AI Colonoscopy Withdrawal Quality Monitoring System Market
The AI colonoscopy withdrawal quality monitoring system market is segmented by Component (Software engines, Edge modules, Display units, Data interfaces), Quality function (Withdrawal timing, Blind-spot review, Bowel scoring, Speed alerts, Report generation), Deployment (Tower-integrated, Standalone carts, Cloud platforms, Hybrid installs), End user (Hospitals, Ambulatory centers, GI clinics, Academic centers), Commercial model (Capital purchase, Subscription license, Per-procedure fee, Service contract), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
AI Colonoscopy Withdrawal Quality Monitoring System Market Size, Market Forecast and Outlook By FMI
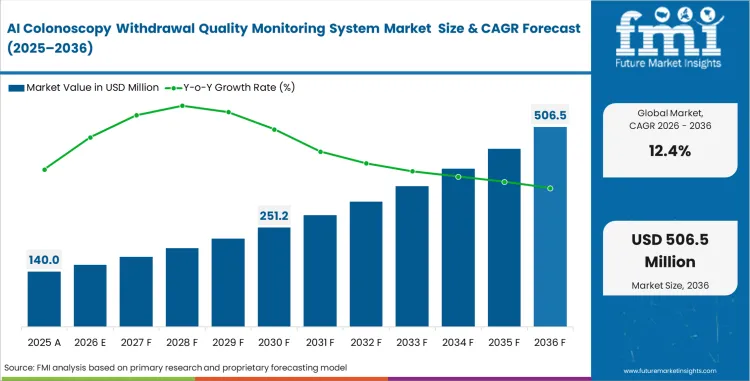
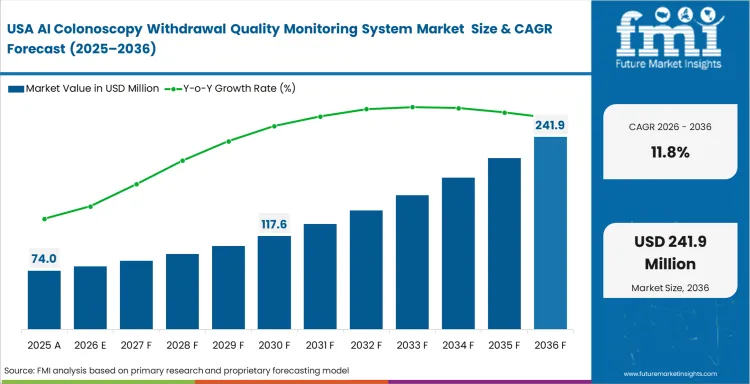
The AI colonoscopy withdrawal quality monitoring system market was valued at USD 124.6 million in 2025. Sector is expected to reach USD 140 million in 2026 at a CAGR of 12.4% during the forecast period. Regulatory mandates tying adenoma detection rates directly to physician reimbursement push total valuation to USD 450.6 million through 2036 as clinical accountability shifts from manual reporting to algorithmic verification.
Summary of AI Colonoscopy Withdrawal Quality Monitoring System Market
- The market is forecast to reach USD 450.6 million by 2036.
- The market is expected to grow at a CAGR of 12.4% from 2026 to 2036.
- The market was estimated at USD 124.6 million in 2025.
- The forecast period represents an incremental opportunity of USD 310.6 million.
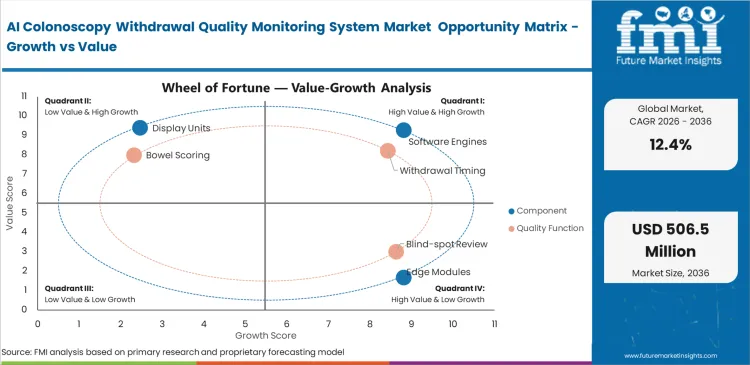
- Software engines lead the component segment with a 42% share, driven by algorithm-based differentiation and workflow analytics.
- Withdrawal timing dominates the quality function segment with a 34% share, aligned with clinical guideline compliance and physician performance metrics.
- Tower-integrated systems account for 46% share, reflecting hospital preference for seamless integration with existing endoscopy setups.
- Hospitals lead the end-user segment with a 49% share, supported by higher procedure volumes and centralized budget control.
- Capital purchase models hold 51% share, although hybrid and subscription-based models are gradually gaining traction.
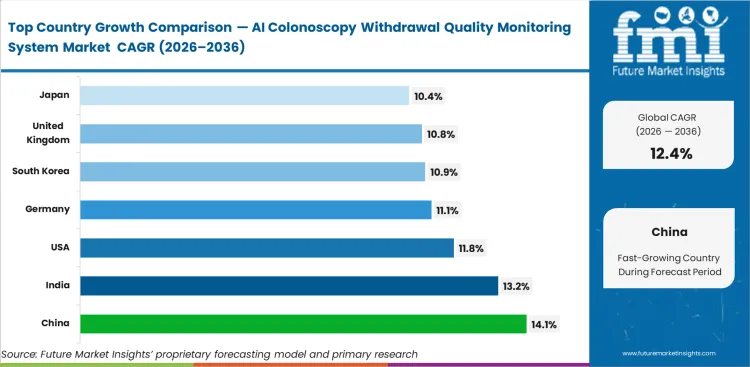
- China and India are the fastest-growing markets, while the United States remains the largest demand base.
- Market growth is driven by stricter clinical quality protocols, regulatory linkage of detection rates to reimbursement, and increasing adoption of AI-enabled real-time procedure monitoring.
AI Colonoscopy Withdrawal Quality Monitoring System Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 140 million |
| Industry Value (2036) | USD 450.6 million |
| CAGR (2026 to 2036) | 12.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Gastroenterology departments are under direct pressure to standardize inspection time during procedures. Clinical leadership must maintain documented compliance with the six-minute minimum withdrawal standard to protect full payer coverage and reduce audit exposure. Any shortfall against that benchmark raises the risk of reimbursement disputes and possible revenue recovery. Automated timing is reshaping how performance is recorded across the colonoscopy quality monitoring market because it removes dependence on manual timestamping and makes procedural records easier to validate. Endoscopy units upgrading their video systems are reducing documentation errors at the point of care. Delayed integration leaves facilities exposed where payer contracts are tied closely to measurable quality metrics.
Algorithm-based verification of withdrawal time is becoming the main trigger for wider adoption. Once reimbursement depends on automated quality scoring, manual reporting loses credibility as a durable operating model. Clinical networks are therefore shifting toward integrated analytics that can support both documentation accuracy and procedural consistency.
China is projected to expand at a CAGR of 14.1% through 2036 as large urban hospitals continue digitizing gastroenterology wards. In India, the sector is likely to post 13.2% CAGR over the forecast period, supported by rising screening volumes across corporate hospital networks. Strict Medicare-linked quality reporting keeps the United States on a positive trend, with the market expected to record 11.8% CAGR by 2036. Germany is forecast to register 11.1% CAGR as clinical upgrades remain closely tied to quality reporting and established hospital IT environments. South Korea follows at 10.9%, where high procedure intensity is accelerating adoption of systems that improve consistency across busy endoscopy units. In the United Kingdom, the industry outlook points to 10.8% CAGR through 2036 as providers work to improve throughput without weakening examination quality. Japan is expected to post 10.4% CAGR over the same period, with adoption advancing in line with established clinical workflows and incumbent imaging ecosystems. Funding gaps across regions continue to influence how quickly these upgrades move from evaluation to full clinical rollout.
Segmental Analysis
AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis by Component
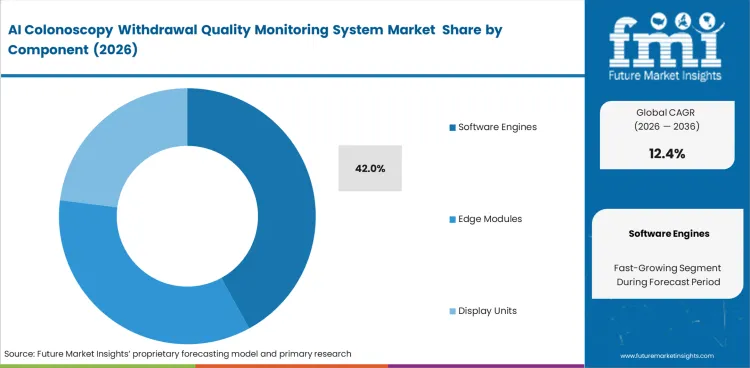
Integration flexibility determines why software engines are expected to account for 42.0% share in 2026. Medical centers operate mixed fleets of endoscopes acquired over multiple capital cycles. Clinical IT directors require colonoscopy withdrawal monitoring software that functions regardless of the underlying camera brand. This hardware-agnostic approach accelerates adoption across large consolidated health networks. Buyers locking themselves into proprietary hardware modules often face massive replacement costs when upgrading their endoscope reprocessing device setups. What the basic share percentage conceals is the rapid shift toward edge-computing architectures that process video feeds locally rather than sending sensitive patient data to external servers. Facilities relying entirely on cloud processing experience unacceptable lag times during live procedures, leading to immediate physician rejection.
- Cross-fleet compatibility: Algorithms must function cleanly across diverse camera resolutions. Clinical IT directors face severe implementation delays if the software requires specific proprietary inputs.
- Local processing prioritization: Edge modules analyze video frames without network dependency. Hospital administrators avoid latency issues that disrupt the physician's natural examination rhythm.
- Data format standardization: Output metrics must export easily into existing patient records. Billing managers lose critical reimbursement revenue if the system generates incompatible documentation.
AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis by Quality function
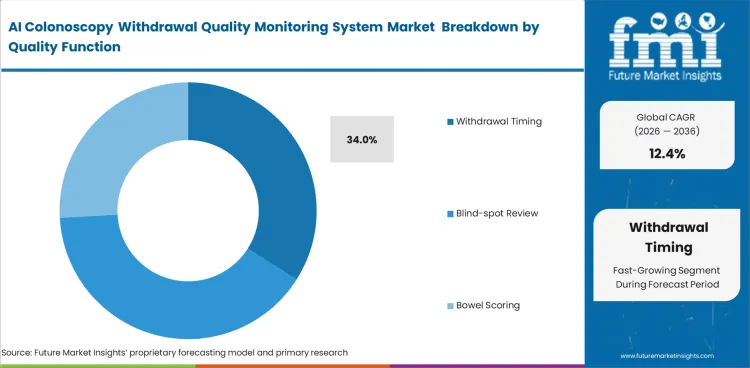
Clinical guidelines dictate why withdrawal timing is projected to secure 34.0% share in 2026. Medical societies universally recognize six minutes as the absolute minimum standard for a thorough mucosal inspection. Endoscopy department heads utilize effective withdrawal time AI to defend their departments against payer audits. The tools managing endoscopy fluid management systems act as secondary priorities compared to sheer withdrawal duration. Practitioners missing these time targets face immediate scrutiny from internal quality review boards. The underlying reality is that timing algorithms are significantly easier to train and validate than complex mucosal scoring models. Many providers are likely to prioritize timing and documentation functions first because they are easier to validate and fit into current workflows.
- Payer audit defense: Accurate timestamping proves clinical thoroughness. Endoscopy department heads secure full reimbursement rates by submitting algorithmic verification.
- Speed boundary alerts: Real-time warnings prevent physicians from rushing examinations. Clinical staff avoid the need to repeat procedures due to inadequate inspection times.
- Automated report generation: Documentation occurs simultaneously with the procedure. Nursing staff save hours of manual data entry while eliminating transcription errors.
AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis by Deployment
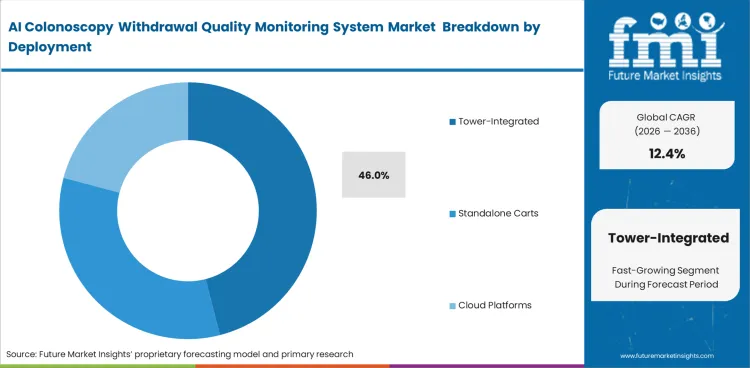
Procedure rooms are already crowded with essential life support and visualization equipment. Purchasing managers strictly prioritize solutions that occupy zero additional floor space. Space limitations inside clinical environments explain why tower-integrated systems are likely to represent 46.0% of the market in 2026. Forcing a standalone cart into a cramped esophagoscope and gastroscope suite creates physical hazards for the nursing staff. Standalone systems remain relevant only in older facilities lacking modern equipment racks. What general market reports ignore is the heavy reliance on proprietary video cables that complicate tower integration for third-party software vendors. Standalone systems remain relevant in legacy rooms, but added floor-space and workflow friction can limit appeal in higher-throughput sites.
- Zero-footprint integration: Modules mount directly onto existing equipment racks. Purchasing managers avoid redesigning procedure rooms to accommodate new technology.
- Direct cable routing: Integrated systems capture raw video feeds before display compression. Clinical technicians guarantee optimal image quality for the analytical engine.
- Power supply consolidation: Tower mounting reduces cord clutter on the clinical floor. Nursing staff avoid tripping hazards during critical patient interventions.
AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis by End user
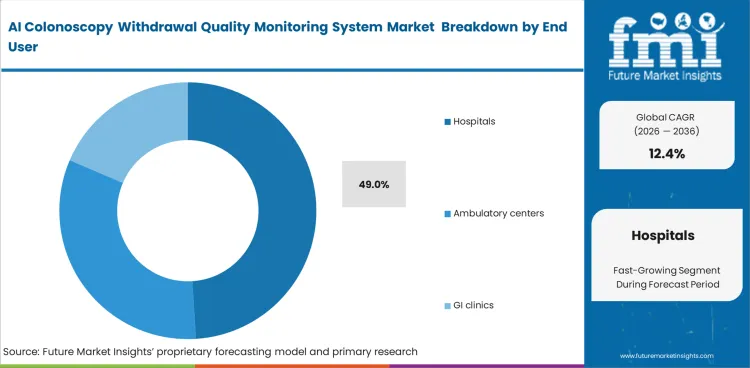
Procedural volume drives the commanding 49.0% share hospitals are forecast to represent in 2026. Massive medical centers conduct thousands of screenings annually, making hospital colonoscopy quality analytics an operational necessity. Chief medical officers rely on algorithmic oversight to manage rotating rosters of junior and senior gastroenterologists. According to estimations, smaller clinics using mobile endoscopic workstations lack the capital budgets for comprehensive fleet upgrades. Hospitals possess the financial resources to absorb the initial installation costs. The non-obvious factor shaping this segment is the role of academic research, as large teaching hospitals utilize these systems to train residents rather than just satisfying payer requirements. Clinics delaying adoption ultimately lose their competitive edge when marketing their quality metrics to local patient populations.
- Volume-driven standardization: High patient throughput requires automated oversight. Chief medical officers maintain consistent quality regardless of which physician performs the examination.
- Resident training applications: Algorithmic feedback accelerates the learning curve for new doctors. Academic directors provide objective performance data to medical trainees.
- Network-wide analytics: Multi-site hospitals aggregate data to identify underperforming departments. Executive boards allocate targeted training resources based on specific institutional weaknesses.
AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis by Commercial model
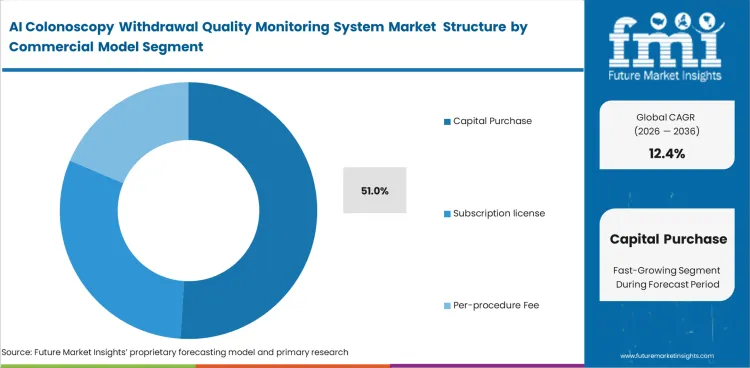
Large medical institutions prefer amortizing equipment costs over several years rather than managing ongoing operational expenses. Finance directors aggressively resist new software subscriptions that complicate their annual budget planning. The development toward recurring revenue models remains slow despite vendor pressure. Financial arrangements heavily favor capital purchase, which is expected to contribute 51.0% of total market share in 2026. Companies selling disposable endoscopes successfully use subscription models, but permanent capital equipment buyers operate differently. What vendors rarely admit is that hospitals purchasing outright often refuse to pay for subsequent algorithmic updates, leading to a fleet of rapidly aging software models. Facilities choosing subscriptions benefit from continuous neural network improvements but surrender long-term cost predictability.
- Budget predictability: Upfront purchases eliminate surprise operational costs. Finance directors secure capital expenditure approvals far easier than recurring software budgets.
- Update resistance: Capital buyers frequently skip optional software enhancements. Clinical IT managers face aging algorithms that eventually fall behind modern detection standards.
- Service contract bundling: Vendors recover recurring revenue through mandatory maintenance agreements. Purchasing committees must carefully evaluate total lifecycle costs before signing initial agreements.
AI Colonoscopy Withdrawal Quality Monitoring System Market Drivers, Restraints, and Opportunities
Quality reporting mandates established by national health insurers force clinical directors to adopt automated verification tools across the AI colonoscopy quality assurance market. Physicians can no longer rely on handwritten timestamps to prove they spent adequate time inspecting the colon. Providers are placing greater value on objective procedural documentation where reimbursement and audit scrutiny are linked to quality reporting. This financial threat compels hospital administrators to prioritize AI integrations over standard equipment upgrades. The integration of small bowel enteroscopes into related procedures further elevates the need for flawless initial mucosal inspections. Clinics without automated tracking may face a weaker documentation position in systems where quality reporting is becoming more formalized
Existing hospital IT configurations act as a massive operational barrier for new deployments. Endoscopy suites utilize legacy video processors that lack the necessary outputs to feed high-definition data into modern AI engines. Clinical IT directors refuse to approve installations that require manual workarounds or jeopardize patient data security alongside colorectal cancer molecular diagnostics records. Vendors must navigate months of cybersecurity reviews before their software is permitted to touch the hospital's internal network. This intense technical validation phase delays revenue realization for suppliers and frustrates clinical champions.
Opportunities in the AI Colonoscopy Withdrawal Quality Monitoring System Market
- Cloud-based aggregated analytics: Vendors providing secure multi-hospital performance benchmarking tools allow chief medical officers to compare their adenoma detection rates against national averages.
- Automated billing integration: Linking withdrawal scores directly to non-invasive colon cancer screening coding software allows billing managers to submit claims instantly.
- Real-time resident coaching: Developing specialized user interfaces tailored for teaching hospitals helps academic directors train junior physicians faster.
Regional Analysis
Based on regional analysis, AI colonoscopy withdrawal quality monitoring system market is segmented into North America, Europe, East Asia, and South Asia across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 14.1% |
| India | 13.2% |
| United States | 11.8% |
| Germany | 11.1% |
| South Korea | 10.9% |
| United Kingdom | 10.8% |
| Japan | 10.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
East Asia AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis
East Asia remains one of the most active regions for adoption because public funding for digital healthcare is feeding directly into hospital modernization. Large urban gastroenterology centers are under pressure to handle higher procedure volumes without allowing examination quality to slip, which makes withdrawal-quality monitoring tools operationally relevant rather than optional. Local technology companies are strengthening their position as they secure regulatory approvals faster and compete more aggressively on price than foreign brands. Precision cancer imaging is also becoming more closely tied to routine screening workflows, so hospitals are putting greater focus on systems that help physicians maintain examination discipline in busy clinical settings.
- China: China is projected to expand at a CAGR of 14.1% through 2036, supported by state-backed hospital modernization and the strong position of domestic vendors in local purchasing programs. Quality targets in leading cities are pushing gastroenterology departments to standardize withdrawal monitoring and reporting rather than leaving performance control to individual physician judgment. Provincial endorsements also matter in this market because they shape which software platforms move into wider hospital adoption.
- South Korea: National screening intensity keeps endoscopy units under constant throughput pressure, and that is lifting interest in tools that help reduce variation between procedures. Clinical teams are using AI timers and monitoring layers to keep daily schedules on track while protecting examination quality. South Korea is anticipated to move ahead at a CAGR of 10.9% during the assessment period. Hospitals upgrading these systems are doing so to improve consistency across busy endoscopy lists.
- Japan: Japan’s installed base, local manufacturing strength, and long-standing OEM relationships continue to shape software adoption patterns. The industry outlook in Japan points to 10.4% CAGR through 2036. Hospitals generally prefer solutions aligned with domestic imaging ecosystems, which gives native developers a practical advantage in procurement discussions. Foreign software firms still face a difficult competitive path because local imaging companies remain deeply embedded in clinical workflows.
FMI's report includes broader regional assessments across East Asia. Government technology subsidies strongly influence which specific digital healthcare software architectures gain initial clinical approval.
South Asia AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis
South Asia is being led primarily by private hospital expansion rather than broad public-sector technology adoption. Large hospital groups in metropolitan areas are using advanced AI capability as part of premium service positioning, especially where international patients and higher-acuity care pathways matter. Public facilities remain more constrained by capital availability, which limits the pace of adoption outside top-tier private networks. Reliability, installation ease, and pricing flexibility carry more weight in this region than technical sophistication alone.
- India: Private hospital groups are taking the lead in adopting algorithm-based monitoring to support standardized colonoscopy practice across multi-site networks. Medical tourism also strengthens the case for these systems, as providers use digital quality oversight to reinforce confidence in procedural standards. Vendors that offer flexible subscription models are better placed to expand in a sector where cost discipline still shapes most buying decisions. India is likely to post 13.2% CAGR in this market by 2036. This keeps the country ahead of most mature healthcare markets in the sector.
FMI’s report includes adjacent markets across South Asia. Pricing flexibility is likely to influence market penetration more than incremental technical differentiation in this region.
North America AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis
North America is shaped less by technology novelty and more by documentation, reimbursement, and system integration requirements. Healthcare providers operate in an environment where withdrawal quality, adenoma detection performance, and audit-ready reporting carry direct commercial importance, so automated monitoring tools are becoming more relevant to routine department management. Hospitals are under steady pressure to reduce documentation risk and tighten quality oversight. Integration with electronic health record infrastructure is also a basic requirement for vendors that want access to large health system contracts.
- United States: Reimbursement discipline and audit exposure are pushing hospitals and endoscopy centers to tighten procedure documentation standards. The United States market is expected to record 11.8% CAGR during the forecast period. Facilities want systems that reduce dependence on manual reporting and create a cleaner digital record around withdrawal quality and related performance measures. Sites that implement automated reporting are doing so to reduce administrative error and support more consistent compliance.
FMI’s report also covers Canadian provincial healthcare networks. Reimbursement framework and budget timing continue to shape when purchasing decisions move forward across North America.
Europe AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis
Europe’s direction is being set by screening-quality mandates and data-governance requirements. Public health systems want stronger procedural consistency, but tender-led buying cycles tend to slow implementation when compared with more commercially driven healthcare markets. Hospitals are giving greater attention to tools that combine video analytics with quality-control functions while still fitting local privacy rules. Local data handling and compliance alignment remain central to vendor selection across the region.
- Germany: German clinics benefit from reimbursement models that support higher spending on advanced procedural tools, especially where workflow integration is already well established. Endoscopy departments place greater value on systems that connect smoothly with registry and documentation platforms used in routine clinical practice. The market in Germany is forecast to register a CAGR of 11.1% over the study period. Vendors that satisfy strict regional privacy and data-handling expectations are in a better position to build durable access.
- United Kingdom: The United Kingdom market is anticipated to move ahead at a CAGR of 10.8% through 2036. Screening backlogs and performance variation across clinical sites continue to shape adoption priorities. NHS-linked providers are using AI-based monitoring to bring more consistency to colonoscopy practice without allowing speed targets to weaken examination quality. Wider use of these tools is helping shift performance review from subjective assessment toward measurable procedural control.
FMI's report includes Nordic and Southern European territories. Disparities in public health funding directly control the speed of clinical endoscopy visualization systems and components digitization across these borders.
Competitive Aligners for Market Players
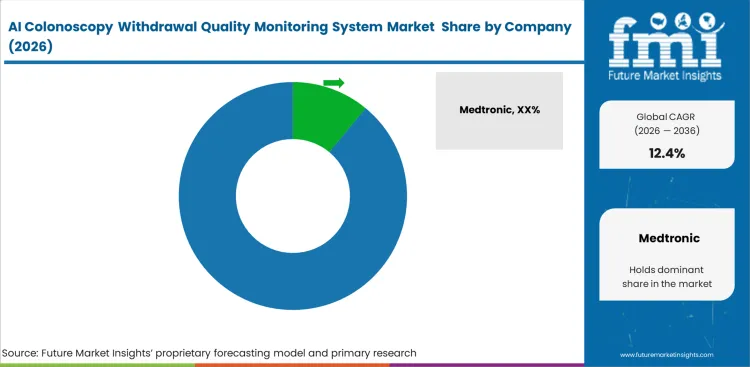
Incumbent medical imaging giants utilize their massive installed hardware base to control the initial software rollout. Companies like Olympus and FUJIFILM offer proprietary algorithms designed specifically to optimize their own optical feeds. They lock hospital networks into closed ecosystems where third-party software faces severe compatibility hurdles. Clinical IT directors naturally gravitate toward native software updates rather than risking technical conflicts with external ai enabled medical devices. This hardware dominance forces independent software developers to compete purely on analytical superiority and cross-platform flexibility.
Independent developers like MAGENTIQ EYE, Wuhan ENDOANGEL, and Chengdu Wision counter this advantage by offering deeply agnostic artificial intelligence in healthcare modules. They build capabilities that interface cleanly with older, mixed-brand equipment racks. Hospital administrators managing tight capital budgets vastly prefer software that functions on legacy endoscopes over financing entirely new visualization towers. These challengers secure their position by proving their algorithms can match or exceed the accuracy of proprietary manufacturer software without requiring a total hardware refresh.
Large medical networks aggressively combat vendor lock-in by mandating open-architecture data standards in their purchasing tenders. Chief medical informatics officers refuse to sign contracts that trap their clinical data inside closed proprietary formats. They explicitly require systems that export raw performance metrics into central hospital data lakes. Moving toward 2036, software vendors who integrate smoothly with generalized healthcare ai computer vision platforms will capture the most lucrative enterprise-level hospital contracts.
Key Players in AI Colonoscopy Withdrawal Quality Monitoring System Market
- Medtronic
- Olympus
- FUJIFILM
- Wuhan ENDOANGEL Medical Technology
- Chengdu Wision Medical Device
- NEC Corporation
- MAGENTIQ EYE
Scope of the Report
| Metric | Value |
|---|---|
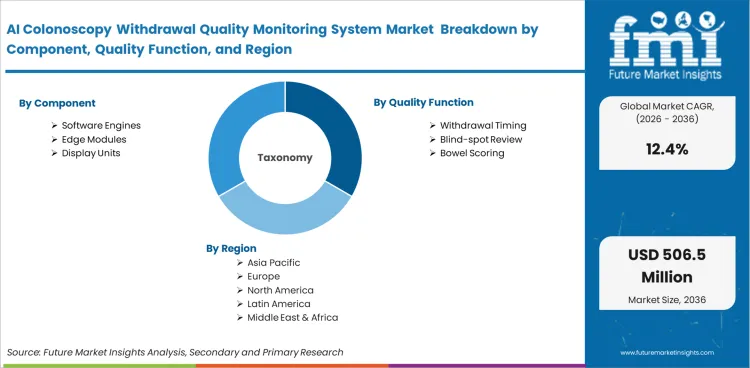
| Quantitative Units | USD 140 million in 2026 to USD 450.6 million by 2036, at a CAGR of 12.4% |
| Market Definition | AI colonoscopy withdrawal quality monitoring systems are specialized algorithmic platforms designed to track, measure, and score the mucosal inspection phase of lower gastrointestinal examinations. These platforms analyze real-time video feeds to ensure physicians spend adequate time examining the colon lining while actively identifying potential missed areas. They function as automated compliance engines verifying medical thoroughness. |
| Segmentation | By Component, By Quality function, By Deployment, By End user, By Commercial model, By Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Brazil, Mexico, Germany, United Kingdom, France, Spain, Italy, China, Japan, South Korea, India, Australia, New Zealand, GCC, South Africa |
| Key Companies Profiled | Medtronic, Olympus, FUJIFILM, Wuhan ENDOANGEL Medical Technology, Chengdu Wision Medical Device, NEC Corporation, MAGENTIQ EYE |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews with endoscopy suite directors combined with clinical installation volume tracking |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
AI Colonoscopy Withdrawal Quality Monitoring System Market Analysis by Segments
By Component:
- Software engines
- Edge modules
- Display units
- Data interfaces
By Quality function:
- Withdrawal timing
- Blind-spot review
- Bowel scoring
- Speed alerts
- Report generation
By Deployment:
- Tower-integrated
- Standalone carts
- Cloud platforms
- Hybrid installs
By End user:
- Hospitals
- Ambulatory centers
- GI clinics
- Academic centers
By Commercial model:
- Capital purchase
- Subscription license
- Per-procedure fee
- Service contract
By Region:
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- East Asia
- China
- Japan
- South Korea
- South Asia
- India
- ASEAN
- Oceania
- Australia
- New Zealand
- Middle East and Africa
- GCC
- South Africa
Bibliography
- Dimopoulou, K., Antonakaki, D., Delis, S., Fragkou, P., & Archimandritis, A. (2025). Artificial intelligence in colonoscopy: Enhancing quality and preventing post-colonoscopy colorectal cancer. World Journal of Gastrointestinal Endoscopy, 17(1), 101391.
- King, S. C., King, J., Thomas, C. C., & Richardson, L. C. (2025). Baseline estimates of colorectal cancer screening among adults aged 45 to 75 years, Behavioral Risk Factor Surveillance System, 2022. Preventing Chronic Disease, 22, 250175.
- Koh, F. H., Ku, M. S. Y., Chan, W. W. Y., et al. (2025). Asia-Pacific consensus on the use of artificial intelligence in gastrointestinal endoscopy. iGIE.
- Lahr, R. E., Wallace, K., Singh, V., et al. (2025). Inspection time during colonoscopy withdrawal as measured by artificial intelligence. Gastrointestinal Endoscopy.
- Maan, S., & Rex, D. K. (2025). Artificial intelligence in endoscopy quality measures. Gastrointestinal Endoscopy Clinics of North America.
- Mori, Y., East, J. E., Rex, D. K., & Neumann, H. (2025). Quality assessment in endoscopy: Artificial intelligence in quality assurance, detection, characterization, and workflow optimization. Best Practice & Research Clinical Gastroenterology.
- Rabba, W., Alhaj Moustafa, M., Al Assar, O., et al. (2025). Artificial intelligence in colonoscopy: A systematic review of efficacy and performance. Cureus, 17(1).
- Rex, D. K., Schoenfeld, P. S., Cohen, J., et al. (2024). Quality indicators for colonoscopy. Gastrointestinal Endoscopy.
- Sabatino, S. A., Thompson, T. D., & White, M. C. (2025). Use of cancer screening tests, United States, 2023. Preventing Chronic Disease, 22.
- World Health Organization, International Agency for Research on Cancer. (2025). Performance of colorectal cancer screening in the European Union Member States: Data from the second European screening report.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Regulatory mandates tying adenoma detection rates to specific physician reimbursement models.
- Hardware constraints affecting the deployment of edge modules in crowded clinical settings.
- Vendor competition between native imaging algorithms and hardware-agnostic software solutions.
- Budgetary pressures favoring capital purchases over recurring software licensing agreements.
- Privacy requirements dictating local video processing over remote cloud analytics.
- Physician resistance to automated clinical surveillance and examination timing constraints.
- The operational divide between high-volume urban hospitals and smaller ambulatory centers.
- The specific technical requirements for integrating algorithmic data into electronic health records.
Frequently Asked Questions
What is the AI in colonoscopy market size expectation for 2036?
Revenue reaches USD 450.6 million by 2036 as clinical directors view automated quality monitoring as a mandatory operational requirement.
Which component segment captures the most share?
Software engines command 42.0% share in 2026 because they run across diverse fleets of existing endoscopes without replacing hardware.
What is AI quality assurance in colonoscopy designed to solve?
These systems provide objective proof that physicians spend adequate time inspecting the colon lining to satisfy payer reimbursement guidelines.
How does AI measure withdrawal time in colonoscopy?
Algorithms analyze real-time video feeds to calculate exact mucosal examination duration, automatically deducting time spent cleaning or pausing.
Which deployment strategy is most prominent?
Tower-integrated systems capture 46.0% share because zero-footprint solutions preserve critical space inside already crowded endoscopy procedure rooms.
What is the fastest-growing region?
China accelerates at a 14.1% CAGR as massive government initiatives fund technological upgrades within large urban medical centers.
Why do hospitals dominate the end user base?
Hospitals retain 49.0% share because chief medical officers require automated oversight to maintain quality across large rotating physician teams.
Why do capital purchases outpace subscription models?
Capital purchases secure 51.0% share because finance directors prefer amortizing equipment costs over managing recurring operational expenses annually.
How does India compare to the United States in growth?
India expands at 13.2% driven by medical tourism, while the United States tracks at 11.8% based on Medicare compliance.
What creates the primary operational friction?
Legacy hospital IT networks limit deployment speed because clinical informatics directors refuse high-bandwidth processors that jeopardize patient data security.
Who are the leading companies in AI colonoscopy quality systems?
Medtronic, Olympus, and FUJIFILM dominate integrated hardware, while Wuhan ENDOANGEL and MAGENTIQ EYE compete heavily in hardware-agnostic software.
What is the core clinical consequence of delayed adoption?
Facilities relying on manual timestamping face increasing claim rejections because payers demand objective algorithmic proof before authorizing full reimbursements.
Explain the AI colonoscopy withdrawal quality monitoring market dynamic in Europe?
The United Kingdom relies on centralized National Health Service mandates, whereas Germany uses high procedural reimbursement rates to fund tools.
Why do edge modules matter for clinical workflow?
Edge computing processes video locally without network latency, giving endoscopists the instantaneous feedback required during critical live diagnostic moments.
What role do academic centers play in software adoption?
Large teaching hospitals utilize algorithmic feedback to train resident physicians, relying on objective performance data rather than subjective peer observation.
Can AI improve colonoscopy withdrawal quality regarding missed areas?
Blind-spot systems map colon geometry in real-time to alert physicians of unexamined tissue folds before they conclude the procedure.
Why do standalone carts remain relevant?
Older medical facilities lacking modern integrated equipment racks still require algorithmic monitoring, making mobile systems a necessary physical alternative.
How do vendors address the capital purchase preference?
Software developers bundle mandatory maintenance contracts with initial hardware sales to capture recurring revenue despite resistance to direct software subscriptions.
What happens when physicians fail to meet the ASGE colonoscopy withdrawal time AI threshold?
Algorithms flag the truncated procedure in electronic health records, forcing department heads to review cases and potentially order repeat examinations.
Why is data integration so crucial for billing managers?
Algorithmic scores must translate seamlessly into standardized billing codes to prevent nursing staff from performing hours of manual data entry.
How do you compare colonoscopy CADe and CAQ systems?
CADe systems exclusively detect polyps, whereas CAQ systems measure procedural mechanics by tracking withdrawal speed and unexamined mucosal surface areas.
What is the primary technical limitation of current computer vision models?
Algorithms struggle during poor bowel preparation because excessive fluid or debris completely neutralizes the software's ability to maintain analytical accuracy.
How do patient privacy laws shape regional deployments?
European regulations force vendors to disable cloud-based storage, guaranteeing that all patient video remains strictly within localized hospital server networks.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Component
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Component , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Component , 2026 to 2036
- Software Engines
- Edge Modules
- Display Value (USD Million)s
- Software Engines
- Y to o to Y Growth Trend Analysis By Component , 2021 to 2025
- Absolute $ Opportunity Analysis By Component , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Quality Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Quality Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Quality Function, 2026 to 2036
- Withdrawal Timing
- Blind-spot Review
- Bowel Scoring
- Withdrawal Timing
- Y to o to Y Growth Trend Analysis By Quality Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Quality Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- Tower-Integrated
- Standalone Carts
- Cloud Platforms
- Tower-Integrated
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Ambulatory centers
- GI clinics
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Commercial Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Commercial Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Commercial Model, 2026 to 2036
- Capital Purchase
- Subscription license
- Per-procedure Fee
- Capital Purchase
- Y to o to Y Growth Trend Analysis By Commercial Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Commercial Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Component
- By Quality Function
- By Deployment
- By End User
- By Commercial Model
- Competition Analysis
- Competition Deep Dive
- Medtronic
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Olympus
- FUJIFILM
- Wuhan ENDOANGEL Medical Technology
- Chengdu Wision Medical Device
- NEC Corporation
- Medtronic
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Quality Function, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Commercial Model, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Quality Function, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Commercial Model, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Quality Function, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Commercial Model, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Quality Function, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Commercial Model, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Quality Function, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Commercial Model, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Quality Function, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Commercial Model, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Quality Function, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Commercial Model, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Quality Function, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Commercial Model, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Component
- Figure 6: Global Market Value Share and BPS Analysis by Quality Function, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Quality Function, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Quality Function
- Figure 9: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Deployment
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Commercial Model, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Commercial Model, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Commercial Model
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Component
- Figure 32: North America Market Value Share and BPS Analysis by Quality Function, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Quality Function, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Quality Function
- Figure 35: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Deployment
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Commercial Model, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Commercial Model, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Commercial Model
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Component
- Figure 48: Latin America Market Value Share and BPS Analysis by Quality Function, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Quality Function, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Quality Function
- Figure 51: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Deployment
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Commercial Model, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Commercial Model, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Commercial Model
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Component
- Figure 64: Western Europe Market Value Share and BPS Analysis by Quality Function, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Quality Function, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Quality Function
- Figure 67: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Deployment
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Commercial Model, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Commercial Model, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Commercial Model
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Component
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Quality Function, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Quality Function, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Quality Function
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Commercial Model, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Commercial Model, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Commercial Model
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Component
- Figure 96: East Asia Market Value Share and BPS Analysis by Quality Function, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Quality Function, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Quality Function
- Figure 99: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Deployment
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Commercial Model, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Commercial Model, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Commercial Model
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Component
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Quality Function, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Quality Function, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Quality Function
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Commercial Model, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Commercial Model, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Commercial Model
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Component
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Quality Function, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Quality Function, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Quality Function
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Commercial Model, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Commercial Model, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Commercial Model
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

