragmentation and a lack of semantic interoperability across EHR versions remain the primary restraints causing high manual normalization burdens.
What is the difference between deterministic and probabilistic matching?
Deterministic matching requires exact matches on unique identifiers, while probabilistic matching uses statistical weights to resolve identities across incomplete datasets.
Which vendors offer privacy-preserving linkage for clinical trial follow-up?
Specialist firms like Datavant or larger platforms like IQVIA facilitate post-trial follow-up linkage to monitor patient outcomes after a study formally concludes.
What are the key implications for CROs in this market?
CRO real world data linkage support is becoming a core service offering as sponsors shift toward hybrid study designs requiring complex data integration.
Why are researchers increasingly linking lab data with EHRs?
Linking these sources allows for much more precise patient stratification and outcome measurement by providing objective biomarkers to validate subjective clinical notes.
How do patient registries benefit from external linkage?
Registry linkage services pharma allow managers to continue tracking patient outcomes through claims data even after patients stop visiting participating registry sites.
What is the structural trajectory of the European market?
Europe is moving toward high-trust, cross-border research frameworks like the European Health Data Space to navigate complex privacy regulations.
How does FMI cross-validate its market forecasts?
FMI cross-validates identity resolution forecasts against API transaction volumes between major EHR vendors and life science analytics platforms.
What will be categorically different about this market by 2036?
By 2036, the Real World Evidence Linkage Services Market is expected to reach USD 3.6 billion, with identity resolution largely automated across major health systems.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Data Source
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Data Source , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Data Source , 2026 to 2036
- Claims Data
- EHR Data
- Others
- Claims Data
- Y to o to Y Growth Trend Analysis By Data Source , 2021 to 2025
- Absolute $ Opportunity Analysis By Data Source , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Linkage Method
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Linkage Method, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Linkage Method, 2026 to 2036
- Tokenization
- Graph Linkage
- Others
- Tokenization
- Y to o to Y Growth Trend Analysis By Linkage Method, 2021 to 2025
- Absolute $ Opportunity Analysis By Linkage Method, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Biopharma
- CROs
- Payers
- Biopharma
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Data Source
- By Linkage Method
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Source
- By Linkage Method
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Data Source
- By Linkage Method
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Source
- By Linkage Method
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Data Source
- By Linkage Method
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Source
- By Linkage Method
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Data Source
- By Linkage Method
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Source
- By Linkage Method
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Data Source
- By Linkage Method
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Source
- By Linkage Method
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Data Source
- By Linkage Method
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Source
- By Linkage Method
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Data Source
- By Linkage Method
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Source
- By Linkage Method
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Source
- By Linkage Method
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Data Source
- By Linkage Method
- By End User
- Competition Analysis
- Competition Deep Dive
- Datavant
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- IQVIA
- TriNetX
- Oracle Life Sciences
- Veradigm
- OM1
- Datavant
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Data Source , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Linkage Method, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Data Source , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Linkage Method, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Data Source , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Linkage Method, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Data Source , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Linkage Method, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Data Source , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Linkage Method, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Data Source , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Linkage Method, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Data Source , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Linkage Method, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Data Source , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Linkage Method, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Data Source , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Data Source , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Data Source
- Figure 6: Global Market Value Share and BPS Analysis by Linkage Method, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Linkage Method, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Linkage Method
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Data Source , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Data Source , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Data Source
- Figure 26: North America Market Value Share and BPS Analysis by Linkage Method, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Linkage Method, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Linkage Method
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Data Source , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Data Source , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Data Source
- Figure 36: Latin America Market Value Share and BPS Analysis by Linkage Method, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Linkage Method, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Linkage Method
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Data Source , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Data Source , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Data Source
- Figure 46: Western Europe Market Value Share and BPS Analysis by Linkage Method, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Linkage Method, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Linkage Method
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Data Source , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Data Source , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Data Source
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Linkage Method, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Linkage Method, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Linkage Method
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Data Source , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Data Source , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Data Source
- Figure 66: East Asia Market Value Share and BPS Analysis by Linkage Method, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Linkage Method, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Linkage Method
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Data Source , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Data Source , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Data Source
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Linkage Method, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Linkage Method, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Linkage Method
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Data Source , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Data Source , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Data Source
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Linkage Method, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Linkage Method, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Linkage Method
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

