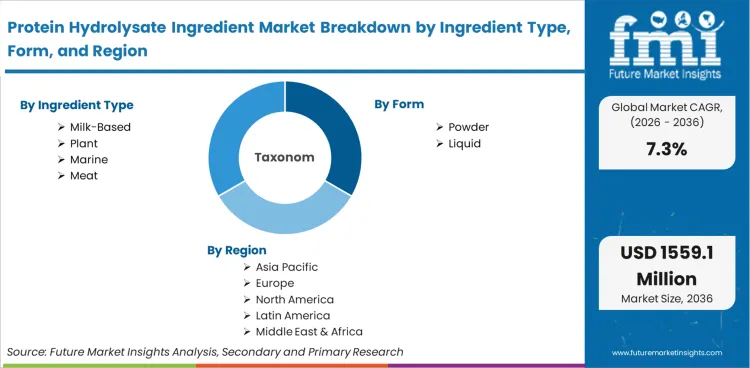
Protein Hydrolysate Ingredient Market
Protein Hydrolysate Ingredient Market is segmented by Ingredient Type (Milk, Plant, Marine, Meat), Form (Powder, Liquid), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
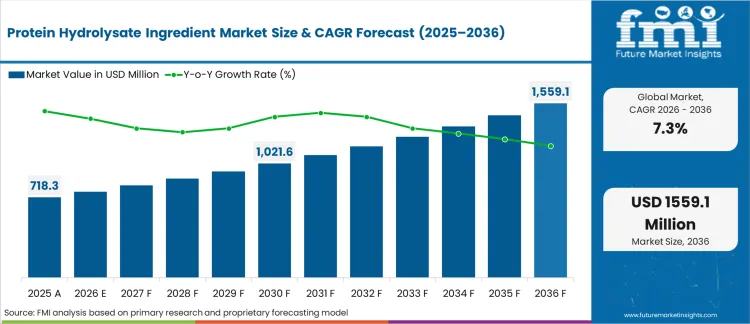
Protein Hydrolysate Ingredient Market Size, Forecast and Outlook By FMI
The global protein hydrolysate ingredient industry generated USD 718.2 million in 2025. According to Future Market Insights, revenues expand to USD 770.7 million in 2026 and advance to USD 1,559.1 million by 2036. The ten-year outlook reflects a CAGR of 7.3% between 2026 and 2036, driven by expanding clinical nutrition mandates, increasing demand for hypoallergenic infant formulations, and growing integration of rapidly absorbable peptide fractions across medical and performance nutrition applications.
Sourcing parameters are shifting away from generic protein concentrates toward highly functionalized variants. Buyers now mandate tighter molecular weight distribution thresholds, driven by an upstream supply base that produced 495 million pounds of whey protein concentrate across American facilities during 2024. [1] Massive raw material availability forces manufacturers to differentiate via enzymatic hydrolysis.
Luis Cubel, Group Vice President and Managing Director of Arla Foods Ingredients, said: “Our acquisition of Volac Whey Nutrition brings together two major manufacturers of whey ingredients, and consolidates our position as a leader in the space. Having Volac’s experts, and its network of trusted supply partners, as part of our team gives us several strategic advantages. It expands our market reach, helping us serve even more customers, and strengthens our global supply chain at a time when demand for whey is growing.” [2]
Summary of Protein Hydrolysate Ingredient Market
- Protein Hydrolysate Ingredient Market Definition
- Sector encompasses enzymatically cleaved protein ingredients engineered for enhanced digestibility and hypoallergenic performance.
- Demand Drivers in Market
- Clinical malnutrition intervention protocols necessitate easily absorbed peptide solutions.
- Rising pediatric food allergy prevalence forces formulation shifts toward specialized ingredients.
- Sports nutrition premiumization creates demand for rapid-recovery protein variants.
- Key Segments Analyzed in FMI Report
- Ingredient Type covers milk, plant, marine, and meat fractions.
- Form analyzes powder versus liquid commercial formats.
- Geography spans major global economic zones.
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage at Future Market Insights, opines, “In the updated Protein Hydrolysate Ingredient Market report for 2026 to 2036, my team and I have evaluated how clinical nutrition and performance recovery trends are elevating demand for highly bioavailable protein formats. Hydrolysates are no longer niche sports ingredients; they are becoming core components in medical nutrition and elderly care formulations. Manufacturers that optimize peptide profiling and bitterness control will gain strategic advantage in premium therapeutic applications.”
- Strategic Implications/Executive Takeaways
- Formulators must secure raw material access through vertical integration or strategic partnerships.
- Manufacturers face pressure to mask bitter flavor profiles inherent to extensive hydrolysis.
- Regulatory compliance capabilities dictate competitive survival in lucrative infant nutrition categories.
- Methodology
- Validated through first-hand corporate production and capacity data.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable industrial benchmarks and verified sources.
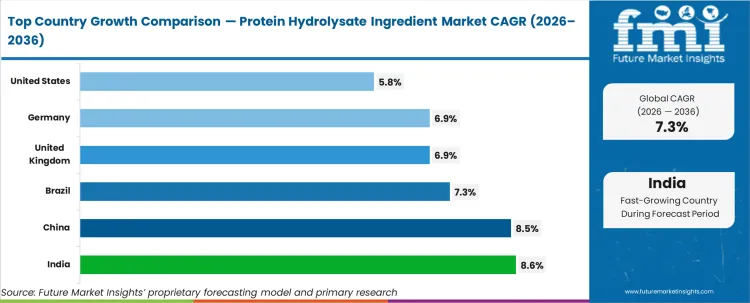
Demand across American channels tracks a 5.8% CAGR, while British and German adoption follows at 6.9% respectively. Expansion in India hits 8.6% and Chinese demand registers 8.5%, fueled by extensive dairy infrastructure investments. Brazilian trajectory supports global metrics with a 7.3% expansion anchored in active nutrition uptake.
Protein Hydrolysate Ingredient Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 770.7 Million |
| Industry Value (2036) | USD 1,559.1 Million |
| CAGR (2026-2036) | 7.3% |
Protein Hydrolysate Ingredient Market Definition
Scope covers partially or extensively digested protein fractions utilizing enzymatic breakdown processes. Specialized ingredients provide accelerated absorption profiles and reduced allergenicity compared to intact proteins. Commercial applications span infant formula, clinical nutrition, and sports supplements.
Protein Hydrolysate Ingredient Market Inclusions
Coverage incorporates revenues generated from selling bulk hydrolysate powders and liquids to food or beverage formulators. Revenue models encompass sales of protein hydrolysate fractions derived from dairy, plant, marine, and meat sources.
Protein Hydrolysate Ingredient Market Exclusions
Analysis omits retail sales of finished consumer products like ready-to-drink shakes or nutritional bars. Amino acid isolates and intact whey protein concentrates fall outside defined reporting boundaries.
Protein Hydrolysate Ingredient Market Research Methodology
- Primary Research: Direct interviews with specialized dairy processors and ingredient procurement heads validate commercial contract pricing.
- Desk Research: Aggregate tracking of global milk pool statistics and customs data maps raw material trade flows.
- Market-Sizing and Forecasting: Volume-based modeling correlates hospital malnutrition rates with clinical formula manufacturing outputs.
- Data Validation and Update Cycle: Internal cross-referencing against verified corporate capacity disclosures ensures ongoing baseline accuracy
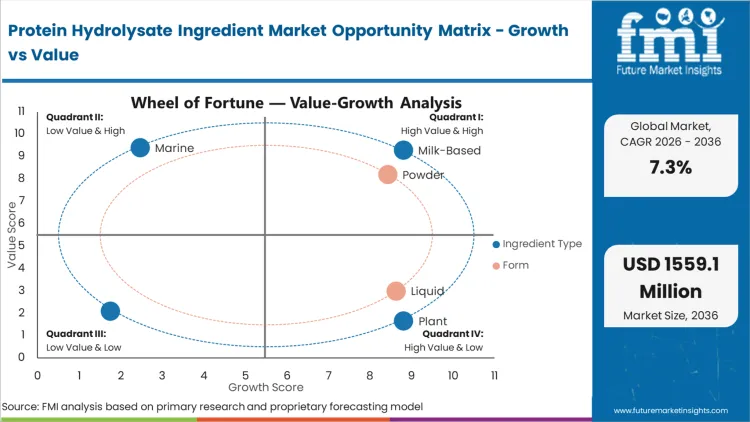
Segmental Analysis
Protein Hydrolysate Ingredient Market Analysis by Ingredient Type
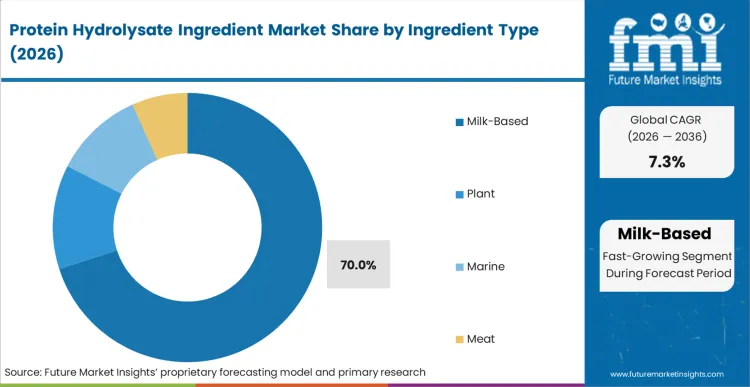
As per FMI's projection, milk-based variants dominate commercial consumption, capturing a 70% share during 2026. Dominance stems from deeply entrenched dairy supply chains and superior amino acid profiles suitable for critical nutrition segments. Raw whey availability provides necessary scale for massive industrial hydrolysis operations. Value creation happens through precise enzymatic control. Advanced technologies permit creation of highly specialized medical nutrition inputs. Arla Foods Ingredients recently launched Lacprodan DI-3092, targeting peptide-based medical formulas. [3] Suppliers commanding advanced technological capabilities dictate premium contract pricing.
- Supply-side move: Production expands into specialized therapeutic categories to capture premium margins. Upgraded purification technologies strip unwanted intact proteins from final outputs. Medical formulators integrate highly refined fractions to meet strict allergen thresholds.
- Buyer priority: Formulators seek ingredients offering balanced amino acid delivery without overwhelming bitter notes. Advanced filtration techniques mitigate off-flavors traditionally associated with extensive breakdown. Better tasting inputs reduce masking agent requirements for finished clinical products.
- Technology shift: Manufacturing processes pivot toward specialized enzymatic blends designed for specific cleavage patterns. Precision processing yields consistent molecular weight profiles across production batches. Predictable performance secures long-term procurement loyalty from major infant formula brands.
Protein Hydrolysate Ingredient Market Analysis by Form
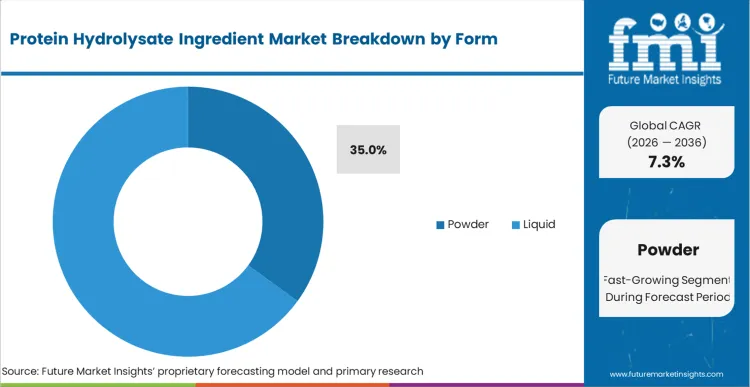
According to FMI's estimates, powder formats hold a 35% stake entering 2026, prized for logistical efficiency and superior shelf stability. Removal of moisture during spray drying protects fragile peptide bonds from premature degradation. Globalized supply chains depend heavily on dry formats to survive extended transit times. Plant-based innovations frequently utilize dry processing techniques to enter new application spaces. Expansion into plant-based arenas diversifies revenue streams away from volatile dairy commodities.
- Procurement trigger: Manufacturers prefer dry formats to minimize freight expenses and extend inventory viability. Moisture elimination prevents microbial proliferation during extended warehouse storage. Factories achieve superior operational flexibility when handling stable dry bulk ingredients.
- Formulation impact: Dry plant protein hydrolysate blends integrate seamlessly into pressed nutritional bars and baked applications. Improved solubility characteristics prevent gritty textures in high-protein consumer goods. Better mouthfeel directly supports repeat consumer purchasing behavior.
- Operational mechanism: Spray drying infrastructure requires massive capital outlay but yields highly consistent particle sizes. Uniform powder density ensures accurate dosing during automated batch mixing procedures. Precision manufacturing reduces costly raw material waste for downstream brands.
Protein Hydrolysate Ingredient Market Drivers, Restraints, and Opportunities
FMI analysts opine that rising clinical malnutrition rates heavily influence peptide procurement strategies. Hospitals face mounting pressure to accelerate patient recovery timelines through targeted nutritional intervention. Partially digested proteins require minimal gastrointestinal processing, ensuring rapid amino acid absorption in compromised patients. Procurement channels prioritize specialized fractions to formulate high-density enteral feeding solutions. Global health burdens dictate sustained baseline demand. Hospital malnutrition risk prevalence reached 11.6% overall during a recent 2025 study. [5] Ongoing clinical requirements shield specialized ingredient suppliers from macroeconomic volatility.
As per FMI's projection, severe flavor degradation presents a formidable barrier to mass consumer adoption. Extensive enzymatic cleavage inevitably exposes hydrophobic amino acids, creating intensely bitter tasting profiles. Formulators struggle to mask off-notes without introducing excessive sugar or artificial sweeteners. Downstream brands reject highly functional ingredients if consumer sensory trials fail. Manufacturers combat flavor challenges by utilizing selective exopeptidases to clip bitter terminals, though advanced processing significantly inflates production costs. High commercial pricing restricts extensive peptide adoption primarily to high-margin infant and medical categories. Mainstream milk protein hydrolysate applications require careful balance between functional benefits and palatable consumer experience.
- Targeted clinical application: Aging populations require accessible nutrition to combat degenerative muscle loss. Manufacturers formulate specific peptide profiles designed to stimulate rapid muscle synthesis in older demographics. Suppliers capturing early demographic shifts secure long-term revenue streams.
- Allergy management expansion: Rising pediatric food sensitivities force complete reformulation of standard milk recipes. Hydrolysis breaks problematic epitopes into unrecognized peptide fragments. Specialized processing secures lucrative contracts with multinational infant nutrition brands.
- Alternative source diversification: Darling Ingredients recently introduced Nextida, a targeted collagen peptide platform. [6] Marine and meat fractions provide novel functional benefits distinct from traditional dairy sources. Alternative base materials reduce exposure to fluctuating raw milk commodity pricing.
Regional Analysis
Based on regional analysis, protein hydrolysate ingredient market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. Full report offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 8.6% |
| China | 8.5% |
| Brazil | 7.3% |
| United Kingdom | 6.9% |
| Germany | 6.9% |
| United States | 5.8% |
Source: Future Market Insights analysis
North America Protein Hydrolysate Ingredient Market Analysis
American demand centers on specialized clinical formulations designed to manage widespread dietary sensitivities. High baseline allergy rates compel vast manufacturing infrastructure dedicated to hypoallergenic formula production. Regulatory scrutiny mandates stringent quality control across domestic manufacturing sites. Strict compliance protocols favor massive established ingredient suppliers capable of funding advanced testing capabilities. Centers for Disease Control and Prevention statistics confirm approximately 7% of American adults currently manage food allergies. [7] High prevalence rates guarantee sustained offtake for extensively processed peptide fractions.
- United States: Growth tracks a 5.8% CAGR through 2036, supported by dominant clinical nutrition networks demanding specialized peptide inputs. Extensive domestic processing infrastructure seamlessly converts raw whey into high-value medical ingredients. Formulators prioritize local supply chains to avoid volatile international freight risks.
FMI’s report includes detailed analysis covering regional developments across America. Canada presents distinct opportunities for advanced casein hydrolysate manufacturing given robust local dairy quotas. Mexico serves as an emerging production hub for cost-effective plant-based variants targeting regional food processors. Export-oriented upgrading remains a dominant theme across opportunistic zones.
Europe Protein Hydrolysate Ingredient Market Analysis
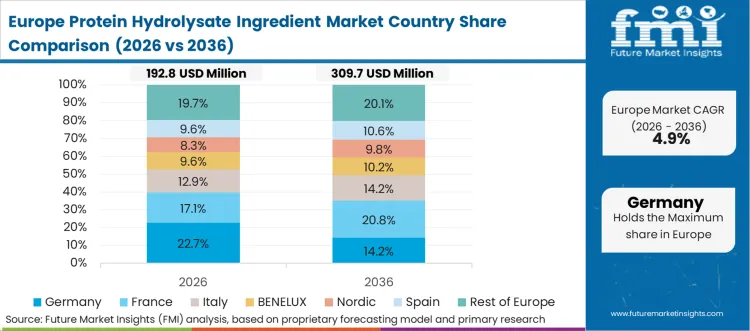
FMI analysts opine that European market dynamics favor high-end infant formula production and strict regulatory adherence. Formulators must navigate complex approval pathways to market specialized nutrition products across member states. High technical barriers prevent low-cost overseas ingredients from penetrating core European supply chains. Established regional cooperatives control massive raw milk pools, channeling premium whey directly into dedicated hydrolysis facilities. Arla recently noted a firm underlying industry trend prioritizing healthy nutritious products across European operations. [8] Rising consumer awareness accelerates transition away from intact concentrates toward functionalized peptide formulations.
- United Kingdom: A 6.9% CAGR reflects increasing domestic reliance on specialized medical nutrition interventions for aging demographics. Local formulators demand highly soluble powders to integrate into ready-to-drink clinical supplements. Rising raw material costs force British manufacturers to extract maximum value via precision enzymatic processing.
- Germany: Expansion at 6.9% CAGR is linked to massive pediatric nutrition exports bound for Asian territories. German factories maintain incredibly strict microbiological standards required for early-life applications. Premium global pricing allows local producers to fund continuous refinement of advanced filtration technologies.
FMI’s report includes extensive coverage across diverse European manufacturing landscapes. France provides critical whey volumes essential for continental ingredient scaling. Italy demonstrates rising demand for specialized meat peptides utilized in premium sports recovery formulas. Strict regional food safety directives heavily influence local procurement behavior regarding infant nutrition hydrolysate ingredients.
Asia Pacific Protein Hydrolysate Ingredient Market Analysis
As per FMI's projection, regional expansion relies on rapid modernization of domestic dairy processing infrastructure. Governments aggressively incentivize local production to reduce reliance on expensive imported infant formula ingredients. Rising middle-class purchasing power fuels unprecedented demand for premium pediatric and performance nutrition products. Local manufacturers struggle to master complex hydrolysis chemistry, creating massive import opportunities for Western ingredient specialists. Joint ventures frequently bridge critical knowledge gaps between foreign technology and local distribution networks. Evolving regulatory frameworks attempt to standardize domestic peptide quality.
- China: Growth tracks an 8.5% CAGR as domestic infant formula brands aggressively upgrade product specifications. Tighter national food safety standards force local formulators to utilize highly purified imported peptide fractions. Strategic alliances secure necessary volume access while mitigating cross-border trade friction.
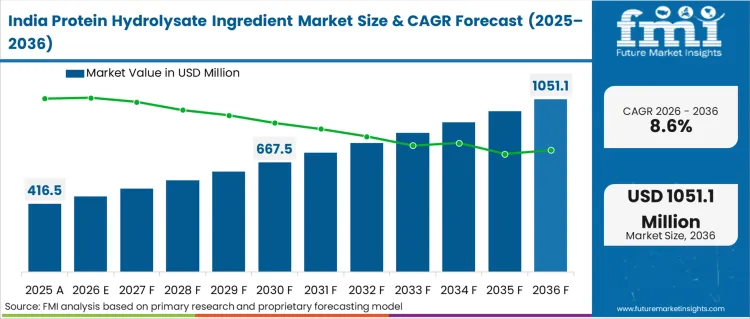
- India: At 8.6% CAGR, regional outlook is shaped by vast unorganized dairy sectors attempting rapid industrialization. Total domestic milk production reached an impressive 239.30 million tons during 2023-24. [9] Massive raw material availability provides a theoretical foundation for immense future hydrolysis capacity.
FMI’s report includes granular tracking of evolving Asian nutritional landscapes. Japan presents high-value opportunities for functional marine peptides targeting mobility enhancement in older populations. South Korea demonstrates strong appetite for imported collagen supplements utilized in advanced cosmetic nutrition applications. Localized functional innovation dictates long-term commercial success.
Latin America Protein Hydrolysate Ingredient Market Analysis
Regional consumption favors cost-effective plant-based options integrated into mainstream functional foods. Macroeconomic volatility often suppresses demand for premium imported dairy peptides. Local manufacturers prioritize affordable soy and pea fractions to fortify staple bakery and beverage products. Expanding regional sports nutrition sectors provide isolated pockets of high-value whey peptide demand. Multi-national brands frequently utilize regional hubs to supply surrounding territories with standard hydrolyzed whey protein blends.
- Brazil: A 7.3% CAGR reflects growing middle-class engagement with functional fitness lifestyles. Domestic processors upgrade manufacturing capabilities to supply local sports nutrition brands with affordable peptide blends. Favorable agricultural output ensures consistent raw material access for emerging plant-based hydrolysis operations.
FMI’s report includes detailed supply chain mapping across major South American economies. Argentina offers strong potential for meat-derived peptide manufacturing given massive beef processing scale. Chile serves as a critical global source for marine protein hydrolysate fractions. Export competitiveness relies on favorable currency valuations.
Competitive Aligners for Market Players
According to FMI's estimates, successful ingredient suppliers maintain absolute control over complex regulatory approval pathways. Securing official recognition for specific peptide fractions creates formidable competitive moats against generic challengers. Deep regulatory expertise allows premium suppliers to command superior contract pricing. FrieslandCampina Ingredients recently secured widespread European Union approval for Hyvital Whey HA 300 usage in infant formulas. [10] Official certification guarantees long-term volume commitments from risk-averse multinational nutrition brands.
Manufacturing scale dictates survival during periodic commodity margin compression. Massive integrated cooperatives cross-subsidize expensive hydrolysis research using profits from standard bulk dairy commodities. Mid-tier independent processors lack financial resilience required to survive prolonged raw material price spikes. Consolidation inevitably absorbs smaller independent facilities holding valuable specialized enzymatic patents.
Application support capabilities increasingly define vendor selection criteria. Procurement teams demand customized processing solutions rather than off-the-shelf bulk powders. Suppliers must provide extensive pilot-plant testing facilities to help downstream brands resolve critical flavor masking challenges. Dedicated technical service teams transform transactional raw material vendors into indispensable strategic formulation partners.
Recent Developments
Report includes full coverage of key trends from competitive benchmarking. Some recent developments covered in reports:
- In November 2024, Arla Foods Ingredients received formal regulatory clearance to formally acquire Volac Whey Nutrition business. [2]
- In February 2024, Roquette expanded functional capabilities by launching NUTRALYS H85 pea protein. [4]
Key Players in Protein Hydrolysate Ingredient Market
- Arla Foods Ingredients
- FrieslandCampina Ingredients
- Roquette Frères
- Darling Ingredients
- Glanbia Nutritionals
- Kerry Group
- Fonterra Co-operative Group
- Tate & Lyle
- Carbery Group
- AMCO Proteins
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 770.7 million (2026) to USD 1,452.8 million (2036), at a CAGR of |
| Market Definition | Sector encompasses enzymatically cleaved protein ingredients engineered for enhanced digestibility and hypoallergenic performance. |
| Ingredient Type Segmentation | Milk, Plant, Marine, Meat |
| Form Segmentation | Powder, Liquid |
| Application Coverage | Infant Nutrition, Clinical Nutrition, Sports Nutrition, Functional Beverages |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40 plus countries |
| Key Companies Profiled | Arla Foods Ingredients, FrieslandCampina Ingredients, Roquette Frères, Darling Ingredients, Glanbia Nutritionals |
| Forecast Period | 2026 to 2036 |
| Approach | Volume-based modeling correlates hospital malnutrition rates with clinical formula manufacturing outputs. |
Protein Hydrolysate Ingredient Market Analysis by Segments
Ingredient Type
- Milk
- Plant
- Marine
- Meat
Form
- Powder
- Liquid
Region
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] USDA. (2025). EconStats Market Information System, US whey protein concentrate 2024.
- [2] Arla Foods Ingredients. (2024). Arla Foods moves forward with acquisition of Volac Whey Nutrition.
- [3] Arla Foods Ingredients. (2024). New medical nutrition ingredient combines high protein with best taste.
- [4] Roquette Frères. (2024). Extension NUTRALYS pea protein range.
- [5] PMC. (2025). Hospital malnutrition risk prevalence.
- [6] Darling Ingredients. (2024). Rousselot introducing Nextida shaping future of collagen peptides.
- [7] CDC. (2026). NCHS Press Release, US adults with food allergy.
- [8] Reuters. (2026). Lurpak maker Arla forecasts drop dairy prices after supply surge.
- [9] Press Information Bureau, Govt of India. (2024). India total milk production 2023-24.
- [10] FrieslandCampina Ingredients. (2024). FrieslandCampina secures EU approval for protein hydrolysate in infant formulas.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Commercial intelligence enables structured strategic decision making across mature clinical nutrition supply chains.
- Revenue forecasting tracks specialized ingredient valuation growth entering 2036.
- Growth opportunity mapping identifies high-value pathways across distinct processing formats.
- Segment modeling isolates specific demand vectors powering milk and plant-based formula adoption.
- Competition strategy assessment evaluates critical regulatory compliance capabilities across top-tier manufacturers.
- Regulatory impact analysis covers shifting global guidelines dictating acceptable infant formula peptide profiles.
- Output delivery provides interactive dashboard formats ensuring rapid executive insight extraction.
Frequently Asked Questions
How large is demand for Protein Hydrolysate Ingredient in global space in 2026?
Revenue reaches USD 770.7 million in 2026.
What will be market size of Protein Hydrolysate Ingredient in global space by 2036?
Revenue hits USD 1,452.8 million by 2036.
What is expected demand growth for Protein Hydrolysate Ingredient globally between 2026 and 2036?
Revenue scales at a 7.3% CAGR.
Which Ingredient Type is poised to lead global sales by 2026?
Milk captures 70% share via established massive whey supply chains.
How significant is role of Powder in driving Protein Hydrolysate Ingredient adoption in 2026?
Powder secures 35% share by extending critical transit viability.
What is driving demand in India?
Unprecedented domestic dairy modernization enables scalable regional extraction.
What compliance standards or regulations are referenced for India?
Local dairy development mandates heavily influence regional processing expansion.
What is India growth outlook in report?
India scales at 8.6% CAGR.
Why is Europe described as priority region in report?
High technical barriers protect lucrative premium pediatric formulation contracts.
What type of demand dominates in Europe?
Strictly regulated medical nutrition sourcing heavily influences regional volumes.
What is China growth outlook in report?
China expands at 8.5% CAGR.
Does report cover United States in regional analysis?
Yes, United States sits within North America.
What are sources referred to for analyzing United States?
Analysis utilizes CDC food allergy prevalence metrics.
What is main demand theme linked to United States in region coverage?
Established clinical networks drive high-volume domestic medical consumption.
Does report cover United Kingdom in regional analysis?
Yes, United Kingdom falls under Europe.
What is main United Kingdom related demand theme in region coverage?
Local formulators increasingly demand specialized soluble inputs.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Dry formats ensure stability across extended international freight channels.
What is Protein Hydrolysate Ingredient and what is mainly used for?
Pre-digested peptide fractions provide rapid absorption for specialized clinical formulations.
What does Protein Hydrolysate Ingredient mean in report?
Scope strictly covers bulk business-to-business functional protein sales.
What is included in scope of Protein Hydrolysate Ingredient report?
Milk, plant, marine, and meat derived peptide ingredients.
What is excluded from scope of report?
Finished retail consumer supplements and completely intact concentrates.
What does market forecast mean on page?
Projections represent strategic revenue modeling based on verifiable baseline indicators.
How does FMI build and validate Protein Hydrolysate Ingredient forecast?
Volume-based tracking maps hospital malnutrition rates against commercial outputs.
What does zero reliance on speculative third party research mean here?
Primary corporate disclosures drive fundamental baseline revenue calculations.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type , 2026 to 2036
- Milk-Based
- Plant
- Marine
- Meat
- Milk-Based
- Y to o to Y Growth Trend Analysis By Ingredient Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Powder
- Liquid
- Powder
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Type
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Type
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Type
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Type
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Type
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Type
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Type
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Type
- By Form
- Competition Analysis
- Competition Deep Dive
- Arla Foods Ingredients
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- FrieslandCampina Ingredients
- Roquette Frères
- Darling Ingredients
- Glanbia Nutritionals
- Kerry Group
- Fonterra Co-operative Group
- Arla Foods Ingredients
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Type
- Figure 6: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Form
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Ingredient Type
- Figure 23: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Form
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Form
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Form
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Form
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Form
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

